Bone Graft and Substitute Market
"Bone Graft and Substitutes Market by Source (Natural and Synthetic), Bone Graft and Substitutes Market by Product Type (Allograft {DBM, BMP, and Cell-Based Matrices}, Xenograft, and Others), Bone Graft and Substitutes Market by Application (Craniomaxillofacial, Dental, Joint Reconstruction, Spinal Fusion, and Others), Bone Graft and Substitutes Market by End-User (Hospitals & Clinics, Specialty Clinics, and Others), and Bone Graft and Substitutes Market by Geography (North America, Europe, Asia-Pacific, and Rest of the World) is expected to grow at a steady CAGR forecast till 2032 owing to the increasing prevalence of bone-related disorders and trauma cases, rising demand for bone grafts in dentistry, the growing geriatric population, and increase in product development activities across the globe
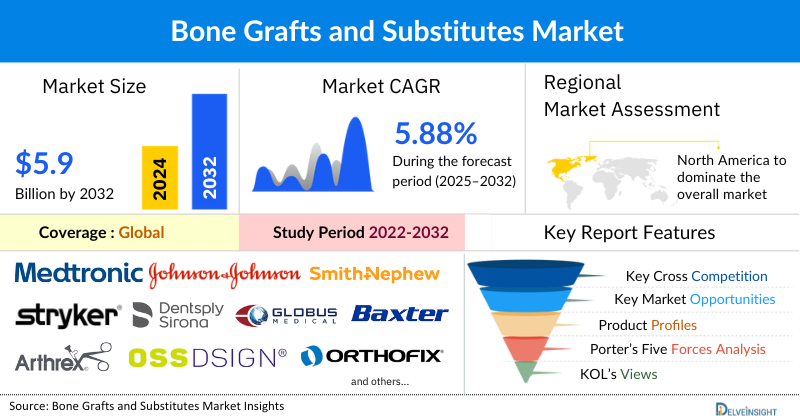
The bone graft and substitutes market was valued at USD 3,786.25 million in 2024, growing at a CAGR of 5.88% during the forecast period from 2025 to 2032 to reach USD 5,957.71 million by 2032. The increasing prevalence of bone-related disorders and trauma cases, coupled with a rising demand for bone grafts in dentistry, is significantly boosting the global market for bone grafts and substitutes. As bone diseases such as osteoporosis and bone fractures become more common, particularly among the aging population, the need for effective bone regeneration and repair solutions has surged. The growing geriatric population, which is more prone to bone degeneration, fractures, and joint issues, further drives demand for bone grafts, especially in orthopedic and dental procedures. Additionally, the increase in product development activities, with innovations in synthetic, allograft, and xenograft materials, is enhancing the effectiveness and availability of bone graft substitutes. These factors collectively contribute to the expansion of the bone graft and substitutes market, with more advanced, biocompatible, and cost-effective solutions being introduced to meet the rising demand during the forecast period from 2025 to 2032.
Bone Graft and Substitutes Market Dynamics:
As per the International Osteoporosis Foundation (2023), over 8.9 million fractures related to osteoporosis occur annually. Osteoporosis is one of the common degenerative bone diseases where the body starts to lose bone mass or makes too little bone. As individuals with osteoporosis are more prone to fractures, especially in areas like the hip, spine, and wrist, the demand for effective solutions to repair and regenerate damaged bone tissue rises. Bone grafts and substitutes, which are used to promote bone healing and provide structural support, are essential in treating fractures caused by osteoporosis, particularly when fractures are complex or difficult to heal thereby boosting the overall market of bone graft and substitute across the globe.
Moreover, the rising number of trauma cases has also contributed to the rising demand for bone grafts and substitutes. As per the World Health Organization factsheet (2023) on road traffic injuries, road injuries are the leading cause of fatality in children and young adults in the age group 5-29 years. The same factsheet further stated that near about 20-50 million people suffer from non-fatal injuries in road accidents resulting in a disability as a result of their injury. In road traffic accidents, the force of impact often leads to complex and severe fractures, including fractures of the limbs, pelvis, spine, and other critical areas, which require immediate and effective treatment. As these injuries often result in substantial bone loss or damage, bone grafts and substitutes become essential for repairing the damaged bones, promoting healing, and ensuring functional recovery.
Another factor contributing to the growth of the bone graft and substitutes market is the rising demand for bone grafts in the dental sector. According to the latest research study “Bone Graft and Substitutes in Dentistry: A Review of Current Trends and Developments” (2021), current estimates suggest that globally, roughly 2.2 million bone transplant surgeries are conducted each year, costing an approximate value of USD 664 million in 2021. The number of surgical procedures for treating bony abnormalities is estimated to grow by approximately 13% yearly.
Additionally, the increase in strategic activities among the key bone grafts and substitutes companies is further boosting the market of bone grafts and substitutes across the globe. For instance, in April 2023, ZimVie announced the launches of RegenerOss® CC allograft particulate and RegenerOss® bone graft plug for filling extraction sockets and periodontal defects.
Thus, the factors mentioned above are expected to boost the overall market of bone grafts and substitutes across the globe during the forecast period from 2025 to 2032.
However, limitations in the management of large bone defects and risks associated with autografts and allografts may hinder the future market of bone grafts and substitutes across the globe during the forecasted period.
