Spinal Cord Injury Market Size in the 7MM is expected to Reach USD 1456 million by 2034.
Get a Sneak Peek at the Latest spinal cord injury market size Report
Spinal Cord Injury Drugs Market 2034: Clinical Trials, Medication, EMA, PDMA, FDA Approval, Epidemiology, Therapies and Companies by DelveInsight
The Spinal Cord Injury Market across the 7 major markets (7MM) — the US, EU4, UK, and Japan — is projected to grow significantly, rising from USD 354 million in 2025 to USD 1,456 million by 2034, at a CAGR of 15.4%.
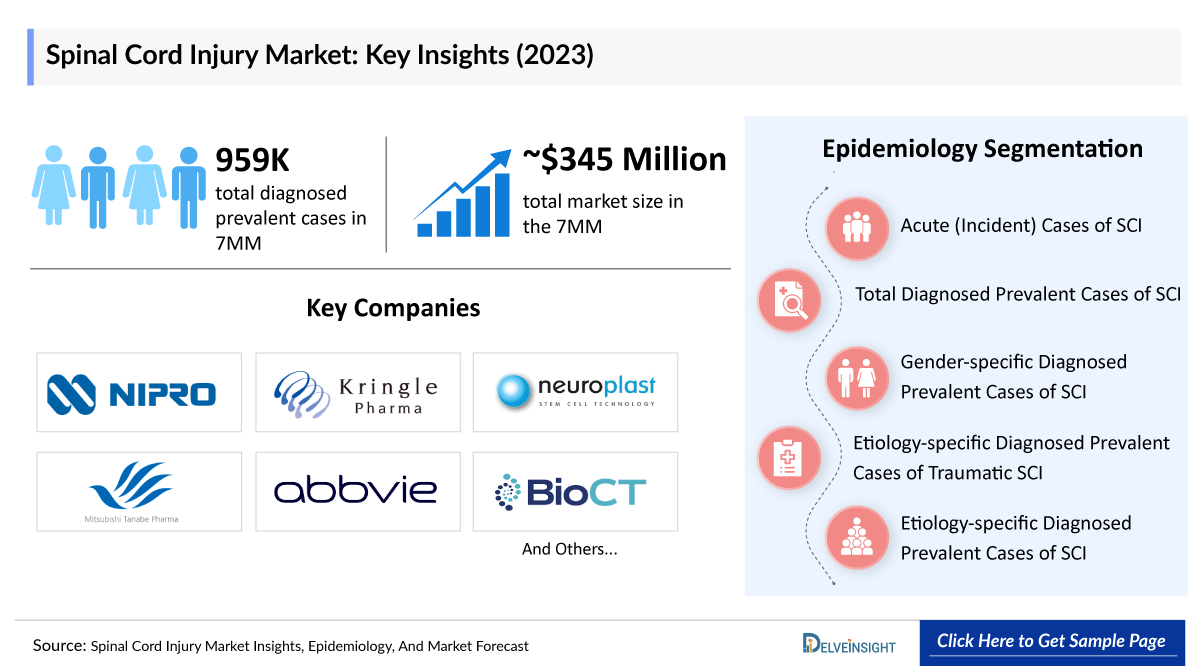
In 2023, there were about 959,000 diagnosed prevalent cases of SCI in the 7MM, with the US accounting for 32%, EU4 and the UK for 56%, and Japan for 12%.
Market growth will be fueled by:
- The launch of novel therapies such as KP-100IT, Neuro-Cells, Elezanumab (ABT-555), and MT-3921.
- Increasing awareness, aging populations, and improved diagnostics leading to a rising prevalence.
- Expanding rehabilitation technologies and a strong R&D pipeline.
Currently, treatment options remain limited, with STEMIRAC being the only approved therapy (in Japan). The market presents a significant unmet need for effective and regenerative treatments.
Key players — Kringle Pharma, Neuroplast, AbbVie, and Mitsubishi Tanabe Pharma America — are actively advancing their pipelines. Notably, Kringle Pharma’s KP-100IT is in Phase III trials and expected to launch in Japan by 2025, marking a major milestone for the SCI treatment landscape.
DelveInsight’s Spinal Cord Injury Market Insights Report provides an in-depth analysis of current treatment practices, emerging therapies, market dynamics, and forecasted growth across the 7 major markets (US, EU4, UK, and Japan) from 2020 to 2034.
The SCI market is primarily driven by rising incidence rates, innovations in regenerative medicine, and the increasing use of neurostimulation technologies. Factors such as accidents, falls, and sports injuries are escalating demand for advanced treatment options. Emerging modalities like stem cell therapy, gene therapy, and exoskeleton-based rehabilitation are transforming the therapeutic landscape.
Despite challenges such as high treatment costs, limited curative options, and regulatory complexities, the market is poised for steady growth as ongoing R&D efforts and technological advancements continue to improve patient outcomes and expand therapeutic possibilities.
