Immunotherapy Drugs Market Summary
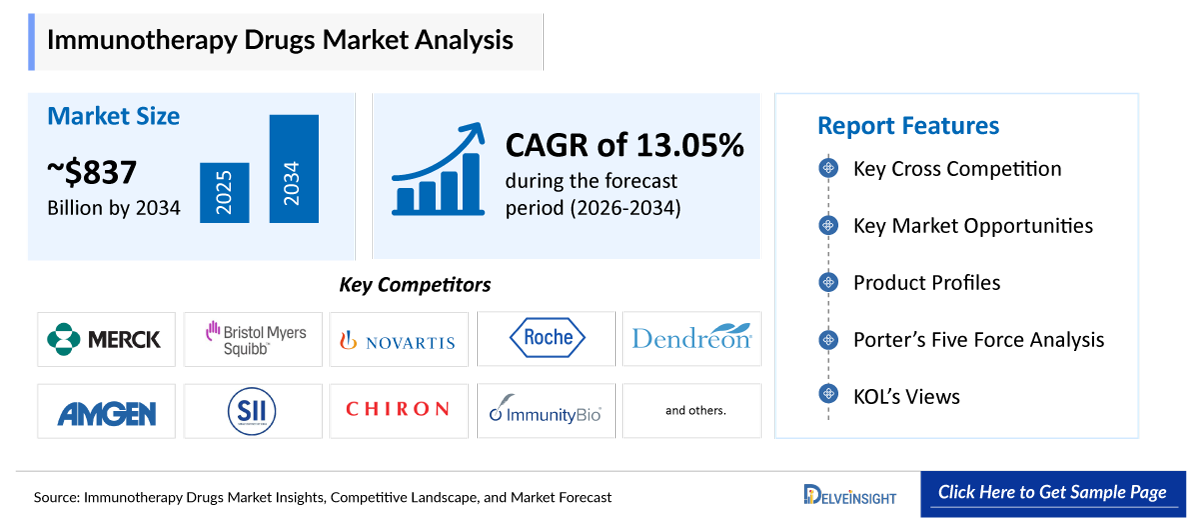
- The global immunotherapy drugs market is expected to increase from USD 280.50 billion in 2025 to USD 837.14 billion by 2034, reflecting strong and sustained growth.
- The global immunotherapy drugs market is growing at a CAGR of 13.05% during the forecast period from 2026 to 2034.
- The market for immunotherapy drugs is primarily driven by the rising global incidence of cancer, rising cases of autoimmune disorders, increasing demand for targeted and personalized treatments, and the superior clinical outcomes of immunotherapies compared to traditional chemotherapy. Additionally, the growing adoption of immune checkpoint inhibitors and cell-based therapies, along with strong investments in research and development by pharmaceutical companies, are further accelerating market growth.Furthermore, supportive regulatory approvals, expanding indications across multiple cancer types, and rising healthcare expenditure in both developed and emerging economies are significantly contributing to the expansion of the immunotherapy drugs market.
- The leading companies operating in the immunotherapy drugs market include Merck & Co., Inc., Bristol-Myers Squibb Company, Novartis AG, F. Hoffmann-La Roche Ltd., Dendreon Pharmaceuticals, Amgen, Serum Institute of India (SII), Chiron Corporation, ImmunityBio, Inc., Kite Pharma Inc., AstraZeneca, Johnson & Johnson and its affiliates, Sanofi, GlaxoSmithKline, Checkpoint Therapeutics, Inc., Adaptimmune, LLC., Iovance Cell Therapy Center, AbbVie, 3M, and others.
