Vascular Dementia Market
- Vascular dementia is an etiological category of dementia characterized by severe cognitive impairment resulting from ischemic or hemorrhagic stroke or hypoperfusion affecting brain regions important for memory, cognition, and behavior.
- In 2023, the market size of Vascular Dementia was highest in the US among the 7MM accounting for approximately USD 1,384 million that is further expected to increase at a compound annual growth rate (CAGR) of 4%.
- The current treatment for Vascular Dementia includes anti-dementia drugs, antihypertensives and cholesterol-lowering drugs, anticoagulants and antiplatelet, antidepressants, and others. Antihypertensives and cholesterol-lowering drugs accounted for the highest market share of 35% in 2023, in the 7MM.
- Vascular Dementia approximately accounted for 12 million diagnosed prevalent cases in 2023 in the 7MM, the treatment market of Vascular Dementia lacks approved therapy specific to Vascular Dementia treatment.
- The vascular dementia market in the 7MM is driven by an aging population, increasing prevalence due to higher rates of cardiovascular diseases and strokes, and advancements in research and diagnostic tools. Growing awareness and earlier diagnosis, rising healthcare expenditures, and investments in dementia care also contribute to market growth
- Pharmacological studies of Charsire Biotechnology Corporation's BAC have that it may improve dementia conditions by reducing pro-inflammatory cytokines in the brain, a mechanism distinct from the commonly hypothesized roles of amyloid beta and Tau protein. This emerging therapy has the potential to create a significant shift in the Vascular Dementia market size.
DelveInsight’s “Vascular Dementia– Market Insights, Epidemiology, and Market Forecast – 2034” report delivers an in-depth understanding of Vascular Dementia, historical and forecasted epidemiology as well as the Vascular Dementia market trends in the United States, EU4, and the UK (Germany, France, Italy, Spain) and the United Kingdom, and Japan.
The Vascular Dementia market report provides current treatment practices, emerging drugs, market share of individual therapies, and current and forecasted 7MM Vascular Dementia market size from 2020 to 2034. The Report also covers current Vascular Dementia treatment practices, market drivers, market barriers, SWOT analysis, reimbursement and market access, and unmet medical needs to curate the best of the opportunities and assess the underlying potential of the market.
Geography Covered
- The United States
- EU4 (Germany, France, Italy, and Spain) and the United Kingdom
- Japan
Study Period: 2020–2034
Disease Understanding and Treatment Algorithm
Vascular Dementia Overview
Vascular dementia, sometimes called vascular cognitive impairment, is a group of conditions that cause a decline in cognitive skills. Vascular Dementia is the second most common form of dementia after Alzheimer’s disease. Blockage or lack of blood flow to the brain is the cause of Vascular Dementia. Reduced blood flow to the brain deprives it of much-needed oxygen. Lack of oxygen and blood can damage the brain, even in a short period of time. People with Vascular Dementia experience problems with reasoning, judgment, and memory. Symptoms can occur suddenly or may be mild and go unnoticed at first.
Several conditions and factors can damage blood vessels. Factors such as age, smoking, and history of stroke or heart attack, hardened arteries, contributing conditions like diabetes, lupus, high blood pressure, and an abnormal heart rhythm all affect how blood flows through the body.
Some people with vascular dementia may have another type of dementia too, like Alzheimer’s disease. This is known as ‘mixed dementia’. Vascular dementia most commonly occurs in people between the ages of 60 and 75. Men seem to be more vulnerable than women.
Vascular Dementia Diagnosis
Diagnosing vascular dementia involves a comprehensive evaluation that includes medical history, physical examination, and various diagnostic tests. Initially, healthcare providers assess symptoms such as cognitive decline, memory loss, and difficulties with reasoning or problem-solving. Imaging techniques, such as MRI or CT scans, are crucial for identifying brain damage related to vascular issues, such as stroke or small vessel disease. Neuropsychological testing helps to evaluate cognitive function and differentiate vascular dementia from other types of dementia. Blood tests may be conducted to rule out other potential causes of cognitive impairment. Accurate diagnosis requires a thorough assessment to distinguish vascular dementia from Alzheimer’s disease and other cognitive disorders, ensuring appropriate treatment and management strategies.