Request for sample report @ Spinal Cord Injury Market Insights
Key Takeaways from the Spinal Cord Injury Market Report
· As per DelveInsight analysis, the spinal cord injury market is expected to grow positively at a significant CAGR during the study period (2020–2034).
· As per the DelveInsight assessment, the total diagnosed cases of SCI in the 7MM was estimated to be ~725K cases in 2022, which is expected to increase in the forecast period (2022-2034).
· Leading spinal cord injury companies such as NervGen Pharma, VA Office of Research and Development, Kessler Institute for Rehabilitation, StemCyte, Inc., Mitsubishi Tanabe Pharma America Inc., AbbVie, and others are developing novel spinal cord injury drugs that can be available in the spinal cord injury market in the coming years.
· Some key therapies for spinal cord injury treatment include NVG-291, Romosozumab, Umbilical Cord Blood Mononuclear Cell, MT-3921, Elezanumab, and others.
Recent Development In The Spinal Cord Injury Treatment Landscape:
- In June 2025, Lineage Cell Therapeutics, Inc. (NYSE American and TASE: LCTX), a clinical-stage biotech company focused on developing innovative allogeneic (“off-the-shelf”) cell therapies for severe neurological and ophthalmic disorders, announced the addition of new presenters for the 3rd Annual Spinal Cord Injury Investor Symposium (3rd SCIIS). This year’s event will be entirely virtual, featuring interactive and on-demand sessions beginning on June 27, 2025.
- In March 2025, Cellino and Matricelf (TASE: MTLF) announced a partnership aimed at advancing the global biomanufacturing of personalized spinal cord injury therapies. This collaboration integrates Cellino’s Nebula™ technology with Matricelf’s innovative regenerative medicine approach. By combining Cellino’s automated production of induced pluripotent stem cells (iPSCs) with Matricelf’s double autologous 3D differentiation process, the partnership seeks to enable scalable and patient-specific regenerative treatments. Cellino’s Nebula™, a proprietary closed-cassette biomanufacturing platform, is specifically designed to generate high-quality iPSCs with exceptional consistency and sterility at a large scale.
- In March 2025, NurExone Biologic Inc. (TSXV: NRX) (OTCQB: NRXBF) (FSE: J90) announced the successful completion of a key preclinical study supporting its upcoming Investigational New Drug (IND) application. This study marks a significant step toward the company’s first-in-human trials. The results demonstrated that ExoPTEN treatment, across various dosing regimens, promoted motor function recovery and notably enhanced blood flow at the spinal cord injury site—both critical factors for tissue repair and functional improvement.
- In March 2025, Phantom Neuro, a neurotech company advancing human-machine interfaces, announced that it has received two prestigious designations from the U.S. Food and Drug Administration (FDA): the Breakthrough Device Designation and the highly selective TAP (Targeted Acceleration Pathway) Designation for its minimally invasive neural interface, the Phantom X platform.
- In February 2025, Neuvotion Receives FDA Clearance for NeuStim™ Providing Non-Invasive, High-Resolution Stimulation for the Hand After Stroke or Spinal Cord Injury
- March 2024: Mitsubishi Tanabe Pharma America Inc. announced a study of Phase 2 clinical trials for MT-3921. The purpose of this study is to compare the efficacy and safety of intravenous (IV) infusions of MT-3921 to placebo in subjects with acute traumatic cervical spinal cord injury. Subjects meeting eligibility criteria will enter the 6-month double-blind period. Subjects will be randomized in a 2:1 ratio to receive MT-3921 or placebo in a double blind manner.
- March 2024: Moleac Pte Ltd announced a study of Phase 4 clinical trials for NeuroAiD. SATURN investigates the promising role of NeuroAiD in patients with spinal cord injury and will provide important information on the feasibility of conducting larger controlled trials.
- March 2024: NervGen Pharma announced a study of Phase 1 & 2 clinical trials for NVG-291. To evaluate the effect of NVG-291 on descending connectivity in subjects with subacute and chronic SCI (20 subjects per Cohort and results will be analyzed separately) using objective electrophysiological measures, in addition to clinical assessments. To evaluate safety and tolerability of NVG-291 in a SCI population, as measured by clinical assessments (Physical Examination, Vital Signs, ECG, etc.) as well as clinical laboratory measures.
Discover which therapies are expected to grab the major spinal cord injury market share @ Spinal Cord Injury Market Report
Spinal Cord Injury Overview
Spinal cord injury is a debilitating condition resulting from trauma or disease that damages the spinal cord, impairing motor, sensory, and autonomic functions below the level of injury. Common causes include motor vehicle accidents, falls, sports injuries, and medical conditions like tumors or infections. Spinal cord injury can be classified as complete, with total loss of function, or incomplete, where some sensation or movement remains.
