Allergy Diagnostics Market Summary
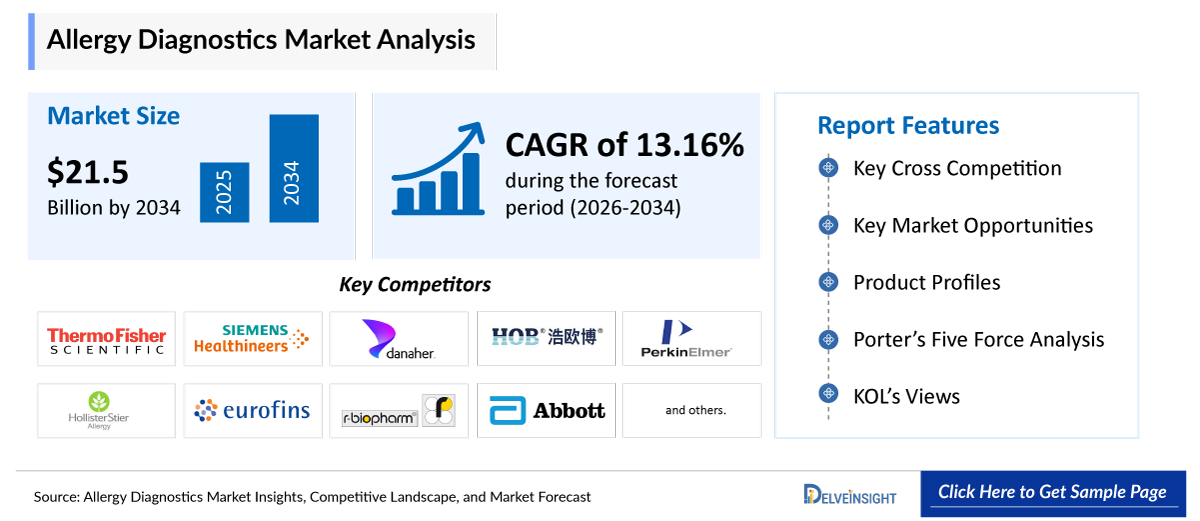
- The global allergy diagnostics market is expected to increase from USD 7,143.89 million in 2025 to USD 21,561.93 million by 2034, reflecting strong and sustained growth.
- The global allergy diagnostics market is growing at a CAGR of 13.16% during the forecast period from 2026 to 2034.
- The allergy diagnostics market is driven by the rising prevalence of allergic diseases such as allergic rhinitis and allergic asthma. Additionally, increasing pollution levels worldwide are worsening respiratory allergies, boosting the demand for screening and monitoring. Moreover, technological advancements like molecular diagnostics, component-resolved testing, and automated immunoassays have improved the accuracy and speed of tests, enabling more personalized treatment and further propelling market growth.
- The leading companies operating in the allergy diagnostics market include Thermo Fisher Scientific Inc., Siemens Healthineers, Danaher Corporation, HOB Biotech Group Corp Ltd., PerkinElmer, Inc., HollisterStier Allergy, Eurofins Scientific, R-Biopharm AG, AESKU Group GmbH, Abbott, Lincoln Diagnostics, Inc., Astra Biotech GmbH, Erba Group, Canone Medical Diagnostics, BioMerieux, Omega Diagnostics Group PLC, Stallergenes Greer, Neogen Corporation, Reacta Healthcare, Allergy Therapeutics and others.
- North America is poised to retain its leadership in the global allergy diagnostics market, largely due to the growing prevalence of allergic conditions such as allergic rhinitis and allergic asthma. The rising number of individuals affected by these disorders has heightened the demand for precise and timely diagnostic solutions. Moreover, increasing pollution in urban and industrial regions has amplified exposure to environmental allergens, further driving the need for allergy testing. The region’s emphasis on innovation and advancements in diagnostic technologies, including enhanced immunoassays and automated testing systems, has improved both the accuracy and efficiency of allergy detection, contributing to steady market growth.
- In the type segment of the allergy diagnostics market, the consumables category is estimated to account for the largest market share in 2025.