Further details related to diagnosis are provided in the report…
Vascular Dementia Treatment
The treatment of vascular dementia focuses on managing symptoms, improving quality of life, and addressing the underlying vascular conditions that contribute to cognitive decline. This often involves a multifaceted approach, including medications to manage risk factors such as hypertension, diabetes, and high cholesterol, which are crucial in slowing the progression of the disease. Anticoagulants or antiplatelet agents may be prescribed to prevent further strokes. Cognitive and behavioral therapies can help patients cope with memory loss and other cognitive impairments. Lifestyle modifications, such as a healthy diet, regular exercise, and smoking cessation, are also recommended to improve overall vascular health. Supportive care, including counseling and assistance with daily activities, plays a vital role in enhancing the well-being of individuals with vascular dementia.
Further details related to treatment are provided in the report…
Vascular Dementia Epidemiology
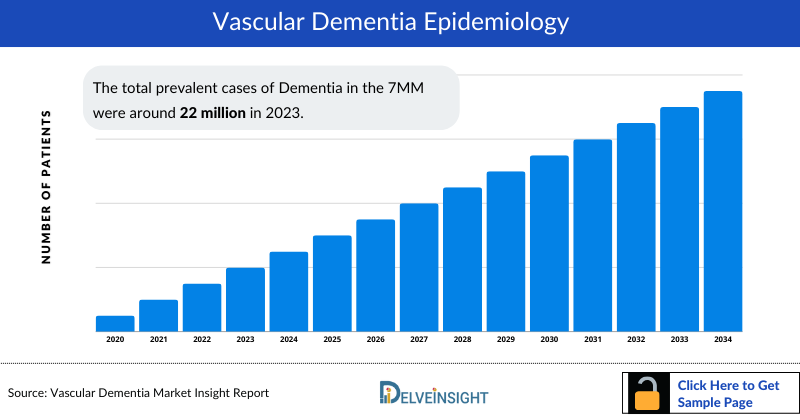
As the market is derived using a patient-based model, the Vascular Dementia epidemiology chapter in the report provides historical as well as forecasted epidemiology segmented by Prevalent Cases of Dementia, Diagnosed Prevalent Cases of Dementia, Diagnosed Prevalent Cases of Vascular Dementia, and Age-specific Diagnosed Prevalent Cases of Vascular Dementia, in the 7MM covering, the United States, EU4 countries (Germany, France, Italy, and Spain), the United Kingdom, and Japan from 2020 to 2034.
- The total prevalent cases of Dementia in the 7MM were around 22 million in 2023.
- The total diagnosed prevalent cases of Vascular Dementia in the United States were around 1 million cases in 2023.
- The United States contributed to the largest diagnosed prevalent population of Vascular Dementia, acquiring ~40% of the 7MM in 2023. Whereas, EU4 and the UK, and Japan accounted for around 32% and 28% of the total population share, respectively, in 2023.
- Among the EU4 countries, Germany accounted for the largest number of prevalent cases of Dementia (1,843 thousand) followed by Italy (1,365 thousand), whereas Spain accounted for the lowest number of cases (934 thousand) in 2023.
- In 2023, it was estimated that the highest number of diagnosed prevalent cases of vascular dementia in Japan was around 320 thousand in the 81–90 years age group.
- According to DelveInsight estimates, in 2023 there were approximately 3 million diagnosed prevalent cases of Dementia in Japan.
- The vascular dementia market has unmet needs including the lack of effective disease-modifying treatments and better diagnostic tools to differentiate it from other dementias. Current therapies primarily manage symptoms rather than address underlying causes. There is also a need for more research to develop targeted therapies and improved care strategies to enhance patient quality of life.
Vascular Dementia Drug Chapters
The drug chapter segment of the Vascular Dementia report encloses a detailed analysis of Vascular Dementia off-label drugs and late-stage (Phase-III and Phase-II) pipeline drugs. It also helps to understand the Vascular Dementia clinical trial details, expressive pharmacological action, agreements and collaborations, approval and patent details, advantages and disadvantages of each included drug, and the latest news and press releases.
Emerging Drugs
BAC: Charsire Biotechnology Corp.
