Non-Cystic Fibrosis Bronchiectasis Market Size was USD 1,455.1 million in 2023 and is projected to grow rapidly by 2034
Get a Sneak Peek at the Latest non cystic fibrosis bronchiectasis ncfb market size and forecast Report
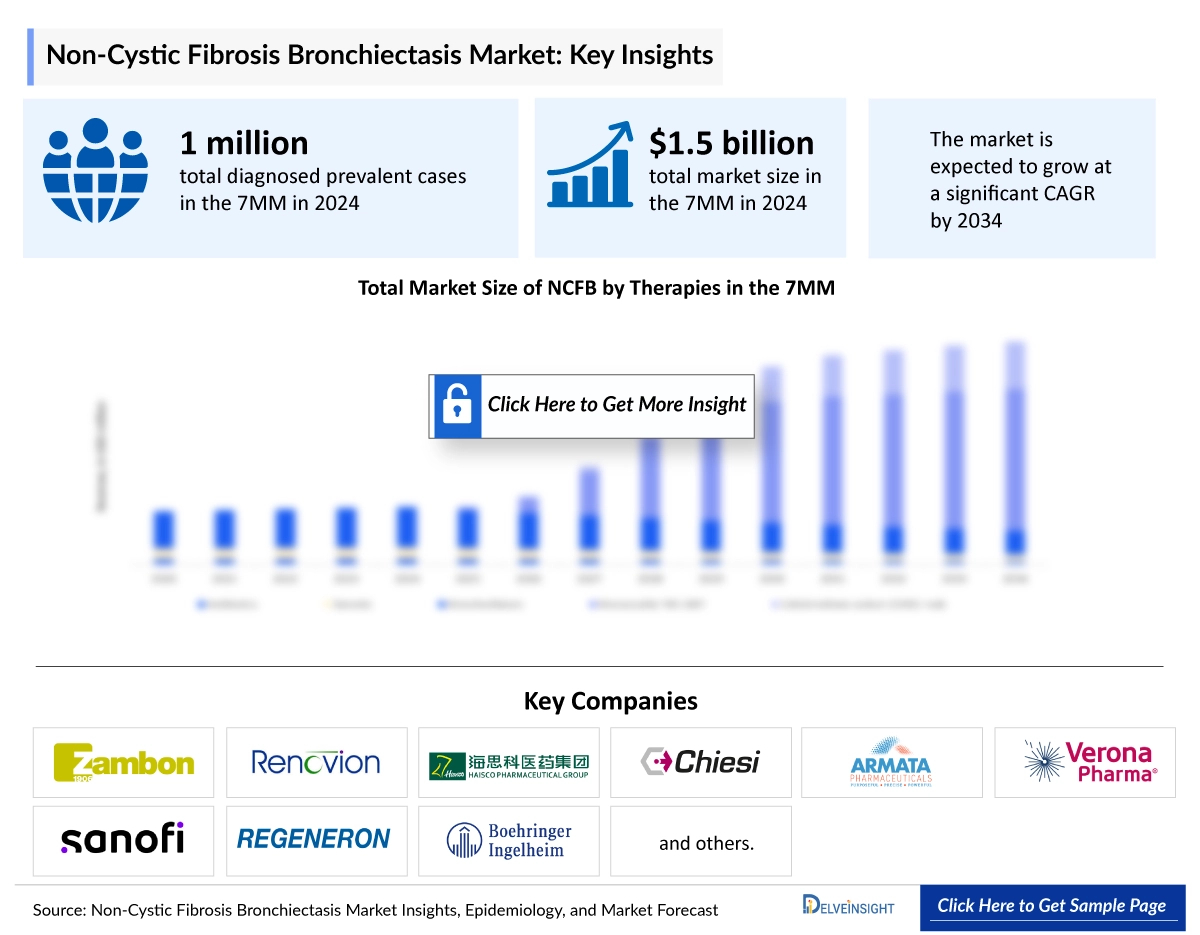
The Non-Cystic Fibrosis Bronchiectasis market size was valued approximately USD 1,455.1 million in 2023 and is anticipated to grow with a significant CAGR during the study period (2020-2034). DelveInsight’s comprehensive market research provides critical insights into such market trends, enabling stakeholders to understand growth drivers, emerging opportunities, and potential challenges within the Non-Cystic Fibrosis Bronchiectasis landscape.
