Polycythemia Vera Market Summary
- The total Polycythemia Vera market size in the 7MM is approximately ~USD 2,000 million in 2025 and is projected to increase during the forecast period (2026–2036).
- In 2025, the Polycythemia Vera market size was highest in the United States among the 7MM, accounting for approximately ~USD 1,500 million, which is further expected to increase by 2036, owing to the launch of emerging therapies, assisted by an increase in the diagnosed prevalent population of Polycythemia Vera.
Polycythemia Vera Insights and Trends
- Most Polycythemia Vera patients are symptomatic, with approximately ~91,000 patients with JAK2 mutation in the United States in 2025.
- The cornerstone of treatment for Polycythemia Vera remains phlebotomy to maintain safe hematocrit levels, often combined with low-dose aspirin. High-risk patients may also receive cytoreductive therapy, such as hydroxycarbamide (hydroxyurea). Ruxolitinib (JAKAFI) and rRopeginterferon alfa-2b (BESREMi) are approved in the US, Europe, and Japan, with Ruxolitinib (JAKAFI) established as a standard second-line therapy since 2015. PEGASYS (peginterferon alfa-2a) received marketing authorization in 2024 and formal approval in the UK in 2025, building on prior off-label use.
- Since BESREMi's label is broad (first and subsequent lines of therapy), PharmaEssentia has more leeway in positioning BESREMi in diverse therapeutic situations. Given BESREMi's position upstream of JAKAFI in the NCCN recommendations, the drug is not a direct competitor for JAKAFI. Still, it has the potential to hinder the progression of patients to JAKAFI usage, which might affect JAKAFI's overall revenue.
- The patent expiration in mid-2028 might be the most significant obstacle to JAKAFI’s supremacy in the Polycythemia Vera market. To address this, Incyte introduced the LIMBER (Leadership in MPNs and GVHD beyond Ruxolitinib) life-cycle management initiative, which was meant to explore different monotherapy and combination methods to enhance and expand therapies for patients with MPNs and GVHD.
- Other emerging Polycythemia Vera therapies in development include Bomedemstat (LSD1 inhibitor, Merck), Sapablursen (TMPRSS6 inhibitor, Ionis Pharmaceutical/Ono Pharmaceutical), Divesiran (TMPRSS6 inhibitor, Silence Therapeutics), PPMX-T003 (anti-TfR1, Perseus Proteomics), and DISC-3405 (anti-TMPRSS6, Disc Medicine), and others highlighting a growing pipeline of innovative treatments.
- Several other potential therapies for Polycythemia Vera are in the early stage of development, which include INCB160058 (Incyte), AG-236 (Agios Pharmaceuticals and Alnylam Pharmaceuticals, Phase I), VGT-1849A and VGT-1849B (Vanda Pharmaceuticals, NA), JAK2V617F (Prelude Therapeutics and Incyte, IND enabling), PN-8047 (Protagonist Therapeutics, IND enabling), and others.
Polycythemia Vera Market size and forecast (7MM)
- 2025 Polycythemia Vera Market Size: ~USD 2,000 million
- 2036 Projected Polycythemia Vera Market Size: ~USD 5,600 million
- Polycythemia Vera Growth Rate (2026–2036): 8.9% CAGR
DelveInsight's ‘Polycythemia Vera (PV) Market Insights, Epidemiology and Market Forecast – 2036’ report delivers an in-depth understanding of the Polycythemia Vera, historical and forecasted epidemiology, as well as the Polycythemia Vera market trends in the United States, EU4 (Germany, Spain, Italy, and France) and the United Kingdom, and Japan.
The Polycythemia Vera market report delivers a comprehensive analysis of the current treatment landscape, including standards of care, clinical practices, and evolving therapeutic algorithms. It evaluates, Polycythemia Vera patient burden trends, revenue & market share dynamics, peak patient share & therapy uptake analysis, and provides an in-depth market size assessment, and growth rate projections (Historical & Forecast 2022–2036) across global regions. The report highlights key unmet medical needs in Polycythemia Vera and maps the competitive and clinical landscape to uncover high-value opportunities, providing a clear outlook on future market growth potential.