BAC (also called CSTC1, CHARSIRE Trauma Complex 1), a component of soybean extraction developed by Charsire Biotechnology Corp, Taiwan, is under investigation to treat a variety of diseases, including Alzheimer’s disease and Vascular Dementia. BAC is a vapor fraction from seeds of Glycine max (L.) BAC in its studies showed to promote the proliferation of neuroblastoma IMR-32 cells, and this observation may correlate to the improvement of the cognitive performance in the heavy metal-induced Alzheimer’s disease mice and the Vascular Dementia resembled rats when treated with BAC.
BAC has been studied and speculated that the possible mechanism is to improve the symptoms of dementia by reducing the expression of inflammatory factors and reducing inflammation in the brain, which is different from the traditional hypothesis of accumulation of amyloid and tau protein.
Further detail in the report…
|
Drug |
MoA |
RoA |
Company |
Logo |
Phase |
|
BAC |
pro-cytokines inhibitor |
Topical |
Charsire Biotechnology Corp. |
II | |
|
XXX |
XXX |
XXX |
XXX |
II |
Note: Detailed emerging therapies assessment will be provided in the final report of Vascular Dementia.
Vascular Dementia Market Outlook
Vascular dementia is a common condition with no effective approved pharmacological treatments available. Vascular dementia arises from ischemic insults such as hemorrhage and hypoperfusion that trigger neurodegeneration by depriving nerve cells of oxygen and glucose.
Vascular Dementia has multifactorial etiopathology, diverse clinical manifestations, and multiple clinical subtypes. Moreover, the diagnostic criteria are not consistent worldwide.
Although patients with Vascular Dementia constitute the second-largest dementia population, treatment data are lacking. There are no effective pharmacological compounds approved for the treatment of vascular dementia in any jurisdiction worldwide. Controlling medical conditions that impact cardiovascular health is recommended to prevent further decline. For example, medications can be prescribed to control blood pressure, cholesterol, heart disease, and diabetes. Aspirin or other drugs may be prescribed to prevent clots from forming in blood vessels.
The absence of treatment creates a problematic situation for those suffering from the disease, their caregivers, and healthcare providers. The aim of prevention treatment is to reduce the symptoms of the disease by eliminating its cause or main risk factors. The treatment goal includes symptomatic improvement of the core symptoms such as cognition, function, and behavior; slowing of progression of disease; and treatment of neuropsychiatric symptoms such as depression, anxiety, and others.
Cholinesterase inhibitors, and the NMDA (the N-methyl-D-aspartate receptor) antagonist, are the only medications currently licensed for AD treatment, have been found to show some cognitive improvements in mild to moderately advanced Vascular Dementia. Cholinesterase inhibitors work by inhibiting acetylcholinesterase, which is accountable for clearing acetylcholine, a neurotransmitter responsible for muscle contractions, blood vessel dilation, and regulating heart rate. With the cholinesterase inhibited, the acetylcholine concentrations rise and lead to better communication between the nerve cells in the brain. An artificial increase in acetylcholine levels by physostigmine, an acetylcholinesterase inhibitor that increases the extracellular acetylcholine levels, impairs memory consolidation and rescue in rodent and human subjects.
Vasodilation drugs mainly work to prevent constriction of the blood vessels anywhere in the body and allow greater blood flow. However, calcium channel blockers prevent calcium from entering the cells in the heart, vascular smooth muscle, and pancreas by lowering the blood pressure. Aspirin is a popular antiplatelet drug that works by blocking the cyclooxygenase chemical (COX) movement via the prostaglandin synthesis pathway (PGH2).
As per the study acetylcholinesterase inhibitors (AChEI) and memantine are often prescribed, sometimes inappropriately, in patients with vascular dementia.
- The therapeutic market size of Vascular Dementia in the 7MM was approximately USD 2,399 million in 2023.
- The market size in the 7MM will increase at a CAGR of 4% due to increasing awareness of the disease, better diagnosis, and the launch of the emerging therapy.
- The United States accounted for the highest market size of Vascular Dementia approximately 58% of the total market size in 7MM in 2023, in comparison to the other major markets i.e., EU4 countries (Germany, France, Italy, and Spain), and the United Kingdom, and Japan.
- Among the European countries, Germany had the highest market size with nearly USD 136 million in 2023, while Spain had the lowest market size of Vascular Dementia with ~USD 74 million in 2023.