In 2023, the US market for NCFB was valued at around USD 664.3 million and is projected to grow by 2034. Within the EU4 and UK, the UK held the largest market share at approximately USD 295 million in 2023, followed by Spain with about USD 195.7 million. In Japan, the NCFB market was valued at roughly USD 67.8 million in 2023 and is expected to expand by 2034. Overall, Japan’s NCFB market accounted for nearly USD 439 million, representing about 25% of the total revenue across the 7 major markets (7MM).
According to DelveInsight's epidemiology model, there were approximately 1,028,651 diagnosed prevalent cases of NCFB across the 7MM in 2023, with numbers expected to rise over the forecast period (2024–2034). The US had the highest diagnosed prevalence, with around 380,711 cases in 2023, which are projected to increase in the coming years. Within the EU4 and UK, the UK reported the most cases at approximately 224,976, followed by Spain with about 149,236 cases, both of which are expected to change by 2034. In Japan, there were roughly 96,580 diagnosed prevalent cases in 2023, with anticipated growth over the forecast period.
In 2023, the US had an estimated 88,706 mild, 156,853 moderate, and 135,152 severe cases of NCFB, with these figures expected to rise by 2034. In the EU4 and UK, diagnosed cases of NCFB were around 247,274 in males and 304,086 in females in 2023, with projections indicating potential changes throughout the 2024–2034 period. The epidemiological model also reported approximately 125,871 postinfective NCFB cases in the EU4 and UK, the highest among etiology-specific cases. In Japan, about 148,867 NCFB patients had Pseudomonas aeruginosa involvement in 2023.
DelveInsight’s report, “Non-Cystic Fibrosis Bronchiectasis Market Insights, Epidemiology, and Market Forecast-2034,” provides a comprehensive analysis of the Non-Cystic Fibrosis Bronchiectasis landscape. The report delivers detailed insights into the disease, including historical and projected epidemiology, helping stakeholders understand the prevalence, incidence, and patient demographics across key regions.
Additionally, it examines Non-Cystic Fibrosis Bronchiectasis market dynamics, offering a thorough assessment of current and emerging market trends, treatment patterns, and therapeutic developments. The analysis spans major markets, including the United States, EU4 (Germany, Spain, Italy, France, and the United Kingdom), and Japan, equipping decision-makers with actionable intelligence for strategic planning, investment, and research initiatives.
To Know in detail about the Non-Cystic Fibrosis Bronchiectasis market outlook, drug uptake, treatment scenario and epidemiology trends, Click here; Non-Cystic Fibrosis Bronchiectasis Market Forecast
Some of the key facts of the Non-Cystic Fibrosis Bronchiectasis Market Report:
- Key Non-Cystic Fibrosis Bronchiectasis Companies: Zambon, AstraZeneca, Boehringer Ingelheim, Zambon, Insmed, AstraZeneca, Zambion, The Marcus Foundation, Inc., Chiesi Farmaceutici S.p.A., SolAeroMed Inc., Zambon SpA, Boehringer Ingelheim, Renovion, Inc., Novartis, Alaxia SAS, and others
- Key Non-Cystic Fibrosis Bronchiectasis Therapies: Inhaled Colistimethate Sodium (CMS I-neb), FASENRA (benralizumab), CMS I-neb, BI 1291583, Brensocatib, Colistimethatesodium, Trikafta, CHF 6333, S-1226, CMS, BI 1323495, ARINA-1, TIP, ALX-009, and others
- The Non-Cystic Fibrosis Bronchiectasis epidemiology based on gender analyzed that in the US, females diagnosed with NCFB are higher in number than males.
- The Non-Cystic Fibrosis Bronchiectasis market is expected to surge due to the disease's increasing prevalence and awareness during the forecast period. Furthermore, launching various multiple-stage Non-Cystic Fibrosis Bronchiectasis pipeline products will significantly revolutionize the Non-Cystic Fibrosis Bronchiectasis market dynamics.