The severity of Spinal cord injury depends on the location and extent of damage. Injuries to the cervical spine can result in quadriplegia, affecting all four limbs, whereas injuries to the thoracic, lumbar, or sacral regions typically cause paraplegia, involving the lower body.
Spinal cord injury Symptoms vary widely and may include paralysis, loss of sensation, pain, spasticity, and autonomic dysfunction affecting bladder, bowel, and cardiovascular regulation. Acute Spinal cord injury is a medical emergency requiring immediate stabilization to prevent further damage.
Spinal cord injury Treatment focuses on minimizing damage and promoting recovery. Initial care includes immobilization, corticosteroids to reduce inflammation, and surgical intervention when needed. Long-term management involves physical rehabilitation, occupational therapy, and assistive technologies to improve independence. Advances in regenerative medicine, including stem cell therapy, neuroprosthetics, and electrical stimulation, offer hope for improving outcomes and quality of life for individuals with Spinal cord injury.
Spinal Cord Injury Epidemiology
Spinal Cord Injury Epidemiology Segmentation
The spinal cord injury epidemiology section provides insights into the historical and current spinal cord injury patient pool and forecasted trends for individual seven major countries. It helps to recognize the causes of current and forecasted trends by exploring numerous studies and views of key opinion leaders.
The spinal cord injury market report proffers epidemiological analysis for the study period 2020–2034 in the 7MM segmented into:
- Total Spinal Cord Injury Cases
- Spinal Cord Injury Diagnosed Cases
- Spinal Cord Injury Gender-specific Cases
- Spinal Cord Injury Cause-specific Cases
- Total cases of SCI at Neurological Level
To know more about spinal cord injury treatment, visit @ Spinal Cord Injury Treatment Drugs
Spinal Cord Injury Treatment Market
A Spinal Cord Injury (SCI) occurs when damage to the spinal cord disrupts communication between the brain and body, often caused by accidents, falls, or violence. Individuals aged 16–30, particularly males and those engaged in high-risk activities, are most vulnerable. Symptoms depend on injury severity and site, typically resulting in loss of movement, sensation, and autonomic function, and may lead to paralysis.
Current treatment primarily focuses on acute medical intervention, surgery, and rehabilitation. Surgical decompression relieves spinal pressure, while pharmacological treatments such as methylprednisolone and other anti-inflammatory and neuroprotective agents aim to minimize damage. Physical therapy and assistive devices aid recovery, with tramadol, gabapentin, and cannabinoids used for pain management. However, there is still no definitive cure for SCI.
LYRICA (pregabalin) remains the only FDA-approved drug for neuropathic pain in SCI, while STEMIRAC, conditionally approved by Japan’s PMDA in 2018, is undergoing post-marketing evaluation, with full approval expected by 2025.
The pipeline for SCI treatment remains limited, with key candidates including KP–100IT, Neuro–Cells, MT-3921, and Elezanumab (ABT-555). These emerging therapies aim to address unmet needs beyond symptomatic relief.
In 2023, the SCI market size across the 7MM was approximately USD 345 million, projected to grow significantly through 2034:
- US: USD 146 million (2023) — expected to rise with new therapy launches.
- EU4 and UK: USD 152 million (44% of total market), led by Germany (USD 51M) and France (USD 40M).
- Japan: USD 47 million, also projected to increase.
By 2034, KP–100IT alone is expected to generate around USD 28 million in the 7MM, highlighting the market’s strong growth potential driven by innovative therapeutics and ongoing clinical advancements.
Key Spinal Cord Injury Therapies and Companies
· NVG-291: NervGen Pharma
· Romosozumab: VA Office of Research and Development/Kessler Institute for Rehabilitation
· Umbilical Cord Blood Mononuclear Cell: StemCyte, Inc.
· MT-3921: Mitsubishi Tanabe Pharma America Inc.
· Elezanumab: AbbVie
Learn more about the FDA-approved drugs for spinal cord injury @ Drugs for Spinal Cord Injury Treatment
Spinal Cord Injury Market Dynamics and Trends
Spinal Cord Injury Market Drivers
· Rising Incidence of Spinal Cord Injuries - Increasing cases due to road accidents, falls, and sports injuries are expanding the patient pool and driving demand for effective therapies.
· Advancements in Regenerative Medicine - Progress in stem cell therapy, gene therapy, and neuroregeneration research offers new possibilities for repairing nerve damage and restoring function.
· Growing Adoption of Neurostimulation and Exoskeleton Devices - Innovative rehabilitation technologies such as robotic exoskeletons, functional electrical stimulation (FES), and brain-computer interfaces are improving patient outcomes and quality of life.
· Rising Awareness and Early Diagnosis - Increased public awareness, better trauma care, and improved diagnostic imaging are leading to timely interventions and more accurate disease management.