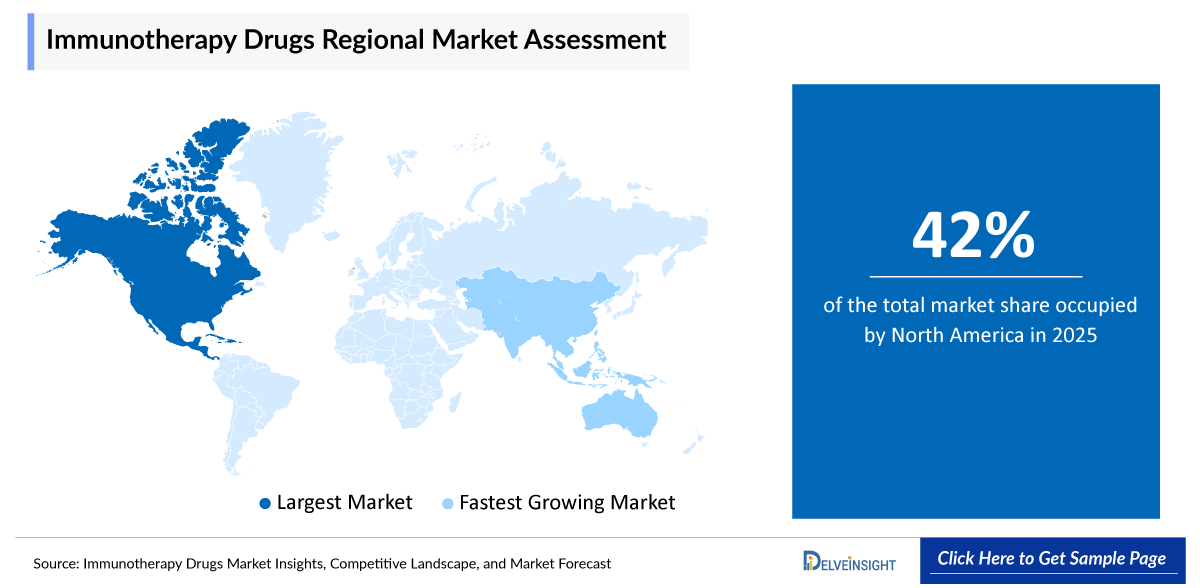
- North America is expected to dominate the immunotherapy drugs market due to the high prevalence of cancer, strong presence of leading biopharmaceutical companies, and advanced healthcare infrastructure in the region. The availability of cutting-edge research facilities, high healthcare expenditure, favorable reimbursement policies, and early adoption of innovative therapies such as immune checkpoint inhibitors and CAR-T Treatments further contribute to market growth. Additionally, supportive regulatory frameworks and continuous product approvals in the United States significantly drive the expansion of the immunotherapy drugs market in North America.
- In the drug type segment of the immunotherapy drugs market, the monoclonal antibodies category is estimated to account for the largest market share in 2025.
Request for unlocking the report of the @ Immunotherapy Drugs Market
Immunotherapy Drugs Market Size and Forecasts
|
Report Metrics |
Details |
|
2025 Market Size |
USD 280.50 billion |
|
2034 Projected Market Size |
USD 837.14 billion |
|
Growth Rate (2026-2034) |
13.05% CAGR |
|
Largest Market |
North America |
|
Fastest Growing Market |
Asia-Pacific |
|
Market Structure |
Moderately Concentrated |
Factors Contributing to the Growth of the Immunotherapy Drugs Market
-
Increasing cases of cancer and autoimmune disorders are leading to a surge in immunotherapy drugs:
The increasing cases of cancer and autoimmune disorders worldwide are significantly driving the demand for immunotherapy drugs. Rising prevalence due to aging populations, lifestyle changes, and improved diagnostic capabilities has expanded the patient pool requiring advanced treatment options. As immunotherapies offer targeted mechanisms and improved long-term outcomes compared to conventional therapies, their adoption continues to grow, leading to a surge in market demand.
-
Rising regulatory approvals, fast-track designations, and new product launches is escalating the market of immunotherapy drugs:
Rising regulatory approvals, fast-track designations, and breakthrough therapy statuses significantly accelerate the growth of the immunotherapy drugs market by shortening development timelines and enabling quicker market entry. These regulatory incentives encourage pharmaceutical companies to invest more in innovative immunotherapies, while faster approvals improve patient access to advanced treatments. Additionally, frequent new product launches expand therapeutic options across various cancer and autoimmune indications, further boosting market expansion.
-
Superior clinical outcomes compared to chemotherapy:
The superior clinical outcomes of immunotherapy drugs compared to traditional chemotherapy are significantly boosting the immunotherapy market. Unlike chemotherapy, which attacks both healthy and cancerous cells, immunotherapies specifically target cancer cells by enhancing the body’s immune response, resulting in improved survival rates, longer-lasting responses, and fewer severe side effects in many patients. These better efficacy and safety profiles increase physician preference and patient acceptance, thereby driving higher adoption and accelerating market growth.
Immunotherapy Drugs Market Report Segmentation
This immunotherapy drugs market report offers a comprehensive overview of the global immunotherapy drugs market, highlighting key trends, growth drivers, challenges, and opportunities. It covers detailed market segmentation by Drug Type (Monoclonal Antibodies {Immune Checkpoint Inhibitors (PD-1, PD-L1, CTLA-4), Targeted Tumor Antigen Antibodies, and Antibody–Drug Conjugates (ADCs)}, Adoptive Cell Therapies {CAR-T Cell Therapies, TCR-T Cell Therapies, and Tumor-Infiltrating Lymphocyte (TIL) Therapies}, Cytokines & Interleukins, Cancer Vaccines, and Others), Route of Administration (Intravenous, Subcutaneous, and Others), Indication (Oncology, Autoimmune Disorders, and Viral Infection), End-Users (Hospitals, Oncology Clinics, and Others), and geography. The report provides valuable insights into the competitive landscape, regulatory environment, and market dynamics across major markets, including North America, Europe, and Asia-Pacific. Featuring in-depth profiles of leading industry players and recent product innovations, this report equips businesses with essential data to identify market potential, develop strategic plans, and capitalize on emerging opportunities in the rapidly growing immunotherapy drugs market.