Non-Cystic Fibrosis Bronchiectasis Overview
Non-Cystic Fibrosis Bronchiectasis (NCFB) is a chronic lung disease characterized by irreversible widening and damage of the bronchial airways, not caused by cystic fibrosis. This structural damage prevents proper clearing of mucus from the lungs, leading to persistent inflammation, recurrent infections, and progressive lung function decline.
Common symptoms include chronic cough, excessive sputum production, shortness of breath, chest discomfort, and frequent respiratory infections. NCFB can develop due to infections, immune system disorders, lung injury, or may occur without a known cause. Early diagnosis and ongoing management are important to reduce flare-ups and prevent disease progression.
Get a Free sample for the Non-Cystic Fibrosis Bronchiectasis Market Forecast, Size & Share Analysis Report:
https://www.delveinsight.com/report-store/non-cystic-fibrosis-bronchiectasis-ncfb-market
Key Trends in Non-Cystic Fibrosis Bronchiectasis Therapeutics Market:
- Growing Focus on Targeted and Personalized Treatment Approaches: Increased understanding of disease heterogeneity and underlying causes such as infection type, inflammation patterns, and comorbid respiratory conditions is driving tailored therapy development.
- Pipeline Expansion with Novel Mechanisms of Action: Emerging therapies including anti-inflammatory agents, neutrophil elastase inhibitors, inhaled antibiotics, and mucolytics are under investigation to address unmet needs where current treatment options offer limited efficacy.
- Increasing Adoption of Inhaled and Advanced Delivery Systems: Controlled-release inhaled formulations and device-based drug delivery solutions are improving medication deposition in the lungs, enhancing patient adherence and therapeutic outcomes.
- Integration of Digital Health and Remote Monitoring: Use of telemedicine platforms, home-based lung function monitoring, and AI-driven exacerbation prediction tools is improving disease management and reducing hospitalizations.
- Rising Clinical Trial Activity and Industry Investment: Growing recognition of NCFBE as a distinct and under-treated condition has led to increased clinical research funding and partnerships between biopharma companies and academic institutions.
Non-Cystic Fibrosis Bronchiectasis Epidemiology
The Non-Cystic Fibrosis Bronchiectasis epidemiology section provides insights into the historical, current, and forecasted epidemiology trends in the seven major countries (7MM) from 2020 to 2034. It helps to recognize the causes of current and forecasted trends by exploring numerous studies and views of key opinion leaders. The epidemiology section also provides a detailed analysis of the diagnosed patient pool and future trends.
Non-Cystic Fibrosis Bronchiectasis Epidemiology Segmentation:
The Non-Cystic Fibrosis Bronchiectasis market report proffers epidemiological analysis for the study period 2020–2034 in the 7MM segmented into:
- Total Diagnosed Prevalent Cases of NCFB in the 7MM
- Gender-specific Diagnosed Prevalent Cases of NCFB in the 7MM
- Severity-specific Diagnosed Prevalent Cases of NCFB in the 7MM
- Etiology-specific Diagnosed Prevalent Cases of NCFB in the 7MM
- Microbiology of NCFB patients in the 7MM
Download the report to understand which factors are driving Non-Cystic Fibrosis Bronchiectasis epidemiology trends @ Non-Cystic Fibrosis Bronchiectasis Epidemiology Forecast
Recent Development In The Non-Cystic Fibrosis Bronchiectasis Treatment Landscape:
- In August 2025, The FDA has approved brensocatib (Brinsupri; Insmed) for treating non–cystic fibrosis bronchiectasis, making it the first therapy approved for this patient population and the first dipeptidyl peptidase 1 (DPP1) inhibitor authorized for a neutrophil-mediated disease. Brensocatib is an oral, small-molecule, reversible DPP1 inhibitor that works by blocking DPP1, thereby reducing the activation of neutrophil serine proteases key contributors to inflammation and tissue damage in chronic lung conditions like bronchiectasis.