Request for unlocking the report of the @Allergy Diagnostics Market
Allergy Diagnostics Market Size and Forecasts
|
Report Metrics |
Details |
|
2025 Market Size |
USD 7,143.89 million |
|
2034 Projected Market Size |
USD 21,561.93 million |
|
Growth Rate (2026-2034) |
13.16% CAGR |
|
Largest Market |
North America |
|
Fastest Growing Market |
Asia-Pacific |
|
Market Structure |
Moderately Concentrated |
Factors Contributing to the Growth of the Allergy Diagnostics Market
- Growing incidence of allergic diseases such as allergic asthma and allergic rhinitis: The allergy diagnostics market is significantly driven by the growing incidence of allergic diseases, particularly allergic asthma and allergic rhinitis. An increasing number of individuals worldwide are affected by these conditions due to factors such as changing lifestyles, environmental exposures, and genetic predisposition. This rising prevalence has created a strong demand for accurate and timely diagnostic solutions, enabling healthcare providers to identify allergens, manage symptoms effectively, and personalize treatment plans. As a result, the need for advanced allergy testing methods, including specific IgE tests, skin prick tests, and molecular diagnostics, continues to expand, making this driver a key contributor to market growth.
- Increasing levels of pollution across the world: Rising global pollution levels are a significant driver of the allergy diagnostics market. Exposure to air pollutants, including particulate matter, industrial emissions, and vehicle exhaust, has been strongly associated with the growing prevalence of allergic conditions. These pollutants can trigger immune responses, intensify symptoms, and lead to more frequent and severe allergic episodes. As a result, there is an increasing demand for accurate and timely allergy testing, encouraging healthcare providers to adopt advanced diagnostic solutions that can identify specific allergens and guide effective treatment. Consequently, escalating environmental pollution continues to drive the need for comprehensive allergy diagnostics worldwide.
- Technological advancements in diagnostic methods: Technological advancements in diagnostic methods are a key driver of the allergy diagnostics market. Innovations such as component-resolved diagnostics, multiplex immunoassays, and automated testing platforms have significantly improved the accuracy, sensitivity, and efficiency of allergy detection. These advancements enable healthcare providers to precisely identify specific allergens, differentiate between cross-reactive substances, and tailor personalized treatment plans for patients. Moreover, the integration of digital tools and laboratory automation has streamlined testing workflows, reduced turnaround times, and enhanced patient outcomes. As a result, ongoing technological progress continues to expand the adoption of advanced allergy diagnostic solutions worldwide.
Allergy Diagnostics Market Report Segmentation
This allergy diagnostics market report offers a comprehensive overview of the global allergy diagnostics market, highlighting key trends, growth drivers, challenges, and opportunities. It covers detailed market segmentation by Type (Products {Instruments and Consumables} and Services), Allergy Type (Food Allergy, Drug Allergy, Inhalant Allergy, and Others), Diagnostic Test Type (In-Vivo, In-Vitro, and Others), End-User (Hospitals and Diagnostic Centers), and Geography. The report provides valuable insights into the competitive landscape, regulatory environment, and market dynamics across major markets, including North America, Europe, and Asia-Pacific. Featuring in-depth profiles of leading industry players and recent product innovations, this report equips businesses with essential data to identify market potential, develop strategic plans, and capitalize on emerging opportunities in the rapidly growing allergy diagnostics market.
Allergy diagnostics refers to the medical procedures and tests used to identify an individual’s allergic sensitivities and reactions to specific substances, known as allergens. These diagnostics help detect immune system responses to environmental factors, foods, medications, or other triggers. Common methods include skin prick tests, specific IgE blood tests, patch tests, and molecular or component-resolved diagnostics, which enable healthcare providers to accurately determine the cause of allergic symptoms and guide effective management and treatment strategies.
The allergy diagnostics market is witnessing substantial growth, driven by the rising prevalence of allergic conditions. Additionally, the growing number of patients affected by these disorders has increased the demand for accurate and timely diagnostic solutions, as early detection is crucial for effective management. Moreover, escalating pollution levels in urban and industrial areas have led to higher exposure to environmental allergens, further fueling the need for comprehensive allergy testing. Consequently, the region’s strong emphasis on innovation and technological advancements, including sophisticated immunoassays and automated testing platforms, has improved the precision and efficiency of allergy detection, supporting sustained market expansion.
Get More Insights into the Report @Allergy Diagnostics Market
What are the latest allergy diagnostics market dynamics and trends?
The allergy diagnostics market is largely driven by the growing prevalence of allergic conditions, including allergic rhinitis, asthma, and food allergies, across the globe. Factors such as rising environmental pollution, urbanization, and changing lifestyles have intensified respiratory and food-related allergies, increasing the demand for precise and timely diagnostic solutions, which in turn has fueled the expansion of the allergy diagnostics market.
According to the Global Initiative for Asthma (2024), asthma was one of the most common chronic non-communicable diseases, affecting more than 260 million people worldwide. Patients with asthma frequently undergo allergy diagnostic tests such as skin prick tests, specific IgE blood tests, and component-resolved diagnostics to identify allergens responsible for airway inflammation and exacerbations. Identifying these triggers supports personalized treatment and immunotherapy decisions, thereby increasing the demand for allergy testing and driving the growth of the allergy diagnostics market.