- The market size for Vascular Dementia in Japan was estimated to be ~USD 480 million in 2023, which accounts for 20% of the total 7MM market.
- With the expected launch of upcoming therapies, such as BAC, the total market size of Vascular Dementia is expected to show change in the upcoming years.
Vascular Dementia Drugs Uptake
This section focuses on the uptake rate of potential drugs expected to launch in the market during 2020–2034. For example, BAC in the US is expected to be launched by 2026 with a peak share of 7%. BAC is anticipated to take 7 years to peak with a slow-medium uptake.
Further detailed analysis of emerging therapies drug uptake in the report…
Vascular Dementia Pipeline Development Activities
The report provides insights into different therapeutic candidates in Phase III, Phase II, and Phase I. It also analyzes key players involved in developing targeted therapeutics.
Pipeline Development Activities
The report covers information on collaborations, acquisitions and mergers, licensing, and patent details for Vascular Dementia emerging therapies.
KOL Views
To keep up with current market trends, we take KOLs and SMEs’ opinions working in the domain through primary research to fill the data gaps and validate the secondary research. Industry Experts were contacted for insights on Vascular Dementia evolving treatment landscape, patient reliance on conventional therapies, patient therapy switching acceptability, and drug uptake along with challenges related to accessibility, including KOL from Stony Brook University, Stony Brook, NY, United States; University of Southern California, Los Angeles, CA, United States; Johns Hopkins University, Baltimore, United States; Professional Association of German Neurologists, Germany; University of Catania, Italy; University of Manchester, United Kingdom; Keio University Global Research Institute (KGRI), Japan; Department of Neurology, University of Tokyo, Japan; and others.
Delveinsight’s analysts connected with 50+ KOLs to gather insights; however, interviews were conducted with 15+ KOLs in the 7MM. Their opinion helps understand and validate current and emerging therapies, treatment patterns, or Vascular Dementia market trends. This will support the clients in potential upcoming novel treatments by identifying the overall scenario of the market and the unmet needs.
Qualitative Analysis
We perform Qualitative and market Intelligence analysis using various approaches, such as SWOT and Conjoint Analysis. In the SWOT analysis, strengths, weaknesses, opportunities, and threats in terms of disease diagnosis, patient awareness, patient burden, competitive landscape, cost-effectiveness, and geographical accessibility of therapies are provided. These pointers are based on the Analyst’s discretion and assessment of the patient burden, cost analysis, and existing and evolving treatment landscape.
Conjoint Analysis analyzes multiple approved and emerging therapies based on relevant attributes such as safety, efficacy, frequency of administration, route of administration, and order of entry. Scoring is given based on these parameters to analyze the effectiveness of therapy.
Further, the therapies’ safety is evaluated wherein the acceptability, tolerability, and adverse events are majorly observed, and it sets a clear understanding of the side effects posed by the drug in the trials. In addition, the scoring is also based on the route of administration, order of entry and designation, probability of success, and the addressable patient pool for each therapy. According to these parameters, the final weightage score and the ranking of the emerging therapies are decided.
Market Access and Reimbursement
The high cost of therapies for the treatment is a major factor restraining the growth of the global drug market. Because of the high cost, the economic burden is increasing, leading the patient to escape from proper treatment.
The report further provides detailed insights on the country-wise accessibility and reimbursement scenarios, cost-effectiveness scenario of approved therapies, programs making accessibility easier and out-of-pocket costs more affordable, insights on patients insured under federal or state government prescription drug programs, etc.
Scope of the Report
- The report covers a segment of key events, an executive summary, and a descriptive overview of Vascular Dementia, explaining its causes, signs and symptoms, and currently available therapies.
- Comprehensive insight has been provided into the epidemiology segments and forecasts, the future growth potential of diagnosis rate, disease progression, and treatment guidelines.
- Additionally, an all-inclusive account of the current and emerging therapies and the elaborative profiles of late-stage and prominent therapies will impact the current treatment landscape.
- A detailed review of the Vascular Dementia market, historical and forecasted market size, market share by therapies, detailed assumptions, and rationale behind the approach is included in the report covering the 7MM drug outreach.
- The report provides an edge while developing business strategies, by understanding trends, through SWOT analysis and expert insights/KOL views, patient journey, and treatment preferences that help shape and drive the 7MM Vascular Dementia market.