Immunotherapy drugs are a class of medications that enhance, stimulate, or restore the body’s immune system to fight diseases, particularly cancer and autoimmune disorders. Unlike traditional treatments such as chemotherapy, immunotherapies work by targeting specific components of the immune system or cancer cells, helping the body recognize and destroy abnormal cells more effectively. These drugs include immune checkpoint inhibitors, monoclonal antibodies, cancer vaccines, cytokines, and cell-based therapies.
The Immunotherapy Drugs Market is primarily driven by the rapidly rising global incidence of cancer and the increasing prevalence of autoimmune disorders, both of which have significantly expanded the patient population requiring advanced therapeutic solutions. Furthermore, the growing awareness about early diagnosis and improved access to healthcare services have further increased the demand for effective and long-lasting treatment options.
In addition, the shift toward targeted and personalized medicine has strengthened the adoption of immunotherapies, as these drugs are designed to specifically enhance or modulate the immune system to recognize and attack diseased cells with greater precision. Compared to traditional chemotherapy, immunotherapies often provide superior clinical outcomes, including improved survival rates, durable responses, and relatively manageable side-effect profiles, making them a preferred treatment choice among physicians and patients. The expanding use of immune checkpoint inhibitors, monoclonal antibodies, and advanced cell-based therapies such as CAR-T has further accelerated market growth. Strong investments in research and development by leading pharmaceutical and biotechnology companies, along with strategic collaborations and pipeline expansions, are continuously introducing innovative products into the market.
Furthermore, supportive regulatory frameworks, including fast-track approvals and breakthrough therapy designations, have shortened drug development timelines and facilitated quicker market entry. Expanding treatment indications across multiple cancer types and increasing healthcare expenditure in both developed and emerging economies are also playing a crucial role in driving the sustained growth and expansion of the global immunotherapy drugs market.
Get More Insights into the Report @ Immunotherapy Drugs Market
What are the latest Immunotherapy Drugs Market Dynamics and Trends?
The global immunotherapy drugs market is growing significantly, driven by the increasing incidence of various cancer such as bladder cancer, central nervous system cancer, lung cancer, breast cancer, colorectal cancer, and the increasing prevalence of autoimmune disorders such as diabetes and arthritis. Additionally, the market is further escalated by the increasing product development activites among the key market players and the presence of strong late stage immunotherapy drug pipeline.
According to data provided by the International Agency for Research on Cancer (2025), the estimated number of new bladder cancer cases worldwide in 2025 was 656,192. Projections further indicate that this number is expected to rise to 1,115,460 cases by 2045, reflecting a significant increase over the forecast period. Additionally, as per the same source, the estimated number of new leukaemia cases worldwide in 2025 was 5,15,145. Projections further indicate that this number is expected to rise to 7,46,019 cases by 2045, reflecting a significant increase over the forecast period.
Bladder cancer, particularly non-muscle invasive bladder cancer (NMIBC), has long been treated with immunotherapies such as BCG, and more recently with immune checkpoint inhibitors, creating sustained demand for advanced immunotherapeutic options. Similarly, leukemia especially subtypes such as acute lymphoblastic leukemia (ALL) and chronic lymphocytic leukemia (CLL) has witnessed major therapeutic advancements through monoclonal antibodies, CAR-T cell therapies, and other targeted immune-based treatments. These cancers often require long-term management or combination regimens, increasing treatment duration and overall immunotherapy drug utilization, thereby escalating the overall market.
Additionally, the superior clinical outcomes of immunotherapy drugs compared to traditional chemotherapy have played a transformative role in boosting the global immunotherapy market. For instance, in January 2026, Bristol Myers Squibb announced that Nivolumab in combination with chemotherapy before surgery (neoadjuvant therapy), followed by nivolumab alone after surgery (adjuvant therapy), was recommended by the National Institute for Health and Care Excellence (NICE) for the treatment of adult patients with resectable non-small cell lung cancer (NSCLC). Furthermore, as per the data provided by the Johns Hopkins (2025), in the CheckMate-816 clinical trial, patients who received nivolumab plus chemotherapy had significantly better long-term survival and remission outcomes compared with those who received chemotherapy alone, with 24% achieving complete remission and many patients showing 95% five-year survival among complete responders. This demonstrates that immunotherapy can not only extend survival but also improve the quality of response, reinforcing confidence among clinicians and patients and driving broader adoption of immunotherapy regimens, which in turn accelerates market growth.