Scope of the Polycythemia Vera Market Report | |
|
Study Period |
2022–2036 |
|
Historical Year |
2022–2025 |
|
Forecast Period |
2026–2036 |
|
Base Year |
2026 |
|
Geographies Covered |
|
|
Polycythemia Vera Market CAGR (Study period) |
8.9% (2026–2036) |
|
Polycythemia Vera Epidemiology Segmentation Analysis |
Patient Burden Assessment
|
|
Polycythemia Vera Companies |
|
|
Polycythemia Vera Therapies |
|
|
Polycythemia Vera Market |
Segmented by
|
|
Analysis |
|
Factors Driving Polycythemia Vera Market Growth
Rising Prevalence of Polycythemia Vera
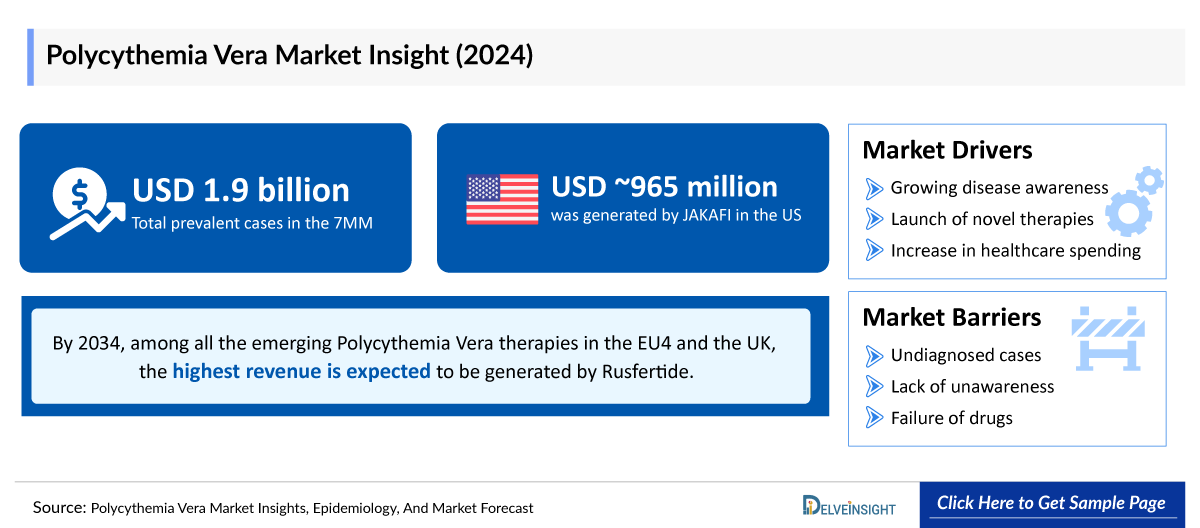
One of the primary drivers of the Polycythemia Vera market is the increasing prevalence of the disease, particularly among the aging population. Polycythemia Vera is more commonly diagnosed in individuals over the age of 60, and as global life expectancy rises, the patient pool continues to expand. This growing disease burden directly increases demand for effective diagnostic tools and long-term treatment options, thereby fueling market growth.
Increasing Awareness and Early Diagnosis
Improved awareness among healthcare professionals and patients has significantly contributed to early detection and diagnosis of Polycythemia Vera. Educational initiatives and advocacy by organizations have enhanced recognition of symptoms and risk factors, leading to higher diagnosis rates. As more cases are identified earlier, the need for therapeutic interventions increases, expanding the overall market size.
Advancements in Diagnostic Technologies
Technological advancements in diagnostic methods, including genetic testing for JAK2 mutations and improved blood profiling techniques, have revolutionized Polycythemia Vera diagnosis. These innovations allow for more accurate and timely identification of the disease, reducing misdiagnosis and enabling earlier treatment initiation. This, in turn, drives demand for both diagnostics and therapeutics in the Polycythemia Vera market.
Emergence of Targeted Therapies
The development of targeted therapies, particularly JAK inhibitors, has transformed the treatment landscape of Polycythemia Vera. These therapies address the underlying molecular mechanisms of the disease, offering better efficacy and improved patient outcomes compared to traditional treatments. The success of such innovative drugs continues to attract investment and accelerate market expansion.
Growing Focus on Personalized Medicine
The shift toward personalized and precision medicine is another critical factor driving the PV market. Therapies tailored to individual genetic profiles and disease characteristics are gaining traction, improving treatment effectiveness and minimizing adverse effects. This trend supports the development of novel treatment approaches and enhances patient compliance, contributing to sustained market growth.