Moreover, as per the stats provided by the Food and Agriculture Organization of the United States (2024), over 220 million people globally lived with food allergies. The increasing prevalence of Food allergy, particularly among infants and children, is significantly driving growth in the allergy diagnostics market. Rising awareness of severe reactions, including Anaphylaxis, has led to greater demand for early and accurate diagnosis through specific IgE blood tests, skin prick tests, and component-resolved diagnostics, thereby escalating the overall market of allergic diagnostic market.
Furthermore, as per the data provided by the Asthma and Allergy Foundation of America (2024), about 5 to 10% of reactions to drugs are true allergic reactions. Suspected allergies to antibiotics (especially penicillins), NSAIDs, anesthetics, and biologic drugs are increasingly leading physicians to recommend confirmatory testing to avoid unnecessary drug avoidance and ensure safe treatment options. Diagnostic approaches such as skin testing, specific IgE assays, and controlled drug challenge tests are being used to differentiate true immune-mediated allergies from non-allergic adverse reactions.
Additionally, according to the Australian Institute of Health and Welfare (2026), in 2022, around 6.1 million people (24%) in Australia were estimated to have allergic rhinitis. Since allergic rhinitis is often chronic and recurrent, physicians frequently recommend skin prick tests and specific IgE blood tests to identify sensitization patterns and guide targeted treatment, including allergen avoidance and immunotherapy.
However, the increase in product development activities is further boosting the overall market of allergy diagnostics. For instance, in April 2025, Beckman Coulter Life Sciences, a global leader in laboratory automation and innovation, introduced its next-generation Basophil Activation Test (BAT) for research use only, designed to more effectively characterize food allergies without exposing patients to potentially harmful allergens.
Thus, the factors mentioned above are expected to boost the overall market of allergy diagnostic during the forecast period from 2026 to 2034.
The allergy diagnostics market faces several restraints and challenges that can limit its growth. Technical and operational issues, such as the presence of skin conditions like eczema or dermatographism, can interfere with the accuracy of tests like skin prick assays, making reliable diagnosis more difficult. Additionally, the premium costs associated with advanced allergy diagnostic products can restrict their accessibility, particularly in low- and middle-income regions, further limiting widespread adoption. These factors collectively pose significant challenges for both healthcare providers and patients in the effective utilization of allergy diagnostics.
Allergy Diagnostics Market Segment Analysis
Allergy Diagnostics Market by Type (Products {Instruments and Consumables} and Services), Allergy Type (Food Allergy, Drug Allergy, Inhalant Allergy, and Others), Diagnostic Test Type (In-Vivo, In-Vitro, and Others), End-User (Hospitals and Diagnostic Centers), and Geography (North America, Europe, Asia-Pacific, and Rest of the World).
By Allergy Type: Inhalant Allergy is projected to account for the highest revenue share in the global market.
The inhalant allergy segment accounts for the largest share of the allergy diagnostics market, contributing approximately 53% of the total market revenue in 2025. This dominance is primarily driven by the high global prevalence of respiratory allergies, particularly allergic rhinitis, allergic asthma, and conjunctivitis, which are commonly triggered by airborne allergens such as pollen, dust mites, mold spores, and animal dander.
One of the key factors supporting the segment’s leadership is the increasing level of air pollution and urbanization, which has significantly increased exposure to airborne allergens, especially in densely populated regions. Additionally, climate change and longer pollen seasons have intensified allergic reactions, leading to a higher number of patients seeking diagnostic testing.
Another important driver is that inhalant allergies affect both children and adults and often require repeated diagnostic testing for effective disease management. Healthcare providers frequently use skin prick tests and specific IgE blood tests to identify inhalant allergens, which further increases testing volumes.
Overall, the high prevalence of respiratory allergies, environmental factors, and the need for continuous diagnosis and monitoring are key factors driving the dominance of the inhalant allergy segment in the allergy diagnostics market.
By Type: Consumables Category Dominates the Market
The consumables segment dominates the allergy diagnostics market with a market share of 60% in 2025, because these items are essential for both routine and specialized tests. Consumables represent one of the most significant revenue-generating segments in the allergy diagnostics market and are a major factor driving overall market expansion due to their recurring, high-volume usage across both in vivo and in vitro testing platforms.
Unlike capital instruments, which are purchased as one-time investments, consumables such as allergen extracts, specific IgE reagents, calibrators, controls, assay kits, microplates, test cassettes, lancets, and sample collection materials are required for every individual test performed, creating a continuous and predictable revenue stream for manufacturers.