Thus, the factors mentioned above are expected to boost the overall market of immunotherapy drugs during the forecast period.
However, Immune-related adverse events (irAEs) and stringent regulatory approval processes act as significant limiting factors for immunotherapy drugs market. Immunotherapies can overstimulate the immune system, leading to serious side effects such as inflammation of vital organs, autoimmune reactions, and other immune-mediated complications, which may require treatment discontinuation and careful monitoring. At the same time, strict regulatory requirements demand extensive clinical trials to demonstrate long-term safety and efficacy, resulting in high development costs and prolonged approval timelines. These challenges can delay market entry, restrict patient eligibility, and ultimately slow the overall growth of the immunotherapy drugs market.
Immunotherapy Drugs Market Segment Analysis
Immunotherapy Drugs Market Drug Type (Monoclonal Antibodies {Immune Checkpoint Inhibitors (PD-1, PD-L1, CTLA-4), Targeted Tumor Antigen Antibodies, and Antibody–Drug Conjugates (ADCs)}, Adoptive Cell Therapies {CAR-T Cell Therapies, TCR-T Cell Therapies, and Tumor-Infiltrating Lymphocyte (TIL) Therapies}, Cytokines & Interleukins, Cancer Vaccines, and Others), Route of Administration (Intravenous, Subcutaneous, and Others), Indication (Oncology, Autoimmune Disorders, and Viral Infection), End-Users (Hospitals, Oncology Clinics, and Others), and Geography (North America, Europe, Asia-Pacific, and Rest of the World)
By Drug Type: Under Monoclonal Antibody Segment, Immune Checkpoint Inhibitors Category Projected to Register Largest Revenue Share
In the drug type segment of the immunotherapy drugs market, under monoclonal antibody segment, immune checkpoint inhibitors category is estimated to account for the largest market share of 66% in 2025, due to their proven clinical efficacy, broad therapeutic applicability, and strong commercial performance. Immune checkpoint inhibitors such as PD-1, PD-L1, and CTLA-4 inhibitors have revolutionized cancer treatment by enhancing the body’s immune response against tumor cells, leading to improved overall survival and durable responses across multiple cancer types including lung cancer, melanoma, renal cell carcinoma, and bladder cancer.
Their expanding indications, combination therapy approvals, and use in both early-stage and advanced cancers have significantly increased patient eligibility and treatment duration, thereby driving higher sales volumes. Additionally, the increasing product development activites are further escalating the overall market of the segment. For instance, in October 2023, Merck & Co. announced that the U.S. FDA had approved Keytruda, its anti-PD-1 therapy, for the treatment of patients with resectable non-small cell lung cancer (NSCLC) (tumors ≥4 cm or node-positive). The approval covered its use in combination with platinum-containing chemotherapy as neoadjuvant treatment, followed by single-agent use as adjuvant therapy after surgery. With this approval, Keytruda had six indications in NSCLC across both metastatic and earlier-stage settings.
From a commercial perspective, blockbuster products like nivolumab, pembrolizumab, and atezolizumab generate multi-billion-dollar annual revenues, contributing substantially to the global oncology market. Pharmaceutical companies continue to invest heavily in lifecycle management strategies, including label expansions, combination regimens, and biomarker-based targeting, which further strengthen market dominance. Additionally, favorable reimbursement policies in developed regions, strong physician adoption, and ongoing clinical trials exploring new indications and earlier lines of therapy reinforce the leading market position of immune checkpoint inhibitors within the monoclonal antibody segment of immunotherapy drugs.
By Route of Administration: Intravenous Category Dominates the Market
In the route of administration segment of the immunotherapy drugs market, intravenous category are projected to hold the largest market share of 55% in 2025. The intravenous (IV) route of administration is significantly boosting the overall immunotherapy drug market because most leading immunotherapies particularly monoclonal antibodies, immune checkpoint inhibitors, and certain cell-based therapies are biologics that require controlled systemic delivery directly into the bloodstream. IV administration ensures rapid bioavailability, precise dosing, and optimal therapeutic concentrations, which are critical in oncology and severe autoimmune conditions. Major blockbuster drugs such as anti-PD-1 and anti-PD-L1 therapies are predominantly delivered intravenously in hospital or specialty infusion center settings, driving higher treatment costs per cycle and increasing overall market value.
Additionally, IV-based regimens often involve combination therapies (e.g., immunotherapy plus chemotherapy), further expanding revenue per patient. The infrastructure growth of oncology infusion centers, improved reimbursement frameworks, and increasing global cancer incidence have collectively accelerated IV immunotherapy adoption. Moreover, because IV biologics typically have longer treatment durations and higher price points compared to oral or subcutaneous alternatives, they contribute disproportionately to total market revenue. As a result, the intravenous segment continues to dominate and expand the immunotherapy market by combining clinical efficacy, premium pricing, and structured healthcare delivery models.