Vascular Dementia Report Insights
- Patient Population
- Therapeutic Approaches
- Vascular Dementia Pipeline Analysis
- Vascular Dementia Market Size and Trends
- Existing and Future Market Opportunities
Vascular Dementia Report Key Strengths
- 11 years Forecast
- The 7MM Coverage
- Vascular Dementia Epidemiology Segmentation
- Key Cross Competition
- Conjoint Analysis
- Drugs Uptake and Key Market Forecast Assumptions
Vascular Dementia Report Assessment
- Current Treatment Practices
- Unmet Needs
- Pipeline Product Profiles
- Market Attractiveness
- Qualitative Analysis (SWOT and Conjoint Analysis)
Key Questions
Market Insights
- What was the Vascular Dementia market share (%) distribution in 2020 and what it would look like in 2034?
- What would be the Vascular Dementia total market size as well as market size by therapies across the 7MM during the forecast period (2024–2034)?
- What are the key findings pertaining to the market across the 7MM and which country will have the largest Vascular Dementia market size during the forecast period (2024–2034)?
- At what CAGR, the Vascular Dementia market is expected to grow at the 7MM level during the forecast period (2024–2034)?
- What would be the Vascular Dementia market outlook across the 7MM during the forecast period (2024–2034)?
- What would be the Vascular Dementia market growth till 2034 and what will be the resultant market size in the year 2034?
- How would the market drivers, barriers, and future opportunities affect the market dynamics and subsequent analysis of the associated trends?
Epidemiology Insights
- What are the disease risk, burden, and unmet needs of Vascular Dementia?
- What is the historical Vascular Dementia patient population in the United States, EU4 (Germany, France, Italy, Spain) and the UK, and Japan?
- What would be the forecasted patient population of Vascular Dementia at the 7MM level?
- What will be the growth opportunities across the 7MM with respect to the patient population pertaining to Vascular Dementia?
- Out of the above-mentioned countries, which country would have the highest prevalent population of Vascular Dementia during the forecast period (2024–2034)?
- At what CAGR the population is expected to grow across the 7MM during the forecast period (2024–2034)?
Current Treatment Scenario, Marketed Drugs, and Emerging Therapies
- What are the current options for the treatment of Vascular Dementia along with the approved therapy?
- What are the current treatment guidelines for the treatment of Vascular Dementia in the US, Europe, And Japan?
- What are the Vascular Dementia-marketed drugs and their MOA, regulatory milestones, product development activities, advantages, disadvantages, safety, efficacy, etc.?
- How many companies are developing therapies for the treatment of Vascular Dementia?
- How many emerging therapies are in the mid-stage and late stages of development for the treatment of Vascular Dementia?
- What are the key collaborations (Industry–Industry, Industry-Academia), Mergers and acquisitions, and licensing activities related to the Vascular Dementia therapies?
- What are the recent therapies, targets, mechanisms of action, and technologies developed to overcome the limitations of existing therapies?
- What are the clinical studies going on for Vascular Dementia and their status?
- What are the key designations that have been granted for the emerging therapies for Vascular Dementia?
- What are the 7MM historical and forecasted market of Vascular Dementia?
Reasons to Buy
- The report will help in developing business strategies by understanding the latest trends and changing treatment dynamics driving the Vascular Dementia Market.
- Insights on patient burden/disease incidence, evolution in diagnosis, and factors contributing to the change in the epidemiology of the disease during the forecast years.
- To understand the existing market opportunity in varying geographies and the growth potential over the coming years.
- Distribution of historical and current patient share based on real-world prescription data along with reported sales of approved products in the US, EU4 (Germany, France, Italy, and Spain), the United Kingdom, and Japan.
- Identification of strong upcoming players in the market will help in devising strategies that will help in getting ahead of competitors.
- Detailed analysis and potential of current and emerging therapies under the conjoint analysis section to provide visibility around leading emerging drugs.
- Highlights of Access and Reimbursement policies of approved therapies, barriers to accessibility of off-label expensive therapies, and patient assistance programs.
- To understand the perspective of Key Opinion Leaders around the accessibility, acceptability, and compliance-related challenges of existing treatment to overcome barriers in the future.
- Detailed insights on the unmet needs of the existing market so that the upcoming players can strengthen their development and launch strategy.