With the rising prevalence of allergic conditions such as Allergic rhinitis, Asthma, and Food allergy, the overall testing volume has increased substantially, directly amplifying demand for consumable products.
The growing adoption of component-resolved diagnostics and multiplex immunoassays further accelerates consumable usage, as these advanced tests require specialized reagents and allergen components that command higher per-test pricing.
For instance, in September 2025, Siemens Healthineers expanded its 3gAllergy assay menu with nine new component allergens, including six targeting peanuts, the leading cause of fatal food-related anaphylaxis.
Additionally, expansion of automated immunoassay analyzers in hospitals and reference laboratories has improved throughput capacity, enabling laboratories to process larger test volumes and thereby consume more reagents on a daily basis. Pediatric allergy screening programs, repeat monitoring for immunotherapy, and confirmatory testing prior to biologic therapy initiation also contribute to repeated testing cycles, strengthening recurring consumable demand.
From a market perspective, consumables often account for the largest share of total allergy diagnostic revenues because they combine high frequency of use with premium pricing for proprietary reagents and allergen panels.
As allergy awareness grows globally and healthcare systems emphasize early and accurate diagnosis, the steady rise in test volumes ensures sustained and scalable growth of the consumables segment, making it a primary engine of expansion in the overall allergy diagnostics market.
By Diagnostic Test Type: In-Vivo Category Dominates the Market
The in-vivo allergy testing segment holds the leading share in the allergy diagnostics market, accounting for approximately 57% of the total market in 2025. This dominance is primarily driven by the widespread clinical adoption of skin-based tests, particularly skin prick tests (SPT), which are considered the first-line diagnostic approach for identifying allergic sensitizations.
One of the key advantages of in-vivo tests is their rapid turnaround time, as results are typically available within 15–20 minutes, allowing physicians to diagnose and plan treatment during the same patient visit. Additionally, these tests are cost-effective and easy to perform, requiring minimal laboratory infrastructure compared to blood-based in-vitro tests. Because of these benefits, in-vivo testing is widely used in allergy clinics, hospitals, and dermatology practices.
Another factor supporting the strong adoption of in-vivo tests is their high sensitivity for detecting immediate allergic reactions, particularly for food allergies, pollen allergies, dust mites, and animal dander. Clinical guidelines from allergy and immunology associations often recommend skin prick testing as the primary screening method, further reinforcing its market dominance.
However, the segment faces certain limitations, including risk of allergic reactions during testing, contraindications in patients with severe skin conditions, and the need to discontinue antihistamines before testing. Due to these factors, some patients may require in-vitro blood tests as an alternative.
Overall, the in-vivo segment continues to dominate the allergy diagnostics market due to its affordability, quick results, and strong clinical acceptance, although the increasing adoption of advanced in-vitro diagnostic technologies may gradually narrow the market share gap in the coming years.
By End-Users: Hospitals Category Dominates the Market
The hospitals segment has consistently emerged as the dominant end-user category in the global allergy diagnostics market due to its major role in boosting the allergy diagnostics market due to their large patient base and access to advanced diagnostic technologies. They offer a wide range of tests, including skin prick tests, serum IgE assays, and component-resolved diagnostics, which are often integrated into patient care for diagnosis, treatment planning, and monitoring therapy outcomes. Hospitals also participate in clinical research and trials for new allergy therapeutics and diagnostic tools, creating additional demand for specialized tests. Moreover, insurance coverage for hospital-based testing and initiatives to educate patients about allergy risks and management further encourage testing. The combination of high test volume, technological capabilities, and comprehensive care services in hospitals significantly contributes to the overall growth of the allergy diagnostics market.