Additionally, the increase in approvals and launches of intravenous immunotherapy drugs is further boosting the overall market. For instance, in October 2025, GSK plc announced that the U.S. FDA had approved Blenrep (belantamab mafodotin-blmf) intravenous therapy in combination with bortezomib and dexamethasone (BVd) for adult patients with relapsed or refractory multiple myeloma who had received at least two prior lines of therapy, including a proteasome inhibitor and an immunomodulatory agent. Thus, the factors mentioned above are expected to boost the market of intravenous category therby escalating the overall market of immunotherapy drugs.
By Indication: Oncology Category Dominates the Market
In the indication segment of the immunotherapy drugs market, oncology category is projected to hold the largest market share of 75% in 2025. Oncology is significantly boosting the overall market of immunotherapy devices due to the rising global incidence of cancer and the increasing adoption of advanced immune-based treatment approaches.
According to data provided by the International Agency for Research on Cancer (2025), the estimated number of new cancer cases worldwide in 2045 would reach upto 32.6 million. Cancer treatment increasingly relies on immunotherapies such as CAR-T cell therapy, immune checkpoint inhibitors, cancer vaccines, and tumor-infiltrating lymphocyte (TIL) therapies, all of which require specialized devices for cell collection (apheresis systems), cell processing and expansion, cryopreservation, infusion pumps, and monitoring systems. The growth of personalized and cell-based therapies in oncology has expanded demand for sophisticated laboratory equipment, automated cell manufacturing platforms, and hospital-based infusion infrastructure.
Additionally, combination regimens and longer treatment durations in cancer care increase device utilization rates, further driving revenue. The expansion of oncology treatment centers, improvements in reimbursement policies, and growing investments in cancer immunotherapy research and clinical trials are also accelerating device adoption. As oncology remains the largest application segment for immunotherapy, it continues to propel technological innovation and capital investment in immunotherapy-related devices, thereby strengthening overall market growth.
By End-Users: Hospitals Category Driving the Market
Hospitals are significantly boosting the overall market of immunotherapy drugs as they serve as the primary administration centers for advanced biologics, particularly intravenous checkpoint inhibitors, CAR-T cell therapies, and combination regimens used in oncology and autoimmune diseases. Most immunotherapies require specialized infrastructure, including infusion centers, oncology departments, intensive monitoring facilities, and trained healthcare professionals, which are predominantly available in hospital settings. The rising number of hospital-based cancer diagnoses, increasing patient admissions for complex immune-related disorders, and expansion of oncology and transplant units have led to higher immunotherapy adoption rates.
Additionally, hospitals benefit from structured reimbursement systems and bulk procurement agreements, enabling consistent purchasing of high-cost biologics. Participation in clinical trials and early adoption of newly approved immunotherapies further strengthen hospital-driven demand. As immunotherapy treatments often involve repeated cycles and long-term management, hospitals generate sustained revenue streams, thereby playing a central role in accelerating overall immunotherapy drug market growth.
Immunotherapy Drugs Market Regional Analysis
North America Immunotherapy Drugs Market Trends
North America is poised to secure the largest share of 42% in the global immunotherapy drugs market in 2025 due to the high prevalence of cancer, strong presence of leading biopharmaceutical companies, and advanced healthcare infrastructure in the region. The availability of cutting-edge research facilities, high healthcare expenditure, favorable reimbursement policies, and early adoption of innovative therapies such as immune checkpoint inhibitors and CAR-T treatments further contribute to market growth. Additionally, supportive regulatory frameworks and continuous product approvals in the United States significantly drive the expansion of the immunotherapy drugs market in North America.
According to data provided by the International Agency for Research on Cancer (2025), the estimated number of new bladder cancer cases in the United States in 2025 was 86,588. Projections further indicate that this number is expected to rise to 1,25,373 cases by 2045, reflecting a significant increase over the forecast period. Additionally, as per the same source, the estimated number of multiple myeloma cases in the United States in 2025 was 34,487. Projections further indicate that this number is expected to rise to 45,338 cases by 2045, reflecting a significant increase over the forecast period.
In bladder cancer, particularly advanced or metastatic urothelial carcinoma, immune checkpoint inhibitors such as PD-1 and PD-L1 inhibitors have become standard treatment options, increasing demand for monoclonal antibody therapies. Similarly, multiple myeloma has witnessed major advancements with immunotherapies including monoclonal antibodies, antibody–drug conjugates, bispecific antibodies, and CAR-T cell therapies, especially in relapsed or refractory cases where patients require multiple lines of treatment. The chronic and recurrent nature of both diseases leads to prolonged therapy duration and combination regimens, thereby increasing overall drug utilization and revenue generation.
