Chondrosarcoma Market Summary
Chondrosarcoma Insights and Trends
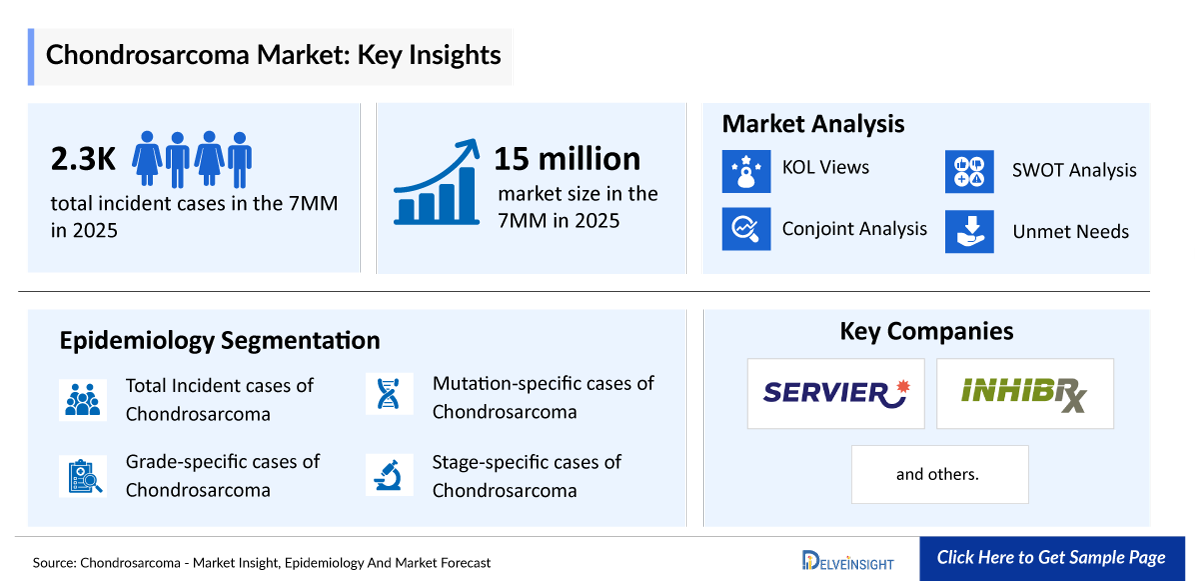
- According to DelveInsight’s analysis, the chondrosarcoma market size was found to be ~USD 15 million in the leading markets (the United States, the EU4 (Germany, France, Italy, and Spain), the United Kingdom, and Japan) in 2025.
- Chondrosarcoma, a type of bone cancer, is categorized into four Grades: I, II, III, and IV. The majority of cases fall within Grade II.
- Chondrosarcoma primarily affects adults and is the second most common primary solid tumor of bone after osteogenic sarcoma. It is classified as primary or secondary depending upon whether the tumor arises de novo or develops in a preexisting benign cartilage neoplasm.
- IDH mutations are found in 50-70% of chondrosarcomas.
- Since there are no approved therapies, the development of molecule-targeting drugs and immunotherapy that improve clinical outcomes and reduce recurrence in patients with advanced chondrosarcoma may have beneficial and vast market opportunities in the chondrosarcoma landscape.
- Surgery remains the mainstay of treatment. Though radiotherapy and chemotherapy are also preferred in chondrosarcoma treatment, resistance to these conventional approaches leaves wide surgical resection as the only option available for both primary and secondary chondrosarcomas, highlighting a major treatment dilemma.