Robust Research and Development Activities
Increasing investment in research and development by pharmaceutical and biotechnology companies is significantly boosting the PV market. Ongoing clinical trials and pipeline therapies aimed at improving efficacy, safety, and dosing convenience are expanding treatment options. The introduction of novel agents, including advanced biologics and next-generation therapies, is expected to further strengthen the market outlook.
Favorable Healthcare Policies and Reimbursement Support
Supportive reimbursement frameworks and increased healthcare spending across both developed and emerging economies are facilitating patient access to advanced therapies. Government initiatives and funding for rare disease treatment also play a crucial role in market growth by encouraging innovation and ensuring affordability of treatment options.
Expanding Therapeutic Pipeline and Drug Approvals
The Polycythemia Vera market is witnessing a strong pipeline of emerging therapies, including interferons, monoclonal antibodies, and novel small molecules. Recent and upcoming drug approvals are expected to enhance treatment efficacy and provide alternative options for patients resistant to existing therapies. This continuous influx of new products is a major growth catalyst for the market.
Increasing Demand for Disease Management and Long-Term Care
Since PV is a chronic condition with no definitive cure, long-term disease management is essential. Patients require ongoing monitoring, treatment, and supportive care to prevent complications such as thrombosis and disease progression. This continuous demand for healthcare services and medications contributes significantly to the expansion of the PV market.
Shift Toward Innovative Treatment Modalities
There is a growing emphasis on therapies that offer improved convenience, such as less frequent dosing and better tolerability. Emerging platforms like RNA-based therapies and advanced biologics are gaining attention for their potential to improve patient adherence and outcomes. This shift toward innovation is expected to create new growth opportunities within the market.
Polycythemia Vera Understanding and Treatment Algorithm
Polycythemia Vera Overview and Diagnosis
Polycythemia Vera is a rare condition characterized by the progressively increased number of red blood cells in the bloodstream, white blood cells (WBC), and platelets also increase in number in the affected people. Polycythemia Vera is the most common myeloproliferative neoplasm (MPN). Out of all the MPNs, Polycythemia Vera is the most common and the only one in which there is an increase in RBC production. Polycythemia Vera usually occurs at an elderly age, and patients are at a higher risk than the usual percentage, as they are more prone to risks and other comorbidities.
Polycythemia Vera Diagnosis
The diagnosis of Polycythemia Vera is based on results from tests assessing elevations in blood counts and the presence of molecular mutations associated with the disease. The initial evaluation should include a focused clinical history, physical examination, hematocrit concentration, and count of RBC, WBC, platelets, serum EPO level, and cytogenetic abnormalities. The patient's journey begins with a referral to a hematologist. However, the current approach to diagnosing these patients is quite similar, as most developing countries establish a patient’s diagnosis journey using WHO criteria, which is based on a composite assessment of clinical and laboratory features, including JAK2 mutation status and serum erythropoietin level. However, when it comes to analyzing the real-world scenario in varying geographies, there are some differences in diagnostic criteria and risk stratification that other organizations in certain European countries have proposed.
For example, even though the WHO classification considers histology useful in distinguishing Polycythemia Vera from other myeloproliferative neoplasms (MPNs), the British Society for Haematology Guideline has also cited several studies that have reported high rates of failure to reach a histological diagnosis in patients with Polycythemia Vera.
Further details are provided in the report.
Polycythemia Vera Treatment
Based on real-world pattern analysis, patients suffering from Polycythemia Vera receive treatment with certain drugs (myelosuppressive drugs) that suppress the formation of blood cells by the marrow. A chemotherapy drug, hydroxyurea, is most often used, along with another chemotherapy drug known as busulfan. Other drugs, such as chlorambucil and radioactive phosphorus, have also been used. JAKAFI and BESREMi are the two FDA-approved drugs for treating Polycythemia Vera.
Although medication adherence can be elusive, it has been found that the patients who were prescribed injectable cytoreductive drugs had lower adherence rates than their counterparts taking oral formulations. There are many factors when it comes to lower adherence to therapies, for instance, when patients are taking too many therapies together, facing side effects, having cost concerns, and facing issues with dosing regimens.
Further details are provided in the report.
Polycythemia Vera Unmet Needs
The section “unmet needs of Polycythemia Vera” outlines the critical gaps between the current state of patient care, diagnosis, and the ideal & effective management of the disease. It highlights the obstacles experienced by patients, clinicians, and researchers and identifies potential solutions for future progress.