Furthermore, the increasing product approvals across the regions are further escalating the overall immunotherapy drugs market. For instance, in August, 2024, the Food and Drug Administration granted accelerated approval to afamitresgene autoleucel (TECELRA, Adaptimmune, LLC), a melanoma-associated antigen A4 (MAGE-A4)-directed genetically modified autologous T cell immunotherapy, for adults with unresectable or metastatic synovial sarcoma who have received prior chemotherapy. Thus, the factors mentioned above are expected to boost the overall immunotherapy drugs market across the region.
Europe Immunotherapy Drugs Market Trends
Europe is increasingly poised to play a leading role in the overall immunotherapy drugs market due to its strong healthcare infrastructure, rising cancer prevalence, supportive regulatory framework, and growing investment in biopharmaceutical research and development. The presence of major pharmaceutical companies, expanding clinical trial activities, and early adoption of advanced therapies such as monoclonal antibodies and CAR-T cell treatments are accelerating market growth.
According to data provided by the International Agency for Research on Cancer (2025), the estimated number of new breast cancer cases in Europe in 2025 was 5,64,059. Projections further indicate that this number is expected to rise to 6,00,704 cases by 2045, reflecting a significant increase over the forecast period. Breast cancer is significantly boosting the overall immunotherapy drugs market due to its high global prevalence and the expanding use of immune checkpoint inhibitors and targeted biologics in treatment, particularly for Triple Negative Breast Cancer (TNBC). The growing adoption of PD-1/PD-L1 inhibitors in combination with chemotherapy has increased drug utilization rates and expanded eligible patient populations.
Additionally, the increasing product development activities are further boosting the overall market of the immunotherapy drugs market. For instance, in March 2025, Bristol Myers Squibb announced that the European Commission (EC) has approved Opdivo® (nivolumab) plus Yervoy® (ipilimumab) for the first-line treatment of adult patients with unresectable or advanced hepatocellular carcinoma (HCC). Thus, the factors mentioned are expected to boost the Immunotherapy Drugs Market in Europe.
Asia-Pacific Immunotherapy Drugs Market Trends
The Asia-Pacific region is rapidly emerging as one of the fastest-growing and most influential markets in the global immunotherapy drugs landscape due to its large patient population, rising cancer incidence, improving healthcare infrastructure, and increasing government investment in biotechnology. According to data provided by the International Agency for Research on Cancer (2025), the estimated number of new Non-Hodgkin lymphoma cases in Asia in 2025 was 2,52,943. Projections further indicate that this number is expected to rise to 3,84,359 cases by 2045. Immunotherapies such as monoclonal antibodies (e.g., anti-CD20 therapies), CAR-T cell therapies, and bispecific antibodies have become standard or emerging treatment options for various NHL subtypes. The recurrent and relapsed nature of the disease often requires multiple lines of therapy, increasing long-term drug utilization and revenue generation.
Additionally, the expanding access to advanced oncology treatments, growing awareness of immunotherapy, and the presence of domestic biopharmaceutical manufacturers are accelerating market penetration. Additionally, supportive regulatory reforms, increasing clinical trial activity, and rising healthcare expenditure in countries such as China, Japan, and India are further strengthening the region’s position as a key growth driver in the global immunotherapy market.
Who are the major players in the immunotherapy drugs market?
The following are the leading companies in the immunotherapy drugs market. These companies collectively hold the largest market share and dictate industry trends.
- Merck & Co., Inc.
- Bristol-Myers Squibb Company
- Novartis AG
- F. Hoffmann-La Roche Ltd.
- Dendreon Pharmaceuticals
- Amgen
- Serum Institute of India (SII)
- Chiron Corporation
- ImmunityBio, Inc.
- Kite Pharma Inc.
- AstraZeneca
- Johnson & Johnson and its affiliates
- Sanofi
- GlaxoSmithKline
- Checkpoint Therapeutics, Inc.
- Adaptimmune, LLC.
- Iovance Cell Therapy Center
- AbbVie
- 3M
How is the competitive landscape shaping the Immunotherapy Drugs market?
The competitive landscape of the immunotherapy drugs market is marked by robust competition among a mix of large, diversified pharmaceutical players and specialized biotech innovators, driving rapid innovation and frequent strategic collaborations. A few well-established multinational companies, such as Roche, Merck & Co., Bristol-Myers Squibb, Novartis, and AstraZeneca, hold significant market share with blockbuster immunotherapies (e.g., checkpoint inhibitors, CAR-T therapies), resulting in a moderately high market concentration at the top tier.