- In February 2025, The FDA granted priority review to brensocatib, an investigational treatment for patients with non-cystic fibrosis bronchiectasis. Following acceptance of the new drug application, the target action date has been set for August 12, 2025. If approved, brensocatib would become the first and only authorized therapy for bronchiectasis and the first drug in the new dipeptidyl peptidase 1 class for treating neutrophil-mediated diseases.
- In February 2025, Verona Pharma is a biopharmaceutical company dedicated to developing and bringing to market innovative treatments for chronic respiratory diseases with considerable unmet medical needs. Ohtuvayre™ (ensifentrine) is the company’s inaugural commercial product and represents the first inhaled therapy for COPD maintenance that uniquely combines bronchodilator and non-steroidal anti-inflammatory effects in a single molecule. Ensifentrine also shows potential for treating non-cystic fibrosis bronchiectasis, cystic fibrosis, asthma, and other respiratory conditions.
- In December 2024, Armata Pharmaceuticals, Inc. (NYSE American: ARMP), a biotech firm focused on developing high-purity, pathogen-specific bacteriophage therapies for chronic pulmonary diseases and antibiotic-resistant infections, has reported encouraging topline results from its Phase 2 ("Tailwind") trial. The study assesses AP-PA02, a novel inhaled multi-phage therapy aimed at treating chronic Pseudomonas aeruginosa infections in patients with non-cystic fibrosis bronchiectasis (NCFB).
- In July 2024, Armata Pharmaceuticals has announced the completion of patient enrollment for its Phase II Tailwind clinical trial, which is assessing the efficacy of inhaled AP-PA02 in individuals with non-cystic fibrosis bronchiectasis (NCFB) and chronic Pseudomonas aeruginosa infection.
- In May 2024, Insmed Incorporated announced encouraging topline results from the ASPEN study, a global Phase III randomized, double-blind, placebo-controlled trial assessing the efficacy, safety, and tolerability of brensocatib in patients with non-cystic fibrosis bronchiectasis.
- In April 2024, 30 Technology announced that the European Medicines Agency (EMA) has authorized the Phase I/IIa NOPA study to evaluate the safety, tolerability, and effectiveness of nebulized Nitric Oxide Formulations in patients with Non-Cystic Fibrosis Bronchiectasis (NCFB) who have Pseudomonas aeruginosa or other potentially pathogenic microorganisms (PPMs).
Non-Cystic Fibrosis Bronchiectasis Drugs Uptake and Pipeline Development Activities
The drugs uptake section focuses on the rate of uptake of the potential drugs recently launched in the Non-Cystic Fibrosis Bronchiectasis market or expected to get launched during the study period. The analysis covers Non-Cystic Fibrosis Bronchiectasis market uptake by drugs, patient uptake by therapies, and sales of each drug.
Moreover, the therapeutics assessment section helps understand the drugs with the most rapid uptake and the reasons behind the maximal use of the drugs. Additionally, it compares the drugs based on market share.
The report also covers the Non-Cystic Fibrosis Bronchiectasis Pipeline Development Activities. It provides valuable insights about different therapeutic candidates in various stages and the key companies involved in developing targeted therapeutics. It also analyzes recent developments such as collaborations, acquisitions, mergers, licensing patent details, and other information for emerging therapies.
Non-Cystic Fibrosis Bronchiectasis Therapies and Key Companies
- Inhaled Colistimethate Sodium (CMS I-neb): Zambon
- FASENRA (benralizumab): AstraZeneca
- CMS I-neb: Zambon
- BI 1291583: Boehringer Ingelheim
- Brensocatib: Insmed/ AstraZeneca
- Colistimethatesodium: Zambion
- Trikafta: The Marcus Foundation, Inc.
- CHF 6333: Chiesi Farmaceutici S.p.A.
- S-1226: SolAeroMed Inc.
- CMS: Zambon SpA
- BI 1323495: Boehringer Ingelheim
- ARINA-1: Renovion, Inc.
- TIP: Novartis
- ALX-009: Alaxia SAS
Discover more about therapies set to grab major Non-Cystic Fibrosis Bronchiectasis market share @ Non-Cystic Fibrosis Bronchiectasis Treatment Landscape
Non-Cystic Fibrosis Bronchiectasis Market Drivers
- Rising prevalence of bronchiectasis globally, especially among older adults, increasing demand for effective therapies.