- Ongoing disease burden due to poor rates of sustained Hematocrit (Hct) control
- Psychosocial and access challenges linked to polycythemia vera rarity and low awareness
- Delayed diagnosis despite established criteria and clear treatment triggers
- Deterioration in quality of life driven by treatment burden and symptom persistence, and others…..
Comprehensive unmet needs insights in Polycythemia Vera and their strategic implications are provided in the full report.
Polycythemia Vera Epidemiology
Key Findings from Polycythemia Vera Epidemiological Analysis and Forecast
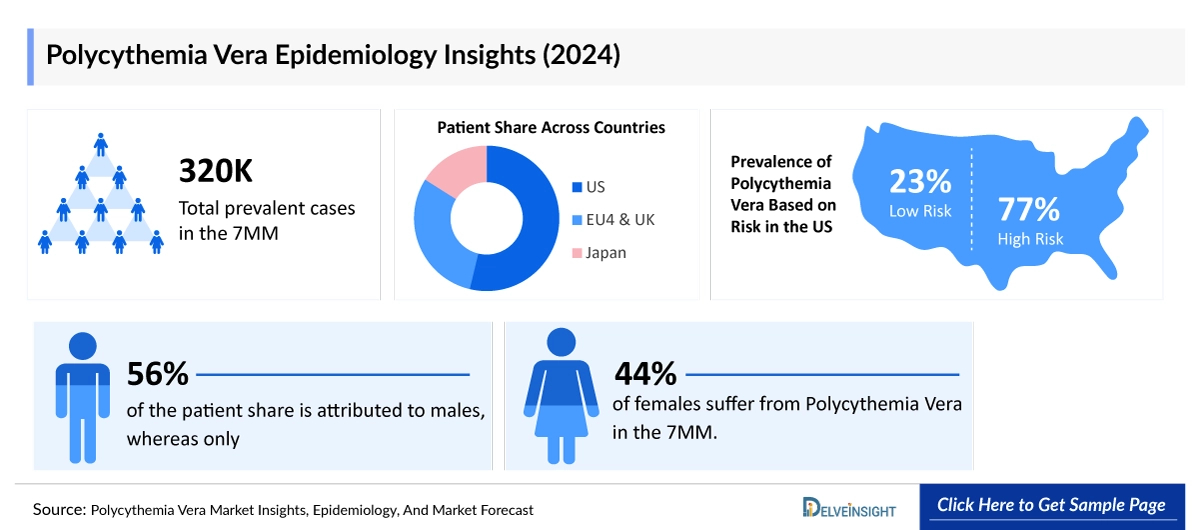
- The United States contributed to the largest prevalent population of Polycythemia Vera, acquiring ~55% of the 7MM in 2025. Whereas EU4 and the UK, and Japan accounted for around ~30% and ~15% of the total population share, respectively, in 2025.
- The total number of prevalent cases of Polycythemia Vera in the United States was around ~158,700 cases in 2025.
- Among the EU4 and the UK, Germany accounted for the largest number of Polycythemia Vera cases based on symptoms, whereas the UK accounted for the lowest number of cases in 2025.
- According to DelveInsight estimates, there were around ~63,000 cases of asymptomatic and ~95,000 cases of symptomatic Polycythemia Vera in the United States in 2025. The prevalence is projected to increase during the forecast period.
- In Japan, the age-specific data revealed that the highest number of people affected with Polycythemia Vera was found in the age group of 65-74 years, i.e., ~6,000 cases in 2025, while people who belonged to the age group 0–34 years, i.e., ~800 cases, were the least affected.
- In 7MM, approximately ~60% of the patient share is attributed to males, whereas only ~40% of females suffer from Polycythemia Vera.
Polycythemia Vera Drug Chapters & Competitive Analysis
The Polycythemia Vera drug chapter provides a detailed, market-focused review of approved therapies and the emerging pipeline across Phase I–III clinical trials. It covers the mechanism of action, clinical trial data, regulatory approvals, patents, collaborations, and strategic partnerships for each therapy, along with their advantages, limitations, and recent developments. This section offers critical insights into the Polycythemia Vera treatment landscape, supporting market assessment, competitive analysis, and growth forecasting for the Polycythemia Vera therapeutics market.