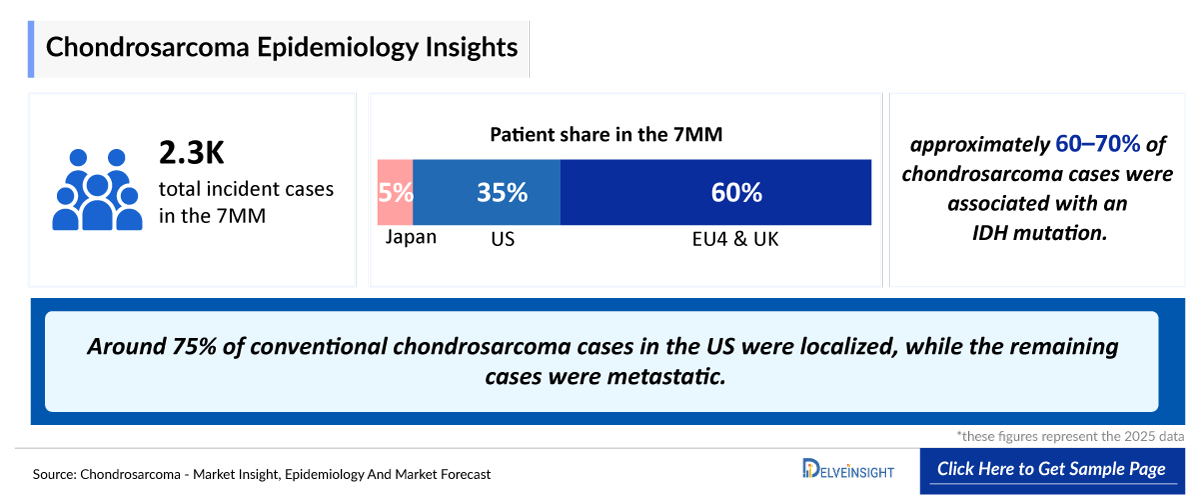
- Novel therapeutic approaches targeting IDH-mutations may sparkle in the chondrosarcoma treatment space and can grab a major share of the untapped market, as nearly 65% of chondrosarcoma cases are related to IDH-mutations.
- Local recurrence and metastatic progression are common in aggressive subtypes, with poor long-term survival rates in metastatic disease.
- Servier and Inhibrx are emerging as key players in the chondrosarcoma landscape, with differentiated targeted approaches in clinical development. Servier is advancing ivosidenib in the Phase III CHONQUER trial for IDH1-mutant chondrosarcoma, targeting tumor metabolism and addressing a genetically defined patient subset with significant unmet need. In parallel, Inhibrx is developing INBRX-109 (ozekibart), a DR5 agonist antibody currently in Phase II trial.
Chondrosarcoma Market Size and Forecast in the 7MM
- 2025 Chondrosarcoma Market Size: ~USD 15 million
- Chondrosarcoma Growth Rate (2026–2036): 16% CAGR
DelveInsight's ‘Chondrosarcoma – Market Insights, Epidemiology and Market Forecast – 2036’ report delivers an in-depth understanding of the chondrosarcoma, historical and forecasted epidemiology, as well as the chondrosarcoma market trends in the United States, EU4 (Germany, Spain, Italy, and France), and the United Kingdom, and Japan.
The Chondrosarcoma market report delivers a comprehensive analysis of the current treatment landscape, including standards of care, clinical practices, and evolving therapeutic algorithms. It evaluates chondrosarcoma patient burden trends, revenue & market share dynamics, peak patient share & therapy uptake analysis, and provides an in-depth market size assessment, and growth rate projections (Historical & Forecast 2022–2036) across global regions. The report highlights key unmet medical needs in chondrosarcoma and maps the competitive and clinical landscape to uncover high‑value opportunities, providing a clear outlook on future market growth potential.
|
Scope of the Chondrosarcoma Market Insights Report | |
|
Study Period |
2022–2036 |
|
Historical Year |
2022–2025 |
|
Forecast Period |
2026–2036 |
|
Base Year |
2026 |
|
Geographies Covered |
|
|
Chondrosarcoma Market CAGR (Forecast period) |
16% (2026–2036) |
|
Chondrosarcoma Epidemiology Segmentation Analysis |
|
|
Chondrosarcoma Companies |
|
|
Chondrosarcoma Therapies |
|
|
Chondrosarcoma Market |
Segmented by
|
|
Analysis |
|
Key Factors Driving the Chondrosarcoma Market
Rising Disease Incidence
Increased detection rates due to advanced imaging and better diagnostic sensitivity are leading to higher reported cases, enhancing the treated patient population.
Emergence of Targeted Therapies
Research into targeted agents, including IDH1/2 inhibitors (Ivosidenib), EZH2 inhibitors, and DR5-targeted agents like INBRX-109, is transforming treatment for unresectable cases.
Emerging Chondrosarcoma Competitive Landscape
Some of the chondrosarcoma drugs in clinical trials include ivosidenib (Servier), INBRX-109 (Inhibrx), and others.
Chondrosarcoma Understanding and Treatment Algorithm
Chondrosarcoma Overview and Diagnosis
Chondrosarcoma comprises a group of locally aggressive or malignant cartilaginous matrix-producing neoplasms with diverse morphological features and clinical behavior. They predominantly arise in bones derived from endochondral ossification, the most common sarcoma of bone in patients above the age of 20. It predominantly affects individuals in the fourth to sixth decades of life; the most commonly involved sites are the pelvic bones, followed by the femur, humerus, and ribs.