Allergy Diagnostics Market Regional Analysis
North America Allergy Diagnostics Market Trends
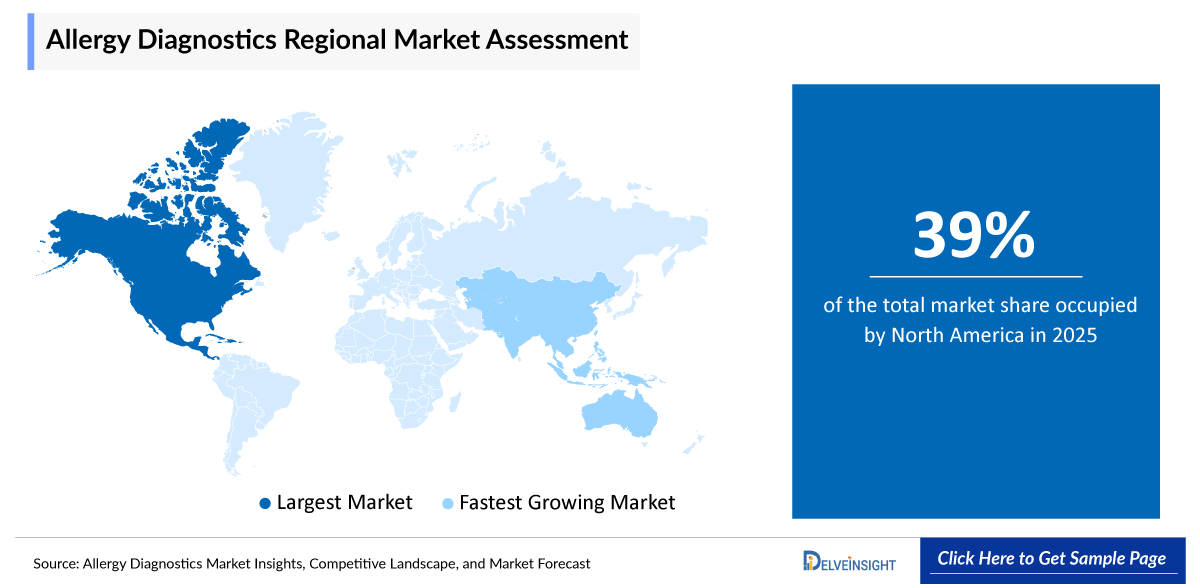
North America is projected to hold a largest share of 39% in 2025 of the allergy diagnostics market, driven by the rising incidence of allergies and strong awareness of various allergic conditions. The presence of leading diagnostic companies and well-established allergy and immunology centers further supports the adoption of allergy diagnostic products in the region.
According to the American College of Allergy, Asthma & Immunology (2023), 7.7% of Americans had asthma. Of the estimated 24.9 million affected individuals, 20.2 million were adults and 4.6 million were children. Asthma, a chronic respiratory condition commonly triggered by allergens such as pollen, dust mites, pet dander, and mold, demands accurate diagnosis to enable targeted treatment and prevent severe flare-ups. Healthcare providers increasingly use diagnostic tools like skin prick tests, specific IgE blood tests, and component-resolved diagnostics to identify the precise allergens driving asthma symptoms, thereby driving growth in the allergy diagnostics market.
Moreover, according to the Centers for Disease Control and Prevention, in 2024, 31.7% of adults in the United States had been diagnosed with a seasonal allergy, eczema, or a food allergy. Specifically, 25.2% had a seasonal allergy, 7.7% had eczema, and 6.7% had a food allergy. Seasonal allergies, caused by triggers like pollen, mold, and dust, affect millions each year, driving the use of diagnostic tools such as skin prick tests, specific IgE blood tests, and component-resolved diagnostics to accurately identify the allergens. Likewise, the growing prevalence of food allergies to common triggers like peanuts, milk, eggs, shellfish, and wheat has heightened the demand for precise testing to prevent severe reactions like anaphylaxis, thereby fueling the growth of the allergy diagnostics market.
Furthermore, the increase in product development activities are further boosting the overall market. For instance, in June 2024, the FDA cleared and granted a CLIA waiver for the Kenota 1 Total IgE system, a semi-quantitative in vitro diagnostic test for measuring total IgE levels from fingerstick blood samples. This allows rapid point-of-care IgE testing in allergist and immunologist offices, aiding clinical diagnosis of IgE-mediated allergic disorders.
However, rising awareness of allergies in the United States is significantly boosting the allergy diagnostics market by encouraging more people to seek early testing, accurate diagnosis, and proactive management of allergic conditions. Increased public education campaigns by health organizations and growing media coverage of allergy prevalence have made individuals more aware of symptoms and available diagnostic options, leading to higher test volumes for specific IgE assays, skin prick tests, and advanced component-resolved diagnostics.
Thus, all the above-mentioned factors are anticipated to propel the market for allergy diagnostics in North America during the forecast period.
Europe Allergy Diagnostics Market Trends
Europe is driving the allergy diagnostics market growth due to the high prevalence of allergic conditions, increasing awareness of allergy management, and widespread adoption of advanced diagnostic technologies.
According to Asthma + Lung UK (2024), asthma was a common condition, with 7.2 million people in the UK affected. Asthma is strongly linked to allergic reactions caused by environmental triggers such as pollen, dust mites, mold, and pet dander. With the global prevalence of asthma on the rise, accurately identifying the allergens responsible for airway inflammation and asthma exacerbations has become increasingly important. This has driven greater demand for allergy diagnostic tests, including skin prick tests, specific IgE blood tests, and component‑resolved diagnostics, to precisely determine allergen sensitivities in asthma patients thereby fueling growth in the allergy diagnostics market.