However, the landscape is far from monopolistic: numerous mid-sized firms and emerging biotechs are actively advancing novel mechanisms of action (including bispecific antibodies, cancer vaccines, and next-generation cell therapies), intensifying competitive pressure. Strategic alliances, licensing deals, and acquisitions are frequent as large players seek to supplement their pipelines while smaller innovators leverage partnerships to access development and commercialization resources. Regulatory approvals and patent expirations also shape this landscape, leading to biosimilar entry in some segments, which further influences competitive dynamics and pricing.
Overall, while a few dominant firms command substantial influence due to their extensive portfolios and global reach, the market remains dynamic and increasingly diversified as new entrants and technologies expand the competitive frontier.
Recent Developmental Activities in the Immunotherapy Drugs Market
- In January 2026, Bristol Myers Squibb announced that Nivolumab in combination with chemotherapy before surgery (neoadjuvant therapy), followed by nivolumab alone after surgery (adjuvant therapy), was recommended by the National Institute for Health and Care Excellence (NICE) for the treatment of adult patients with resectable non-small cell lung cancer (NSCLC). Sun Pharmaceutical Industries Limited announced that UNLOXCYT™ (cosibelimab-ipdl) had become available in the United States for healthcare professionals to prescribe to adults with mCSCC or laCSCC who were not candidates for curative surgery or curative radiation.
- In October 2025, GSK plc announced that the U.S. FDA had approved Blenrep (belantamab mafodotin-blmf) intravenous therapy in combination with bortezomib and dexamethasone (BVd) for adult patients with relapsed or refractory multiple myeloma who had received at least two prior lines of therapy, including a proteasome inhibitor and an immunomodulatory agent.
- In March 2025, Bristol Myers Squibb announced that the European Commission (EC) has approved Opdivo® (nivolumab) plus Yervoy® (ipilimumab) for the first-line treatment of adult patients with unresectable or advanced hepatocellular carcinoma (HCC).
- In August, 2024, the Food and Drug Administration granted accelerated approval to afamitresgene autoleucel (TECELRA, Adaptimmune, LLC), a melanoma-associated antigen A4 (MAGE-A4)-directed genetically modified autologous T cell immunotherapy, for adults with unresectable or metastatic synovial sarcoma who have received prior chemotherapy.
- In October 2023, Merck & Co. announced that the U.S. FDA had approved Keytruda, its anti-PD-1 therapy, for the treatment of patients with resectable non-small cell lung cancer (NSCLC) (tumors ≥4 cm or node-positive).
|
Report Metrics |
Details |
|
Study Period |
2023 to 2034 |
|
Base Year |
2025 |
|
Forecast Period |
2026 to 2034 |
|
Immunotherapy Drugs Market CAGR (2026-2034) |
XX% |
|
Key Companies in the Immunotherapy Drugs Market |
Merck & Co., Inc., Bristol-Myers Squibb Company, Novartis AG, F. Hoffmann-La Roche Ltd., Dendreon Pharmaceuticals, Amgen, Serum Institute of India (SII), Chiron Corporation, ImmunityBio, Inc., Kite Pharma Inc., AstraZeneca, Johnson & Johnson and its affiliates, Sanofi, GlaxoSmithKline, Checkpoint Therapeutics, Inc., Adaptimmune, LLC., Iovance Cell Therapy Center, AbbVie, 3M, and others. |
|
Immunotherapy Drugs Market Segments |
by Drug Type, by Route of Administration, by Indication, by End-users, and by Geography |
|
Immunotherapy Drugs Regional Scope |
North America, Europe, Asia Pacific, the Middle East, Africa, and South America |
|
Immunotherapy Drugs Country Scope |
U.S., Canada, Mexico, Germany, United Kingdom, France, Italy, Spain, China, Japan, India, Australia, South Korea, and key Countries |
Immunotherapy Drugs Market Segmentation
· Immunotherapy Drugs by Drug Type Exposure
Monoclonal Antibodies
-
-
- Immune Checkpoint Inhibitors (PD-1, PD-L1, CTLA-4)
- Targeted Tumor Antigen Antibodies
- Antibody–Drug Conjugates (ADCs)
-
Adoptive Cell Therapies
§ CAR-T Cell Therapies
§ TCR-T Cell Therapies
§ Tumor-Infiltrating Lymphocyte (TIL) Therapies}
o Cytokines & Interleukins
o Cancer Vaccines
o Others
· Immunotherapy Drugs Drug Route of Administration Exposure
o Intravenous
o Subcutaneous
o Others
· Immunotherapy Drugs Indication Exposure
o Oncology
o Autoimmune Disorders
o Viral Infection
· Immunotherapy Drugs End-Users Exposure
o Hospitals
o Oncology Clinics
o Others
· Immunotherapy Drugs Geography Exposure
o North America Immunotherapy Drugs Market
§ United States Immunotherapy Drugs Market
§ Canada Immunotherapy Drugs Market
§ Mexico Immunotherapy Drugs Market