Chondrosarcoma diagnosis involves a thorough process beginning with a review of the patient's medical history and a physical examination to identify potential signs and symptoms. Imaging tests such as X-rays, CT scans, and MRI scans are then utilized to visualize the tumor and assess its size, location, and extent. If a tumor suspicious of chondrosarcoma is detected, a biopsy is performed to obtain a tissue sample for microscopic examination by a pathologist, confirming the diagnosis. The tumor tissue is further graded on a scale from I to IV based on its appearance under the microscope, aiding in determining its aggressiveness and prognosis. Staging tests may follow to assess the cancer's spread.
Further details are provided in the report.
Current Chondrosarcoma Treatment Landscape
The grade of malignancy and the underlying subentity strongly determine the therapy and prognosis of chondrosarcoma. However, for all types of chondrosarcoma, the premise for curative treatment is adequate surgery. Only the wide compartmental tumor resection, according to Enneking, can control the tumor locally and systemically in a curative setting in high and intermediate-grade tumors. Neoadjuvant or adjuvant chemotherapy may provide a survival advantage in dedifferentiated chondrosarcoma. The chemotherapy regimen is often based on a combination of doxorubicin and cisplatin or ifosfamide. The pipeline therapies include INBRX-109, ivosidenib, and others.
Further details related to country-based variations are provided in the report.
Chondrosarcoma Unmet Needs
The section “unmet needs of chondrosarcoma” outlines the critical gaps between the current state of patient care, diagnosis, and the ideal & effective management of the disease. It highlights the obstacles experienced by patients, clinicians, and researchers and identifies potential solutions for future progress.
- Lack of effective systemic therapy
- Radiation resistance
- Lack of treatment guidelines for rare subtype management
- Diagnostic Challenges, and others…..
Note: Comprehensive unmet needs insights in chondrosarcoma and their strategic implications are provided in the full report.
Chondrosarcoma Epidemiology
Key Findings from Chondrosarcoma Epidemiological Analysis and Forecast
- According to DelveInsight’s estimates, in 2025, the total number of incident cases of chondrosarcoma in the 7MM was ~2,300.
- Among the stage-specific incident cases of chondrosarcoma, localized cases contribute the most in Japan.
- Conventional chondrosarcomas constitute approximately 80% of all chondrosarcoma cases, making them the most prevalent subtype.
- IDH mutations are detected in approximately 50% of chondrosarcoma patients in Japan.
Chondrosarcoma Drug Analysis & Competitive Landscape
The chondrosarcoma drug chapter provides a detailed, market-focused review of approved therapies and the emerging pipeline across Phase III-II clinical trials. It covers the mechanism of action, clinical trial data, regulatory approvals, patents, collaborations, strategic partnerships, upcoming key catalysts for each therapy, along with their advantages, limitations, and recent developments. This section offers critical insights into the chondrosarcoma treatment landscape, supporting market assessment, competitive analysis, and growth forecasting for the chondrosarcoma therapeutics market.
Approved Therapies for Chondrosarcoma
There is no approved drug for the treatment of Chondrosarcoma. As per the updated NCCN guidelines, ivosidenib has been added as a treatment option for patients with IDH1-mutant conventional or dedifferentiated chondrosarcoma; however, for all types of chondrosarcoma, the premise for curative treatment is adequate surgery.
Note: Detailed market assessment will be provided in the final report.
Chondrosarcoma Pipeline Analysis
Ivosidenib: Servier
Ivosidenib is an isocitrate dehydrogenase-1 (IDH1) inhibitor developed by Servier. A Phase III, multicenter, double-blind, randomized, placebo-controlled study of ivosidenib is underway for participants aged 18 years and older with locally advanced or metastatic conventional chondrosarcoma harboring an IDH1 mutation, whether untreated or previously treated with one systemic treatment regimen. These mutations lead to abnormal forms of IDH1 proteins in the cancer cells.
INBRX-109 (OZEKIBART): Inhibrx
INBRX-109 is the most advanced therapeutic candidate of Inhibrx and is designed to target tumor-biased direct cell death induction by DR5 activation in numerous cancer types. INBRX-109 is a tetravalent DR5 agonistic antibody that can potentize DR5 through efficient receptor clustering, causing cell death. Inhibrx plans to file a BLA in Q2 of 2026. Currently, the drug is being evaluated in a potentially registration-enabling Phase II trial.