Additionally, according to the European Food Safety Authority (2025), 1% of the EU population approximately 4.5 million people were affected by food allergens. As the prevalence of food allergies rises globally, impacting both children and adults, the need to accurately identify the specific allergens responsible for adverse reactions has grown. This has driven increased demand for diagnostic tools such as skin prick tests, specific IgE blood tests, oral food challenges, and component-resolved diagnostics, which enable clinicians to precisely pinpoint allergenic triggers thereby fueling growth in the allergy diagnostics market.
However, the increase in product development activities is further boosting the overall market of allergy diagnostics. For instance, in July 2024, AliveDx proudly announced that it had received the IVDR CE mark for its groundbreaking microarray immunoassay, which was designed to detect specific IgE antibodies directed against a protein allergen in human serum. Hence, all the factors mentioned above are expected to drive the market for allergy diagnostics in Europe during the forecast period.
Asia-Pacific Allergy Diagnostics Market Trends
The Asia-Pacific region is driving growth in the allergy diagnostics market due to rising prevalence of allergic diseases, increasing awareness of allergy testing, rapid urbanization, and improving healthcare infrastructure, which together are boosting demand for accurate and early diagnosis of allergies.
According to the Global Asthma Report 2022, about 35 million people in India suffered from asthma. With the rising global prevalence of asthma, there has been a growing need to identify the specific allergens responsible for airway inflammation and asthma episodes. This has increased demand for diagnostic tools such as skin prick tests, specific IgE blood tests, and component-resolved diagnostics, enabling clinicians to accurately detect allergen sensitivities and, in turn, fueling growth in the allergy diagnostics market.
Moreover, according to a study conducted in 2023, allergic rhinitis (AR) was the most prevalent allergy, affecting approximately one in every six people in India. As one of the most prevalent allergic conditions, allergic rhinitis (AR) causes symptoms such as sneezing, nasal congestion, and itchy eyes, affecting millions worldwide especially in regions experiencing increasing pollution and urbanization. The growing need to accurately identify the specific allergens triggering these reactions has driven demand for diagnostic tests, including skin prick tests, specific IgE blood tests, and component-resolved diagnostics, thereby fueling growth in the allergy diagnostics market.
Thus, the factors mentioned above are expected to boost the market of allergy diagnostics across the Asia-Pacific region.
Who are the major players in the allergy diagnostics market?
The following are the leading companies in the allergy diagnostics market. These companies collectively hold the largest market share and dictate industry trends.
- Thermo Fisher Scientific Inc.
- Siemens Healthineers
- Danaher Corporation
- HOB Biotech Group Corp Ltd.
- PerkinElmer, Inc.
- HollisterStier Allergy
- Eurofins Scientific
- R-Biopharm AG
- AESKU Group GmbH
- Abbott
- Lincoln Diagnostics, Inc.
- Astra Biotech GmbH
- Erba Group
- Canone Medical Diagnostics
- BioMerieux
- Omega Diagnostics Group PLC
- Stallergenes Greer
- Neogen Coporation
- Reacta Healthcare
- Allergy Therapeutics
- Others
How is the competitive landscape shaping the allergy diagnostics market?
The competitive landscape of the allergy diagnostics market is highly dynamic and characterized by the presence of both established global players and emerging regional companies. Major companies such as Thermo Fisher Scientific, Siemens Healthineers, bioMérieux, and Stallergenes Greer are competing to expand their product portfolios, introduce innovative diagnostic technologies, and strengthen their geographical presence. Additionally, strategic collaborations, mergers, and acquisitions are common as companies aim to gain access to advanced testing platforms, novel reagents, and emerging markets. Innovation-driven competition is particularly strong, with players focusing on the development of high-throughput in-vitro tests, multiplex panels, and automated immunoassay systems to improve accuracy, speed, and patient safety. Moreover, smaller and niche companies are increasingly contributing to market growth by offering specialized allergy test kits, allergen extracts, and reagents targeting specific patient segments, thereby intensifying market rivalry. Companies are also leveraging digital solutions and laboratory automation to differentiate their offerings, reduce operational costs, and enhance diagnostic efficiency. As a result, the market is witnessing continuous technological advancements, aggressive product launches, and competitive pricing strategies, all of which are shaping the global allergy diagnostics market and driving its evolution toward more precise, convenient, and patient-friendly solutions.
Recent Developmental Activities in the Allergy Diagnostics Market
- In September 2025, Siemens expanded its 3gAllergy assay portfolio on the IMMULITE 2000 XPi system by introducing additional component allergen tests for peanut, wheat, and house dust mite, providing more detailed molecular-level insights.