Approved Therapies for Polycythemia Vera
Ruxolitinib (JAKAFI/JAKAVI): Incyte/Novartis
JAKAFI/JAKAVI is an oral inhibitor of the JAK 1 and JAK 2 tyrosine kinases. It is approved for treating adult patients resistant to or intolerant of hydroxyurea. Incyte’s flagship product, JAKAFI, received approval as a second-line therapy for Polycythemia Vera patients in December 2014 in the United States, followed by approvals in both the EU and Japan in 2015.
In December 2024, the American Society of Hematology (ASH) Annual Meeting and Exposition featured novel research on the long-term safety and efficacy of ruxolitinib for patients with polycythemia vera. The poster presentations in particular shed light on the superiority of ruxolitinib over hydroxyurea and how ruxolitinib can allow patients to rely less on corticosteroids.
Polycythemia Vera Competitive Landscape: Marketed Therapies | ||||||
|
Drug/Therapy |
Company |
Indication |
Molecule Type |
MoA |
RoA |
Marketed Region |
|
Ruxolitinib (JAKAFI/JAKAVI) |
Incyte/Novartis |
Patients with Polycythemia Vera who have had an inadequate response to or are intolerant of hydroxyurea |
Small molecule |
JAK inhibitor |
Oral |
US: 2014 EU: 2015 JP: 2015 |
|
Ropeginterferon alfa-2b (BESREMi) |
PharmaEssentia/AOP Orphan Pharmaceuticals |
Polycythemia Vera patients, regardless of their treatment history |
Recombinant interferon alpha subtype 2b (IFN-a2b) protein |
Binds to the interferon alfa receptor (IFNAR) |
SC |
US: 2021 EU: 2019 JP: 2023 |
|
Peginterferon alfa-2a (PEGASYS) |
pharmaand GmbH |
Polycythemia Vera |
Type 1 interferon |
Antiviral and anti-proliferative activities |
SC |
EU: 2024 UK: 2025 |
Polycythemia Vera Pipeline Analysis
Rusfertide (PTG-300): Protagonist Therapeutics
Rusfertide is a novel injectable synthetic mimetic of the natural hormone hepcidin that offers greater potency, solubility, and stability, which translates to better in vivo pharmacokinetics and pharmacodynamics characteristics and manufacturability in comparison to the natural hormone. In December 2024, the final results of rusfertide from the Phase II REVIVE trial were presented at the ASH (4559) meeting. The data demonstrated that rusfertide, when added to therapeutic phlebotomy with or without cytoreductive therapy, effectively controlled erythrocytosis, provided long-term durable control of hematocrit, and reduced the need for therapeutic phlebotomy in patients with Polycythemia Vera.
According to Protagonist Therapeutics’ corporate presentation published in January 2026, the company filed an NDA for rusfertide in 2025, with an anticipated early launch in H2 2026.
Polycythemia Vera Competitive Landscape: Emerging Therapies | |||||||
|
Drug Name |
Company |
Highest Phase |
Indication |
RoA |
MoA |
Molecule Type |
Anticipated Launch in the US |
|
Rusfertide (PTG-300) |
Protagonist Therapeutics and Takeda Pharmaceuticals |
III |
Polycythemia vera |
SC |
Targeting the underlying mechanism of iron dysregulation |
Hepcidin mimetic peptide |
2026 |
|
DISC-3405 |
Disc Medicine’s |
II |
Polycythemia vera |
SC |
TMPRSS6 protein inhibitors |
DISC-3405 |
Information is available in the full report |
|
GLB-001 |
Hangzhou GluBio Pharmaceutical |
I |
Relapsed, refractory, or intolerant polycythemia vera |
Oral |
Casein Kinase 1 Alpha (CK1α) protein inhibitor |
Small molecule |
Information is available in the full report |
|
Note: Launch insights are provisional and may change with future report updates or the occurrence of major key catalysts. | |||||||
Polycythemia Vera Key Players, Market Leaders and Emerging Companies
- Protagonist Therapeutics and Takeda Pharmaceuticals
- Italfarmaco
- Merck (Imago BioSciences)
- Ionis Pharmaceuticals and Ono Pharmaceutical
- Disc Medicine’s, and others
Polycythemia Vera Drug Updates
- In the J.P. Morgan Healthcare Conference 2026 presentation, Incyte stated that ruxolitinib extended-release (XR), a once-daily (QD) formulation for Polycythemia Vera, is expected to receive regulatory approval and be launched around mid-2026.
- Ionis Pharmaceuticals announced at the J.P. Morgan Healthcare Conference 2026 that it plans to initiate a Phase III clinical trial of sapablursen for the treatment of Polycythemia Vera in 2026.