o Europe Immunotherapy Drugs Market
§ United Kingdom Immunotherapy Drugs Market
§ Germany Immunotherapy Drugs Market
§ France Immunotherapy Drugs Market
§ Italy Immunotherapy Drugs Market
§ Spain Immunotherapy Drugs Market
§ Rest of Europe Immunotherapy Drugs Market
o Asia-Pacific Immunotherapy Drugs Market
§ China Immunotherapy Drugs Market
§ Japan Immunotherapy Drugs Market
§ India Immunotherapy Drugs Market
§ Australia Immunotherapy Drugs Market
§ South Korea Immunotherapy Drugs Market
§ Rest of Asia-Pacific Immunotherapy Drugs Market
o Rest of the World Immunotherapy Drugs Market
§ South America Immunotherapy Drugs Market
§ Middle East Immunotherapy Drugs Market
§ Africa Immunotherapy Drugs Market
Immunotherapy Drugs Market Recent Industry Trends and Milestones (2022-2026)
|
Category |
Key Developments |
|
Immunotherapy Drugs Regulatory Approvals |
Merck & Co. - Keytruda, for non-small cell lung cancer (FDA), GSK plc - Blenrep (belantamab mafodotin-blmf) (FDA), Bristol Myers Squibb - Opdivo® (nivolumab) plus Yervoy® (ipilimumab) (EU) |
|
Company Strategy |
Merck reorganized its pharmaceutical business by separating oncology (including immunotherapy assets like Keytruda) from non-cancer segments. Gilead announced plans to acquire cancer therapy developer Arcellx (a CAR-T cell therapy company) for up to $7.8 billion. This move significantly strengthens its immunotherapy and cell therapy portfolio especially in adoptive cell therapies and builds on existing collaborations in the space. |
|
Emerging Technology |
Checkpoint Inhibitor Next-Generations, Bispecific and Multispecific Antibodies, CAR-T Cell Therapy (Next-Gen CARs), TCR (T-Cell Receptor) Therapies, and others |
Impact Analysis
U.S. Tariff Impact Analysis on Immunotherapy Drugs Market:
The U.S. tariff impact on immunotherapy drugs is generally limited for finished products because most biologics and advanced therapies are either manufactured domestically or fall under trade exemptions for essential medicines. However, tariffs on active pharmaceutical ingredients (APIs), raw materials, laboratory equipment, and bioprocessing components, especially those imported from major manufacturing hubs such as China or Europe—can increase production costs for companies developing and manufacturing immunotherapy drugs.
This may lead to higher overall R&D expenses, supply chain disruptions, and potential price pressures. Additionally, retaliatory trade measures or geopolitical tensions can affect cross-border partnerships, clinical trial supplies, and contract manufacturing arrangements. Overall, while direct tariff effects on final immunotherapy drugs are relatively constrained, indirect impacts through supply chains and manufacturing inputs can influence cost structures and market dynamics in the U.S. biotech sector.
How This Analysis Helps Clients
- Cost Management: By understanding the tariff landscape, clients can anticipate cost increases and adjust pricing strategies accordingly, ensuring profitability.
- Supply Chain Optimization: Clients can identify alternative sourcing options and diversify their supply chains to reduce dependency on high-tariff regions, enhancing resilience.
- Regulatory Navigation: Expert guidance on navigating the evolving regulatory environment helps clients maintain compliance and avoid potential legal challenges.
- Strategic Planning: Insights into tariff impacts enable clients to make informed decisions about manufacturing locations, partnerships, and market entry strategies.
Startup Funding & Investment Trends
|
Company Name |
Total Funding |
Main Products |
Stage of Development |
Core Technology |
|
Exeliom Biosciences |
€2.85 M |
EXL01 |
Series A |
It is a precision innate immune modulator delivered via engineered live bacteria for cancer and immune-related diseases. |
Key Takeaways from the Immunotherapy Drugs Market Report Study
- Market size analysis for the current immunotherapy drugs market size (2025), and market forecast for 8 years (2026 to 2034)
- Top key product/technology developments, mergers, acquisitions, partnerships, and joint ventures happened over the last 3 years.
- Key companies dominating the immunotherapy drugs market.
- Various opportunities available for the other competitors in the immunotherapy drugs market space.
- What are the top-performing segments in 2025? How these segments will perform in 2034?
- Which are the top-performing regions and countries in the current immunotherapy drugs market scenario?
- Which are the regions and countries where companies should have concentrated on opportunities for the immunotherapy drugs market growth in the future?
Stay updated with us for Recent Articles @ New DelveInsight Blogs