- In May 2025, Revvity, Inc. announced the launch of the IDS i20™ analytical random access platform from EUROIMMUN, enabling fully automated chemiluminescence immunoassays (ChLIA). The versatile IDS i20 allowed simultaneous testing of 20 analytes across six diagnostic areas, including endocrinology, allergy, autoimmune and infectious diseases, Alzheimer’s disease, and therapeutic drug monitoring.
- In April 2025, Beckman Coulter Life Sciences, a global leader in laboratory automation and innovation, introduced its next-generation Basophil Activation Test (BAT) for research use only, designed to more effectively characterize food allergies without exposing patients to potentially harmful allergens.
- In July 2024, AliveDx proudly announced that it had received the IVDR CE mark for its groundbreaking microarray immunoassay, which was designed to detect specific IgE antibodies directed against a protein allergen in human serum.
|
Report Metrics |
Details |
|
Study Period |
2023 to 2034 |
|
Base Year |
2025 |
|
Forecast Period |
2026 to 2034 |
|
Allergy Diagnostics Market CAGR |
XX% |
|
Key Companies in the Allergy Diagnostics Market |
Thermo Fisher Scientific Inc., Siemens Healthineers, Danaher Corporation, HOB Biotech Group Corp Ltd., PerkinElmer, Inc., HollisterStier Allergy, Eurofins Scientific, R-Biopharm AG, AESKU Group GmbH, Abbott, Lincoln Diagnostics, Inc., Astra Biotech GmbH, Erba Group, Canone Medical Diagnostics, BioMerieux, Omega Diagnostics Group PLC, Stallergenes Greer, Neogen Corporation, Reacta Healthcare, Allergy Therapeutics and others. |
|
Allergy Diagnostics Market Segments |
by Type, by Allergy Type, by Diagnostic Test Type, by End-Users, and by Geography |
|
Allergy Diagnostics Regional Scope |
North America, Europe, Asia Pacific, Middle East, Africa, and South America |
|
Allergy Diagnostics Country Scope |
U.S., Canada, Mexico, Germany, United Kingdom, France, Italy, Spain, China, Japan, India, Australia, South Korea, and key Countries |
Allergy Diagnostics Market Segmentation
- Allergy Diagnostics by Type Exposure
- Products
- Instruments
- Consumables
- Services
- Products
- Allergy Diagnostics by Allergy Type Exposure
- Food Allergy
- Drug Allergy
- Inhalant Allergy
- Others
- Allergy Diagnostics by Diagnostic Test Type Exposure
- In-Vivo
- In-Vitro
- Others
- Allergy Diagnostics End-Users Exposure
- Hospitals
- Diagnostic Centers
- Allergy Diagnostics Geography Exposure
- North America Allergy Diagnostics Market
- United States Allergy Diagnostics Market
- Canada Allergy Diagnostics Market
- Mexico Allergy Diagnostics Market
- Europe Allergy Diagnostics Market
- United Kingdom Allergy Diagnostics Market
- Germany Allergy Diagnostics Market
- France Allergy Diagnostics Market
- Italy Allergy Diagnostics Market
- Spain Allergy Diagnostics Market
- Rest of Europe Allergy Diagnostics Market
- Asia-Pacific Allergy Diagnostics Market
- China Allergy Diagnostics Market
- Japan Allergy Diagnostics Market
- India Allergy Diagnostics Market
- Australia Allergy Diagnostics Market
- South Korea Allergy Diagnostics Market
- Rest of Asia-Pacific Allergy Diagnostics Market
- Rest of the World Allergy Diagnostics Market
- South America Allergy Diagnostics Market
- Middle East Allergy Diagnostics Market
- Africa Allergy Diagnostics Market
Allergy Diagnostics Market Recent Industry Trends and Milestones (2022-2026)
|
Category |
Key Developments |
|
Allergy Diagnostics Product Approvals |
AliveDx - Microarray Immunoassay on MosaiQ® platform (IVDR-CE) |
|
Allergy Diagnostics Product Launch |
Beckman Coulter Life Sciences launched Basophil Activation Test |
|
Acquisition in the Allergy Diagnostics Market |
Thermo Fisher Scientific acquired The Binding Site Group, Canon Medical Systems acquired Minaris Medical Co., Ltd. |
|
Company Strategy |
Thermo Fisher Scientific Inc.- focuses on innovation-driven growth, expanding its life sciences and diagnostics portfolio through acquisitions, R&D investment, and strategic partnerships. The company emphasizes advanced technologies, automation, and high-throughput solutions to serve hospitals, laboratories, and research institutions globally, while strengthening its presence in emerging markets and specialty diagnostics like allergy and molecular testing. Danaher Corporation- focuses on innovation-driven growth, expanding its portfolio through strategic acquisitions and continuous development of advanced diagnostic technologies. The company emphasizes high-quality, automated, and high-throughput solutions to improve lab efficiency and test accuracy. Additionally, Danaher invests in research partnerships and global expansion to strengthen market presence, particularly in in-vitro diagnostics, immunoassays, and laboratory automation |
|
Emerging Technology |
Component-Resolved Diagnostics (CRD), Multiplex Allergy Testing Panels, Automated Immunoassay Systems, Microfluidics-Based Allergy Testing, Artificial Intelligence & Machine Learning Integration, Basophil Activation Test (BAT), Nanotechnology & Biosensors and others. |