Note: A detailed emerging therapies assessment will be provided in the final report.
Competitive Landscape of Pipeline Drugs | ||||||
|
Drug Name |
Company |
Highest Phase |
Indication |
RoA |
MoA |
Anticipated Launch in the US |
|
Ivosidenib |
Servier |
III |
Locally Advanced or Metastatic Conventional Chondrosarcoma |
Oral |
IDH1 inhibitor |
Information is available in the full report |
|
INBRX-109 (OZEKIBART) |
Inhibrx |
II |
Conventional Chondrosarcoma |
IV |
Death receptor 5 (DR5) agonist |
Information is available in the full report |
|
Note: Launch insights are provisional and may change with future report updates or the occurrence of major key catalysts. | ||||||
Chondrosarcoma Key Players, Emerging Companies
- Servier
- Inhibrx and others
Chondrosarcoma Drug Updates
- In October 2025, Inhibrx announced positive data from ozekibart (INBRX-109) registration-enabling trial in advanced or metastatic, unresectable chondrosarcoma. Ozekibart has key data or milestone events expected in 2026.
- In January 2025, Servier announced an update of its Phase III program evaluating TIBSOVO (ivosidenib tablets) in isocitrate dehydrogenase 1 (IDH1)-mutated cancers. The first patient has been enrolled in the CHONQUER study, a pivotal Phase III clinical trial evaluating the efficacy and safety of TIBSOVO versus placebo in patients with IDH1-mutated conventional chondrosarcoma.
Chondrosarcoma Market Outlook
The current management of chondrosarcoma depends on the type, the grade, and the location of the tumor, while the best predictor of clinical behavior is the grading system from I to III. It is well-established that surgical treatment is the best and the only treatment for chondrosarcoma, as other treatments, such as radiotherapy and chemotherapy, are rendered unresponsive and refractory. Curettage, radical resection, and amputation are the primary surgical treatment options, while these procedures often require considerable judgment and are based on factors such as the size and site of the lesion and its degree of malignancy.
However, a major roadblock while estimating the revenue generated by systemic agents in chondrosarcoma is chemotherapy, which is generally ineffective in conventional chondrosarcoma, and there is no standard systemic therapy for advanced conventional chondrosarcoma either, unlike other solid tumors.
The rarity of these cancers makes randomized clinical trials to evaluate the efficacy of systemic therapy more challenging. Current chemotherapy recommendations include cisplatin and doxorubicin, extrapolated from recommended osteosarcoma and Ewing sarcoma treatment regimens. Other combinations of Gemcitabine, Ifosfamide, Dasatinib, and Pazopanib have also been used but with limited success, especially in dedifferentiated and metastatic cases.
Key players like Inhibrx, Servier, and others are evaluating their lead candidates in different stages of clinical development.
- Among the 7MM, the US accounted for the largest market size of chondrosarcoma. i.e., USD ~10 million in 2025.
- The entry of mid-stage candidates such as INBRX-109 (OZEKIBART) is expected to intensify competition in the chondrosarcoma treatment landscape during the latter half of the forecast period.
Further details will be provided in the report….
Drug Class/Insights into Leading Emerging and Marketed Therapies in Condrosarcoma (2022–2036 Forecast)
The chondrosarcoma market comprises humanized IgG1 agonist antibodies, and small molecules, each targeting different aspects of chondrosarcoma.
- Humanized IgG1 agonist antibody: It is designed to activate specific receptors involved in tumor cell death or immune stimulation rather than directly inhibiting growth pathways. INBRX-109 (OZEKIBART) targets death receptor 5 (DR5) on tumor cells. By binding and activating DR5, this antibody mimics the natural ligand (TRAIL), triggering the extrinsic apoptotic pathway through caspase activation, leading to programmed tumor cell death.
- Small molecules: Ivosidenib is a small-molecule targeted therapy that selectively inhibits mutant isocitrate dehydrogenase 1 (IDH1), an enzyme that, when mutated, acquires a neomorphic activity converting α-ketoglutarate into the oncometabolite 2-hydroxyglutarate (2-HG).
Chondrosarcoma Drug Uptake
This section focuses on the uptake rate of potential drugs expected to be launched in the market during the forecast period (2026–2036). The analysis covers the chondrosarcoma drug’s uptake, performance at peak, factors affecting performance during prime years of growth, patient uptake by therapy, and anticipated sales generated by each drug.