- In August 2025, Vanda announced that the FDA had granted Orphan Drug Designation for VGT-1849B, a selective peptide nucleic acid-based JAK2 inhibitor for the treatment of Polycythemia Vera.
- According to Italfarmaco's pipeline activity, the company anticipates the launch of DUVYZAT in 2027 for the treatment of Polycythemia Vera.
Polycythemia Vera Market Outlook
Polycythemia Vera treatment in the US is entering a new era with changing dynamics. Therapeutic options for Polycythemia Vera are limited, and no cure is available. The treatment landscape was dominated by legacy, generic therapies such as Hydroxyurea, second-generation interferon, and Phlebotomy until the approval of BESREMi as both the first and second line of treatment, and JAKAFI in the second line of treatment only; options for third-line treatment are still limited. Adding to that, Low-dose aspirin and phlebotomy are recommended as first-line treatment options for patients at low risk of thrombotic events. Cytoreductive therapy (usually hydroxyurea or interferon alpha) is recommended for high-risk patients. Polycythemia Vera treatment is still inadequate, as approximately 30% of Hydroxyurea-treated patients become resistant/intolerant.
The current market has been segmented into different commonly used therapeutic classes based on the prevailing treatment pattern across the 7MM, which presents minor variations in the overall prescription pattern. JAK inhibitor, Interferons, Hepcidin mimetic, TMPRSS6 inhibitor, CK1a, BCL-2 inhibitor, and LSDI inhibitor are the major classes covered in the forecast model.
Key players involved in developing targeted therapies to treat Polycythemia Vera include Protagonist Therapeutics and Takeda Pharmaceuticals (rusfertide), Merck (bomedemstat), Italfarmaco (givinostat), Ionis Pharmaceuticals and Ono Pharmaceutical (sapablursen), Silence Therapeutics (Divesiran (SLN124)), Perseus Proteomics (PPMX-T003), Disc Medicines (DISC-3405), and others are evaluating their lead candidates in different stages of clinical development, respectively. They aim to investigate their products for the treatment of Polycythemia Vera.
Key findings from Polycythemia Vera Market Forecast and Analysis
- The total Polycythemia Vera market size in the 7MM is approximately ~USD 2,000 million in 2025 and is projected to increase during the forecast period (2026-2036).
- Among EU4 and the UK, Germany accounts for the maximum market size in 2025, while the United Kingdom occupies the bottom of the ladder in 2025.
- In 2025, among the current therapies for Polycythemia Vera, the largest revenue was generated by JAKAFI, i.e., ~USD 1,000 million in the United States.
- By 2036, among all the emerging therapies in the EU4 and the UK, the highest revenue is expected to be generated by Rusfertide.
Further details will be provided in the report….
Polycythemia Vera Drug Class/Insights
The existing Polycythemia Vera treatment is mainly dominated by classes such as antimetabolites, platelet-reducing agents, Interferons, and JAK inhibitors.
JAK inhibitors have been developed following the discovery of the JAK2V617F in 2005 as the driver mutation of most non-BCR-ABL1 MPNs. A decade after the beginning of clinical trials, only one JAK inhibitor, JAKAFI, has been approved by the US Food and Drug Administration for hydroxyurea-resistant or intolerant Polycythemia Vera patients since 2014. JAKAFI competes with platelet-reducing agents (Anagrelide), Interferon alpha, and BESREMi in the second line of treatment. Even though there are safety concerns regarding JAK inhibitors, we cannot misconstrue the strong uptake of JAKAFI in the already treated patients with this blood disorder.
Moving on to Interferon’s, BESREMi is not the first one being used in these patients, though it is the first approved interferon for Polycythemia Vera patients. For years, PEGASYS has been commercially available, which is present all along. PEGASYS is a form of pegylated interferon that is used off-label by patients living with a myeloproliferative neoplasm. However, many patients have dealt with access and reimbursement issues, as it is accessed off-label.
Moreover, the upcoming treatment landscape is poised to see further expansion after the emergence of new classes such as iron modulator, LSD1 inhibitor, Pan-HDAC inhibitor, TMPRSS6 inhibitor, TfR targeting, BCL-2 inhibitor, Immunostimulant, CK1a, and others.
Further details will be provided in the report….