Impact Analysis
AI-Powered Innovations and Applications:
The integration of AI-powered innovations is significantly transforming the allergy diagnostics market by enhancing accuracy, efficiency, and predictive capabilities. Artificial intelligence and machine learning algorithms are being applied to analyze complex datasets from patient histories, in-vitro test results, and multiplex allergy panels, enabling clinicians to identify allergen sensitivities more precisely and rapidly. Additionally, AI-driven platforms facilitate personalized allergy profiling, helping healthcare providers predict potential allergic reactions and recommend targeted treatment or immunotherapy. Automation of data interpretation and diagnostic workflows through AI reduces human error, streamlines laboratory operations, and shortens turnaround times for test results. Moreover, AI applications in allergy diagnostics support the development of predictive models for emerging allergens and seasonal trends, aiding both clinicians and public health authorities in proactive disease management. Companies are increasingly investing in AI-enabled diagnostic devices, cloud-based analytics, and decision-support tools, which not only improve patient outcomes but also drive the adoption of technologically advanced solutions in hospitals, diagnostic laboratories, and specialty clinics. Consequently, AI-powered innovations are reshaping the competitive landscape, fostering market growth, and setting new standards for precision, efficiency, and personalized care in allergy diagnostics.
U.S. Tariff Impact Analysis on Allergy Diagnostics Market:
The imposition of tariffs in the United States has had a notable impact on the allergy diagnostics market, particularly affecting the import of diagnostic instruments, reagents, and consumables from international suppliers. Increased tariffs have raised the cost of imported testing kits, reagents, and specialized laboratory equipment, leading to higher operational expenses for hospitals, diagnostic laboratories, and clinics. Consequently, manufacturers and distributors are adjusting pricing strategies to offset these additional costs, which may influence purchasing decisions and slow down the adoption of certain high-value diagnostic technologies. Moreover, some companies are exploring local manufacturing or establishing regional supply chains to mitigate the impact of tariffs, maintain competitiveness, and ensure uninterrupted product availability. While domestic production helps reduce dependency on imports, it also requires significant investment in infrastructure and quality compliance. Overall, U.S. tariff policies have introduced cost considerations and strategic planning into the allergy diagnostics market, influencing supply chain dynamics, product pricing, and long-term market growth trajectories.
How This Analysis Helps Clients
- Cost Management: By understanding the tariff landscape, clients can anticipate cost increases and adjust pricing strategies accordingly, ensuring profitability.
- Supply Chain Optimization: Clients can identify alternative sourcing options and diversify their supply chains to reduce dependency on high-tariff regions, enhancing resilience.
- Regulatory Navigation: Expert guidance on navigating the evolving regulatory environment helps clients maintain compliance and avoid potential legal challenges.
- Strategic Planning: Insights into tariff impacts enable clients to make informed decisions about manufacturing locations, partnerships, and market entry strategies.
Startup Funding & Investment Trends
|
Company Name |
Total Funding |
Main Products |
Stage of Development |
Core Technology |
|
ATANIS Biotech |
$2million |
FAST-PASE |
- |
detects the release of mediators from patient-derived mast cells when exposed to specific allergens, allowing rapid, high-throughput, and safe identification of drug or food allergies without exposing the patient directly to allergens |
Key takeaways from the allergy diagnostics market report study
- Market size analysis for the current allergy diagnostics market size (2025), and market forecast for 8 years (2026 to 2034)
- Top key product/technology developments, mergers, acquisitions, partnerships, and joint ventures happened over the last 3 years.
- Key companies dominating the allergy diagnostics market.
- Various opportunities available for the other competitors in the allergy diagnostics market space.
- What are the top-performing segments in 2025? How these segments will perform in 2034?
- Which are the top-performing regions and countries in the current allergy diagnostics market scenario?
- Which are the regions and countries where companies should have concentrated on opportunities for the allergy diagnostics market growth in the future?