The uptake of therapies in chondrosarcoma is expected to vary based on clinical positioning, mechanism of action, and stage of development. Ongoing clinical trials are evaluating the clinical activity of novel IDH inhibitors. AG-120 or ivosidenib, an oral IDH2 inhibitor, is currently being tested in a Phase III trial to see whether AG-120 is an effective and safe treatment for people with advanced/metastatic or recurrent chondrosarcoma that has an IDH1 mutation. The IDH inhibitor AG-120 is also under clinical evaluation in a Phase II study of advanced solid tumors that harbor an IDH1 and/or IDH2 mutation, including gliomas, cholangiocarcinomas. Moreover, an ongoing Phase Ib, open-label, single-center, nonrandomized clinical trial evaluates the toxicity and efficacy of metformin in combination with chloroquine in IDH1/2 mutated patients with a glioma, intrahepatic cholangiocarcinoma, or chondrosarcoma.
Detailed insights of emerging therapies' drug uptake are included in the report.
Market Access and Reimbursement of Approved Therapies in Chondrosarcoma
The report further provides detailed insights on the country-wise accessibility and reimbursement scenarios, cost-effectiveness scenario of approved therapies, programs making accessibility easier and out-of-pocket costs more affordable, insights on patients insured under federal or state government prescription drug programs, etc.
Reimbursement is a crucial factor that affects the drug’s access to the market. Often, the decision to reimburse comes down to the price of the drug relative to the benefit it produces in treated patients. To reduce the healthcare burden of these high-cost therapies, many payment models are being considered by payers and other industry insiders.
NOTE: Further Details are provided in the final report….
Chondrosarcoma Therapies Price Scenario & Trends
Pricing and analogue assessment of chondrosarcoma therapies highlights evolving price dynamics structures. This section summarizes the cost of approved treatments, the closest and most appropriate analogue selection for emerging therapies, and understanding of how pricing influences market access, adherence, and long-term uptake.
Industry Experts and Physician Views for Chondrosarcoma
To keep up with chondrosarcoma market trends, we take Key Opinion Leaders (KOLs) and Subject Matter Experts (SMEs) opinions working in the domain through primary research to fill the data gaps and validate our secondary research. Industry experts were contacted for insights on the chondrosarcoma emerging therapies, evolving treatment landscape, patient adherence to conventional therapies, therapy switching trends, drug adoption and uptake, accessibility challenges, and epidemiology and real-world prescription patterns in chondrosarcoma, including MD, PhD, Instructor, Postdoctoral Researcher, Professor, Researcher, and others.
DelveInsight’s analysts connected with 15+ KOLs to gather insights at the country level. Centers such as the University of Washington, University of Würzburg, German Cancer Research Center (DKFZ) & KiTZ Heidelberg, and University College London Hospital, etc. were contacted. Their opinion helps understand and validate current and emerging chondrosarcoma therapies, highlight unmet medical needs, provide epidemiological context, and support strategic decisions for market access, therapy adoption, and pipeline prioritization in chondrosarcoma.
|
Region |
Key Opinion Leaders (KOLs) and Subject Matter Experts (SMEs) |
|
United States |
“In a small study of fewer than 20 patients, trabectedin controlled the disease for up to two years in some patients who had previously failed standard chemotherapy. Moving forward, there will first need to be collaboration between institutions and pharmaceutical companies to determine if similar results have been seen before.” |
|
Germany |
IDH1 inhibitors have demonstrated prolonged stability of disease with potential tumor shrinkage in patients with chondrosarcoma. Studies evaluating IDH1/2 inhibitors in this setting are in early phases, but these data suggest that metabolic therapy may be an effective approach in treating patients with chondrosarcoma in the future.” |
Qualitative Analysis: SWOT and Conjoint Analysis
We perform qualitative and market Intelligence analysis using various approaches, such as SWOT analysis and conjoint analysis.
In the SWOT analysis of chondrosarcoma, strengths, weaknesses, opportunities, and threats in terms of disease diagnosis, patient awareness, patient burden, competitive landscape, cost-effectiveness, and geographical accessibility of therapies are provided.
Conjoint analysis analyzes emerging therapies based on relevant attributes such as safety, efficacy, frequency of administration, route of administration, and order of entry. Scoring is given based on these parameters to analyze the effectiveness of therapy.
The team of analysts analyzes promising emerging therapies based on relevant attributes such as safety, efficacy, frequency of administration, route of administration, and order of entry. In efficacy, the trial’s primary and secondary outcome measures are evaluated, whereas the therapies’ safety is evaluated, wherein the acceptability, tolerability, and adverse events are mainly observed. In addition, the scoring is also based on the route of administration, order of entry, probability of success, and the addressable patient pool for each therapy. According to these parameters, the final weightage score and the ranking of the emerging therapies are decided.