- Advancements in diagnostic imaging technologies like high-resolution CT scans enabling early and accurate diagnosis.
- Development of novel therapies and inhalation treatments to manage symptoms and reduce exacerbations.
- Growing awareness among healthcare professionals and patients about disease management.
- Supportive healthcare policies and reimbursement initiatives promoting treatment adoption and R&D investment.
Non-Cystic Fibrosis Bronchiectasis Market Barriers
- High cost of treatment and long-term management, limiting patient access in certain regions.
- Variability in disease severity and underlying causes, complicating standardized treatment approaches.
- Limited patient awareness leading to underdiagnosis or delayed diagnosis.
- Challenges in managing recurrent infections and comorbidities, affecting treatment outcomes.
- Scarcity of large-scale clinical trials due to the rare and heterogeneous nature of the disease.
Scope of the Non-Cystic Fibrosis Bronchiectasis Market Report
- Study Period: 2020–2034
- Coverage: 7MM [The United States, EU5 (Germany, France, Italy, Spain, and the United Kingdom), and Japan]
- Key Non-Cystic Fibrosis Bronchiectasis Companies: Zambon, AstraZeneca, Boehringer Ingelheim, Zambon, Insmed, AstraZeneca, Zambion, The Marcus Foundation, Inc., Chiesi Farmaceutici S.p.A., SolAeroMed Inc., Zambon SpA, Boehringer Ingelheim, Renovion, Inc., Novartis, Alaxia SAS, and others
- Key Non-Cystic Fibrosis Bronchiectasis Therapies: Inhaled Colistimethate Sodium (CMS I-neb), FASENRA (benralizumab), CMS I-neb, BI 1291583, Brensocatib, Colistimethatesodium, Trikafta, CHF 6333, S-1226, CMS, BI 1323495, ARINA-1, TIP, ALX-009, and others
- Non-Cystic Fibrosis Bronchiectasis Therapeutic Assessment: Non-Cystic Fibrosis Bronchiectasis current marketed and Non-Cystic Fibrosis Bronchiectasis emerging therapies
- Non-Cystic Fibrosis Bronchiectasis Market Dynamics: Non-Cystic Fibrosis Bronchiectasis market drivers and Non-Cystic Fibrosis Bronchiectasis market barriers
- Competitive Intelligence Analysis: SWOT analysis, PESTLE analysis, Porter’s five forces, BCG Matrix, Market entry strategies
- Non-Cystic Fibrosis Bronchiectasis Unmet Needs, KOL’s views, Analyst’s views, Non-Cystic Fibrosis Bronchiectasis Market Access and Reimbursement
To know more about Non-Cystic Fibrosis Bronchiectasis companies working in the treatment market, visit @ Non-Cystic Fibrosis Bronchiectasis Clinical Trials and Therapeutic Assessment
About DelveInsight
DelveInsight is a leading Healthcare Business Consultant, and Market Research firm focused exclusively on life sciences. It supports Pharma companies by providing comprehensive end-to-end solutions to improve their performance.
It also offers Healthcare Consulting Services, which benefits in market analysis to accelerate the business growth and overcome challenges with a practical approach.
Need more?
- ✅ Connect with our analyst to learn how this research was developed
- ✅ Expand the scope with additional segments or countries through free customization
- ✅ Discover how this report can directly influence your business growth
Related Reports
Non-Cystic Fibrosis Bronchiectasis (NCFB) - Epidemiology Forecast - 2034
DelveInsight's Non-Cystic Fibrosis Bronchiectasis - Epidemiology Forecast 2034 report delivers an in-depth understanding of the disease, historical, and forecasted epidemiology..
Non-Cystic Fibrosis Bronchiectasis (NCFB) - Pipeline Insight, 2025
Non-Cystic Fibrosis Bronchiectasis (NCFB) Pipeline Insights, 2025 report by DelveInsight outlays comprehensive insights of present clinical development scenario and growth prospects across..
-epidemiology.png&w=256&q=75)