Bone Graft and Substitutes Market Segment Analysis:
Bone Graft and Substitutes Market by Source (Natural and Synthetic), Bone Graft and Substitutes Market by Product Type (Allograft {DBM, BMP, and Cell-Based Matrices}, Xenograft, and Others), Bone Graft and Substitutes market by Application (Craniomaxillofacial, Dental, Joint Reconstruction, Spinal Fusion, and Others), Bone Graft and Substitutes market by End-User (Hospitals & Clinics, Specialty Clinics, and Others), and Bone Graft and Substitutes market by Geography (North America, Europe, Asia-Pacific, and Rest of the World)
The natural category in the bone graft and substitutes market is estimated to dominate the market with a market share of 66.85% in 2024. This can be attributed to the various advantages and applications provided by grafts taken from natural sources which will be responsible for driving the demand for natural bone grafts and substitutes market during the given forecast period. Natural bone grafts typically refer to biological tissues or bone structures transplanted from one organism to another within the same species or sometimes across species. These grafts include tissues such as bones and cartilage.
These bone grafts are derived from biological sources, making them compatible with the recipient's body tissues. This further reduces the risk of rejection and promotes integration with surrounding tissues. Additionally, natural bone grafts, such as autografts (tissues taken from the patient's own body) or allografts (tissues from a donor of the same species), have a lower risk of infection compared to synthetic materials because they do not introduce foreign substances into the body. Natural bone grafts, once integrated into the recipient's bone tissue, provide long-term strength and support, ensuring the health of the treated area.
Thus, the factors mentioned above are expected to boost the segment thereby boosting the overall Bone Graft and Substitutes market globally.
North America is expected to dominate the overall bone graft and substitutes market:
Among all regions, North America Bone Graft and Substitutes market is estimated to hold the largest share of 47.92% in the global bone graft and substitutes market in 2024, growing at a CAGR of 5.54% during the forecast period from 2025 to 2032. This can be attributed to the growing prevalence of spine-related disorders, and increasing cases of tooth loss, coupled with other supportive factors such as increasing orthopedic surgical procedures, new product launches, and increased focus on research and development among other factors that are driving the overall growth of the bone graft and substitutes market over the forecast period.
As per the data provided by the United Spinal Association (2022), every year approximately 17,700 Americans acquire a spinal cord injury. Roughly 78% are men, and their average age is 43. Most spinal cord injuries happened as a result of a vehicle accident (39.3%), followed closely by falls (31.8%), trailed by violence (13.2%), sports (8%), and medical/surgery (4.3%). Conditions such as degenerative disc disease, spinal deformities, fractures, and spinal stenosis often require surgical interventions, including spinal fusion procedures, to stabilize the spine and alleviate pain. Bone grafts and substitutes are essential in these surgeries to promote bone healing, ensure proper fusion of vertebrae, and restore spinal integrity.
Additionally, as per the data provided by the Centre for Disease Control and Prevention (2024), about 1 in 10 adults (11%) aged 65 to 74 years had lost all their teeth. Tooth loss, often caused by periodontal disease, trauma, tooth decay, or aging, results in alveolar bone resorption a condition where the jawbone deteriorates due to the absence of a tooth root to stimulate bone growth. To address this, bone grafts and substitutes are commonly used to regenerate lost bone, providing a stable foundation for dental implants and restoring oral functionality and aesthetics. The rising global prevalence of tooth loss, particularly among the aging population, has created a substantial demand for bone regeneration solutions in dentistry.
Furthermore, the increasing number of product development activities in the region is further going to accelerate the growth of the spinal non-fusion devices market. For instance, in September 2024, Cerapedics Inc., a global orthopedics company focused on improving bone repair treatments, announced that the U.S. Food and Drug Administration (FDA) had approved an expanded use and labeling for its i-FACTOR P-15 Peptide Enhanced Bone Graft. With this approval, i-FACTOR was cleared for use in single-level anterior cervical discectomy fusion (ACDF) procedures. It could be used with an allograft bone ring or combined with a PEEK, titanium alloy, or PEEK/titanium fusion interbody device approved by the FDA for cervical spine use, along with supplemental anterior plate fixation.
Thus, the above-mentioned factors are expected to escalate the Bone Graft and Substitutes market in North America.
Bone Graft and Substitutes Market Key Players:
Some of the key market players operating in the bone graft and substitutes market include Medtronic plc., Johnson & Johnson, Smith & Nephew, Stryker, Dentsply Sirona Inc., Globus Medical Inc., Baxter, Arthrex, Inc., OssDsign, Orthofix Medical Inc., Xtant Medical Holdings, Inc., Halma, B. Braun Melsungen AG, Exactech Inc., Zimmer Biomet, Institut Straumann AG, Southern Implants, MedBone Biomaterials, Noraker, BioHorizons, and others.
Recent Developmental Activities in the Bone Graft and Substitutes Market:
- In September 2022, Ivory Dentin Graft, the first and only dentin graft, received CE mark approval.
- In August 2022, Orthofix Medical and CGBio announced the strategic partnership agreement for Novosis rhBMP-2 bone graft solutions.
Key Takeaways From the Bone Graft and Substitutes Market Report Study
- Market size analysis for current bone graft and substitutes size (2024), and market forecast for 8 years (2025 to 2032)
- Top key product/technology developments, mergers, acquisitions, partnerships, and joint ventures happened over the last 3 years.
- Key companies dominating the bone graft and substitutes market.
- Various opportunities available for the other competitors in the bone graft and substitutes market space.
- What are the top-performing segments in 2024? How these segments will perform in 2032?
- Which are the top-performing regions and countries in the current bone graft and substitutes market scenario?
- Which are the regions and countries where companies should have concentrated on opportunities for bone graft and substitute market growth in the coming future?
Target Audience Who Can be Benefited From This Bone Graft and Substitutes Market Report Study
- Bone graft and substitute product providers
- Research organizations and consulting companies
- Bone graft and substitutes-related organizations, associations, forums, and other alliances
- Government and corporate offices
- Start-up companies, venture capitalists, and private equity firms
- Distributors and traders dealing in bone grafts and substitutes
- Various end-users who want to know more about the bone graft and substitutes market and the latest technological developments in the bone graft and substitutes market.