Scope of the Chondrosarcoma Market Insights Report
- The report covers a segment of key events, an executive summary, a descriptive overview of chondrosarcoma, explaining its causes, signs and symptoms, pathogenesis, and currently available treatments.
- Comprehensive insight has been provided into the epidemiology segments and forecasts, the future growth potential of the diagnosis rate, and disease progression along treatment guidelines.
- Additionally, an all-inclusive account of both the current and emerging treatments, along with the elaborate profiles of late-stage and prominent therapies, will have an impact on the current treatment landscape.
- A detailed review of the chondrosarcoma market, historical and forecasted market size, market share by therapies, detailed assumptions, and rationale behind our approach is included in the report, covering the 7MM drug outreach.
- The report provides an edge while developing business strategies by understanding trends through SWOT analysis and expert insights/KOL views, patient journey, and treatment preferences that help in shaping and driving the 7MM chondrosarcoma market.
Chondrosarcoma Market Insights Report Insights
- Chondrosarcoma Patient Population Forecast
- Chondrosarcoma Therapeutics Market Size
- Chondrosarcoma Pipeline Analysis
- Chondrosarcoma Market Size and Trends
- Chondrosarcoma Market Opportunity (Current and forecasted)
Chondrosarcoma Market Insights Report Key Strengths
- Epidemiology‑based (Epi‑based) Bottom‑up Forecasting
- Artificial Intelligence (AI)-Enabled Market Research Report
- 11-Year Forecast
- Chondrosarcoma Market Outlook (North America, Europe, Asia-Pacific)
- Patient Burden Trends (By Geography)
- Chondrosarcoma Treatment Addressable Market (TAM)
- Chondrosarcoma Competitive Landscape
- Chondrosarcoma) Major Companies Insights
- Chondrosarcoma Price Trends and Analogue Assessment
- Chondrosarcoma Therapies Drug Adoption/Uptake
- Chondrosarcoma Therapies Peak Patient Share Analysis
Chondrosarcoma Market Insights Report Assessment
- Chondrosarcoma Current Treatment Practices
- Chondrosarcoma Unmet Needs
- Chondrosarcoma Clinical Development Analysis
- Chondrosarcoma Emerging Drugs Product Profiles
- Chondrosarcoma Market Attractiveness
- Chondrosarcoma Qualitative Analysis (SWOT and conjoint analysis)
Key Questions Answered in the Chondrosarcoma Market Insights Report
Market Insights
- What was the chondrosarcoma market size, the market size by therapies, the market share (%) distribution in 2025, and what would it look like by 2036? What are the contributing factors for this growth?
- What are the anticipated pricing variations among different geographies for the emerging therapies in the future?
- What can be the future treatment paradigm of chondrosarcoma?
- What are the disease risks, burdens, and unmet needs of chondrosarcoma? What will be the growth opportunities across the 7MM concerning the patient population with chondrosarcoma?
- Who is the major future competitor in the market, and how will the competitors affect their market share?
- What are the current options for the treatment of chondrosarcoma? What are the current guidelines for treating chondrosarcoma in the US, Europe, and Japan?
Reasons to Buy Chondrosarcoma Market Insights Report
- The report will help in developing business strategies by understanding the latest trends and changing treatment dynamics driving the chondrosarcoma market.
- Bottom‑up forecasting builds from the affected population to product forecasts, delivering a robust, data‑driven approach ideal for new therapies and novel classes.
- Insights on patient burden/disease incidence, evolution in diagnosis, and factors contributing to the change in the epidemiology of the disease during the forecast years.
- Understand the existing market opportunities in varying geographies and the growth potential over the coming years.
- Identifying strong upcoming players in the market will help devise strategies to help get ahead of competitors.
- Detailed analysis and ranking of class-wise potential current and emerging therapies under the conjoint analysis section to provide visibility around leading classes.
- To understand KOLs’ perspectives on the accessibility, acceptability, and compliance-related challenges of existing treatment to overcome barriers in the future.
- Detailed insights into the unmet needs of the existing market so that the upcoming players can strengthen their development and launch strategy.
- This Artificial Intelligence (AI)‑enabled report summarizes and simplifies complex datasets within the report into clear, actionable insights for stakeholders, investors, and healthcare providers, enabling faster, data‑driven decisions.