· Strong R&D Pipeline and Emerging Therapies - Promising candidates like KP-100IT, Neuro-Cells, MT-3921, and Elezanumab (ABT-555) are expected to transform the treatment landscape.
· Supportive Government and Healthcare Initiatives - Funding for rehabilitation programs, clinical research, and assistive technology development is helping accelerate innovation in SCI care.
Spinal Cord Injury Market Barriers
· Limited Approved Therapies - Few effective or curative options exist; STEMIRAC (Japan) and LYRICA (for neuropathic pain) remain the only approved treatments, highlighting a major unmet need.
· High Treatment and Rehabilitation Costs - Long-term care, surgeries, and assistive technologies are financially burdensome, limiting access for many patients.
· Regulatory and Clinical Trial Challenges - Stringent approval requirements and complex clinical endpoints slow down drug development and commercialization timelines.
· Complex Pathophysiology of SCI - The irreversible nature of spinal cord damage and the lack of effective nerve regeneration mechanisms hinder therapeutic progress.
· Limited Awareness in Low- and Middle-Income Regions - Inadequate trauma management infrastructure and lack of access to specialized rehabilitation centers restrict market expansion.
· Uncertain Long-term Efficacy of Emerging Therapies - Many investigational treatments require long-term validation to confirm safety, efficacy, and sustainability of clinical benefits.
Scope of the Spinal Cord Injury Market Report
· Study Period: 2020–2034
· Coverage: 7MM [The United States, the EU-4 (Germany, France, Italy, and Spain), the United Kingdom, and Japan]
· Key Spinal Cord Injury Companies: NervGen Pharma, VA Office of Research and Development, Kessler Institute for Rehabilitation, StemCyte, Inc., Mitsubishi Tanabe Pharma America Inc., AbbVie, and others
· Key Spinal Cord Injury Therapies: NVG-291, Romosozumab, Umbilical Cord Blood Mononuclear Cell, MT-3921, Elezanumab, and others
· Therapeutic Assessment: Spinal Cord Injury current marketed and emerging therapies
· Spinal Cord Injury Market Dynamics: Conjoint Analysis of Emerging Spinal Cord Injury Drugs
· Competitive Intelligence Analysis: SWOT analysis and Market entry strategies
· Unmet Needs, KOL's views, Analyst's views, Spinal Cord Injury Market Access and Reimbursement
Discover more about spinal cord injury drugs in development @ Spinal Cord Injury Clinical Trials
Table of Contents
1. Spinal Cord Injury Market Key Insights
2. Spinal Cord Injury Market Report Introduction
3. Spinal Cord Injury Market Overview at a Glance
4. Spinal Cord Injury Market Executive Summary
5. Disease Background and Overview
6. Spinal Cord Injury Treatment and Management
7. Spinal Cord Injury Epidemiology and Patient Population
8. Patient Journey
9. Spinal Cord Injury Marketed Drugs
10. Spinal Cord Injury Emerging Drugs
11. Seven Major Spinal Cord Injury Market Analysis
12. Spinal Cord Injury Market Outlook
13. Potential of Current and Emerging Therapies
14. KOL Views
15. Unmet Needs
16. SWOT Analysis
17. Appendix
18. DelveInsight Capabilities
19. Disclaimer
20. About DelveInsight
About DelveInsight
DelveInsight is a leading Business Consultant and Market Research firm focused exclusively on life sciences. It supports pharma companies by providing comprehensive end-to-end solutions to improve their performance.
Need more?
- ✅ Connect with our analyst to learn how this research was developed
- ✅ Expand the scope with additional segments or countries through free customization
- ✅ Discover how this report can directly influence your business growth
Related Reports
Spinal Cord Injury Market Insight, Epidemiology And Market Forecast - 2034
Spinal Cord Injury Market Insights, Epidemiology, and Market Forecast-2032" report delivers an in-depth understanding of the Spinal Cord Injury epidemiology segmentation
Spinal Cord Injury (SCI) - Epidemiology forecast- 2034
DelveInsight's Spinal Cord Injury (SCI) - Epidemiology Forecast 2034 report delivers an in-depth understanding of the disease, historical, and forecasted epidemiology of the disease...
Spinal Cord Injury - Pipeline Insight, 2025
Spinal Cord Injury Pipeline Insights, 2025 report by DelveInsight outlays comprehensive insights of present clinical development scenario and growth prospects across..
Chronic Spinal Cord Injury - Epidemiology Forecast - 2034
DelveInsight's Chronic Spinal Cord Injury - Epidemiology Forecast 2034 report delivers an in-depth understanding of the disease, historical, and forecasted epidemiology of Chronic Spinal Cord Injury in the United States, EU5 (Germany, France, Italy, Spain, and the United Kingdom), and Japan.