Polycythemia Vera Drug Uptake
This section focuses on the uptake rate of potential Polycythemia Vera drugs expected to be launched in the Polycythemia Vera market during 2022–2036. For example, for Rusfertide, although the FDA rescinded its Breakthrough Designation, we expect the drug uptake to be medium-fast with a probability-adjusted peak share of 5.7% in the first line and second line, and years to the peak are expected to be 7 years from the year of launch. The drug was proven to be better among all the other therapies in terms of safety and efficacy.
Polycythemia Vera Therapies Price Scenario & Trends
Pricing and analogue assessment of Polycythemia Vera therapies highlights evolving price dynamics structures. This section summarizes the cost of approved treatments, closest and most appropriate analogue selection for emerging therapies, and understanding of how pricing influences market access, adherence, and long-term uptake.
Pricing of Polycythemia Vera Approved Drugs
Recommended starting dose of BESREMi is 100 mcg by subcutaneous injection every 2 weeks (50 mcg if receiving hydroxyurea). Further increasing the dose by 50 mcg every 2 weeks (up to a maximum of 500 mcg) until hematological parameters are stabilized. Hence, on this basis we have assumed 22 syringes would be needed in a year. PharmaEssentia launched BESREMi at a wholesale acquisition cost (WAC) of USD 6,988 per syringe, translating to an annual cost of USD 153,736
Further details are provided in the final report….
Industry Experts and Physician Views for Polycythemia Vera
To keep up with Polycythemia Vera market trends, we take Key Opinion Leaders (KOLs) and Subject Matter Experts (SMEs) opinions working in the domain through primary research to fill the data gaps and validate our secondary research. Industry Experts were contacted for insights on Polycythemia Vera emerging therapies, evolving treatment landscape, patient adherence to conventional therapies, therapy switching trends, drug adoption and uptake, accessibility challenges, and epidemiology and real-world prescription patterns in Polycythemia Vera, including MD, PhD, Instructor, Postdoctoral Researcher, Professor, Researcher, and others.
Delveinsight’s analysts connected with 20+ KOLs to gather insights; however, interviews were conducted with 10+ KOLs in the 7MM. Centers such as MD Anderson Cancer Center, Texas, from UT Southwestern Medical Center in Dallas, Cancer Research UK Barts Centre in London, MD Anderson Cancer Center, etc., were contacted. Their opinion helps understand and validate current and emerging therapy treatment patterns or Polycythemia Vera market trends. This will support the clients in potential upcoming novel treatments by identifying the overall scenario of the market and the unmet needs.
What KOLs are saying on Polycythemia Vera Patient Trends? | |
|
Region |
Key Opinion Leaders (KOLs) and Subject Matter Experts (SMEs) |
|
United States |
“The approval of JAKAFI represents an important advance for patients with uncontrolled polycythemia vera. For the first time, we can provide these patients a treatment that has been shown to provide effective and consistent control of their blood counts and reduce spleen volume.” |
|
Germany |
“Polycythemia Vera patients could benefit from a novel treatment option that effectively manages their condition without causing serious adverse effects. In the Phase I portion of the SANRECO study, Divesiran substantially reduced the need for phlebotomy and lowered hematocrit levels following infrequent dosing in a range of Polycythemia Vera patients. The long duration of effect and clean safety/tolerability profile were impressive. These data are very exciting and support further development of Divesiran in polycythemia vera.” |
Polycythemia Vera Qualitative Analysis: SWOT and Conjoint Analysis
We perform qualitative and market Intelligence analysis using various approaches, such as SWOT analysis and conjoint analysis. In the SWOT analysis of Polycythemia Vera, strengths, weaknesses, opportunities, and threats in terms of disease diagnosis, patient awareness, patient burden, competitive landscape, cost-effectiveness, and geographical accessibility of therapies are provided.
Conjoint analysis analyzes emerging therapies based on relevant attributes such as safety, efficacy, frequency of administration, route of administration, and order of entry. Scoring is given based on these parameters to analyze the effectiveness of therapy.
The team of analysts analyzes promising emerging therapies based on relevant attributes such as safety, efficacy, frequency of administration, route of administration, and order of entry. In efficacy, the trial’s primary and secondary outcome measures are evaluated, whereas the therapies’ safety is evaluated, wherein the acceptability, tolerability, and adverse events are majorly observed. In addition, the scoring is also based on the route of administration, order of entry, probability of success, and the addressable patient pool for each therapy. According to these parameters, the final weightage score and the ranking of the emerging therapies are decided.
Scope of the Polycythemia Vera Market Report
- The report covers a segment of key events, an executive summary, a descriptive overview of Polycythemia Vera, explaining its causes, signs and symptoms, pathogenesis, and currently available treatments.
- Comprehensive insight has been provided into the epidemiology segments and forecasts, the future growth potential of the diagnosis rate, and disease progression along treatment guidelines.
- Additionally, an all-inclusive account of both the current and emerging treatments, along with the elaborative profiles of late-stage and prominent therapies, will have an impact on the current treatment landscape.
- A detailed review of the Polycythemia Vera market, historical and forecasted market size, market share by therapies, detailed assumptions, and rationale behind our approach is included in the report, covering the 7MM drug outreach.
- The report provides an edge while developing business strategies by understanding trends through SWOT analysis and expert insights/KOL views, patient journey, and treatment preferences that help in shaping and driving the 7MM Polycythemia Vera market.
Polycythemia Vera Market Report Insights
- Polycythemia Vera patient population forecast
- Polycythemia Vera therapeutics market size
- Polycythemia Vera pipeline analysis
- Polycythemia Vera market size and trends
- Polycythemia Vera market opportunity (current and forecasted)
Polycythemia Vera Market Report Key Strengths
- Epidemiology-based (Epi-based) bottom-up forecasting
- Artificial Intelligence (AI)-enabled market research report
- 11-year forecast
- Polycythemia Vera market outlook (North America, Europe, Asia-Pacific)
- Patient Burden trends (by geography)
- Polycythemia Vera Treatment addressable Market (TAM)
- Polycythemia Vera) Competitive Landscape
- Polycythemia Vera major companies Insights
- Polycythemia Vera Price trends and analogue assessment
- Polycythemia Vera Therapies Drug Adoption/Uptake
- Polycythemia Vera Therapies Peak Patient Share analysis
Polycythemia Vera Market Report Assessment
- Polycythemia Vera Current treatment practices
- Polycythemia Vera Unmet needs
- Polycythemia Vera Clinical development Analysis
- Polycythemia Vera emerging drugs product profiles
- Polycythemia Vera Market attractiveness
- Polycythemia Vera Qualitative analysis (SWOT and conjoint analysis)
Frequently Asked Questions from Polycythemia Vera Market Report
Polycythemia Vera Market Insights
- What was the Polycythemia Vera market size, the market size by therapies, market share (%) distribution in 2025, and what would it look like by 2036? What are the contributing factors for this growth?
- What are the anticipated pricing variations among different geographies for the emerging therapies in the future?
- What can be the future treatment paradigm of Polycythemia Vera?
- What are the disease risks, burdens, and unmet needs of Polycythemia Vera? What will be the growth opportunities across the 7MM concerning the patient population with Polycythemia Vera?
- Who is the major future competitor in the market, and how will the competitors affect their market share?
- What are the current options for the treatment of Polycythemia Vera? What are the current guidelines for treating Polycythemia Vera in the US, Europe, and Japan?
Reasons to Buy the Polycythemia Vera Market Report
- The Polycythemia Vera Market report will help in developing business strategies by understanding the latest trends and changing treatment dynamics driving the Polycythemia Vera market.
- Bottom up forecasting builds from the affected population to product forecasts, delivering a robust, data driven approach ideal for new therapies and novel classes.
- Insights on patient burden/disease incidence, evolution in diagnosis, and factors contributing to the change in the epidemiology of the disease during the forecast years.
- Understand the existing Polycythemia Vera Market opportunities in varying geographies and the growth potential over the coming years.
- Identifying strong upcoming Polycythemia Vera companies in the Polycythemia Vera Market will help devise strategies to help get ahead of competitors.
- Detailed analysis and ranking of class-wise potential current and emerging therapies under the conjoint analysis section to provide visibility around leading classes.
- To understand KOLs’ perspectives on the accessibility, acceptability, and compliance-related challenges of existing treatment to overcome barriers in the future.
- Detailed insights on the unmet needs of the existing Polycythemia Vera Market so that the upcoming Polycythemia Vera companies can strengthen their development and launch strategy.
- This Artificial Intelligence (AI) enabled report summarize and simplify complex datasets within the report into clear, actionable insights for stakeholders, investors, and healthcare providers, enabling faster, data driven decisions.
-Market-Vera-(PV)-Market-.png&w=256&q=75)