Huntington's Disease Market Summary
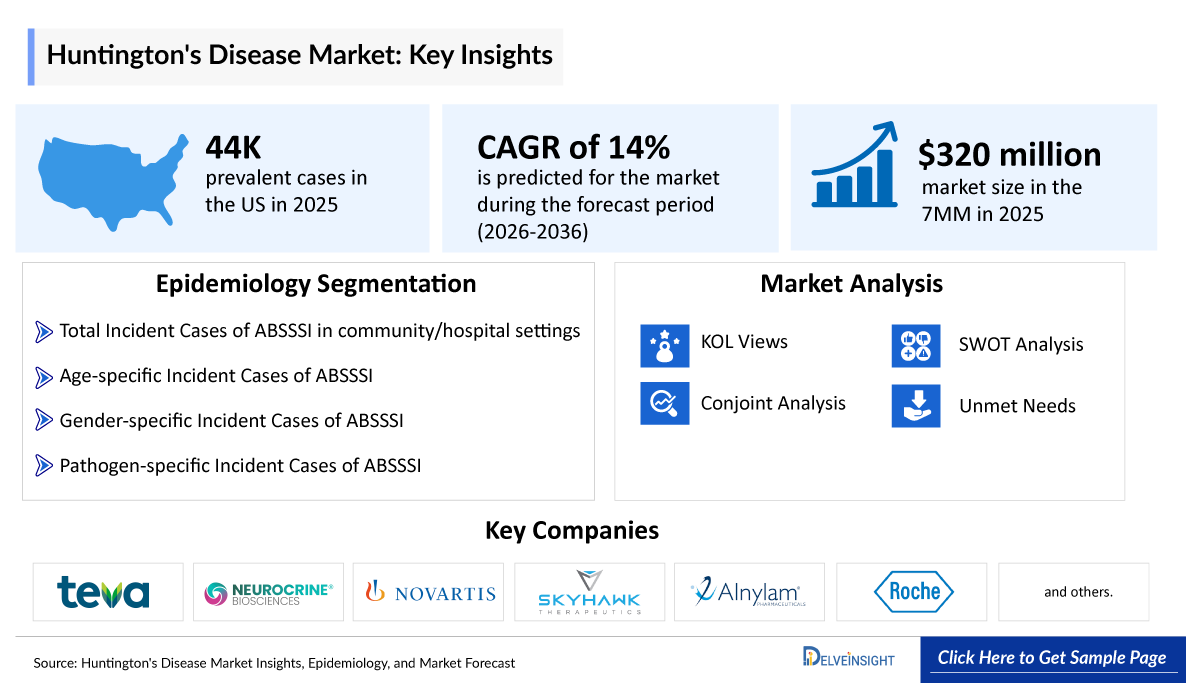
- The Huntington's Disease market size was valued at USD 320 million in 2025 and is projected to grow at a CAGR of 14% during the forecast period from 2026 to 2036.
- The leading Huntington's Disease companies develooping therapies in the treatment market include - Teva Pharmaceuticals, Neurocrine Biosciences, Novartis Pharmaceuticals, Skyhawk Therapeutics, Alnylam Pharmaceuticals, Roche, UniQure Biopharma, and others.
Huntington's Disease Market & Epidemiology Insights
- Despite advances in understanding disease pathology, treatment remains largely symptomatic, with no curative options currently available, highlighting a significant unmet medical need.
- Growing focus on genetic and biomarker-driven approaches, including mutant huntingtin (mHTT) lowering strategies, is shaping the future treatment paradigm and enabling earlier intervention.
- The therapeutic landscape is evolving with the development of gene therapies, antisense oligonucleotides, and RNA-based approaches aimed at slowing disease progression rather than only managing symptoms.
- Currently approved therapies, including AUSTEDO (Teva Pharmaceuticals), INGREZZA (Neurocrine Biosciences), and Tetrabenazine (available as generics), primarily focus on symptomatic management, especially chorea, rather than modifying disease progression.
- The treatment landscape faces key challenges, including the lack of disease-modifying therapies, variability in disease progression, limited efficacy of symptomatic treatments, and safety/tolerability concerns associated with long-term use.
- Emerging pipeline candidates such as SKY-0515, PTC518, and others highlight a shift toward disease-modifying approaches, including gene-silencing and RNA-targeted therapies.
- Increasing research investments, patient advocacy, and clinical trial activity are accelerating innovation, while improved disease awareness is expected to support market expansion over the forecast period.
Huntington’s Disease Market Size and Forecast in the 7MM
- 2025 Huntington’s Disease Market Size: USD 320 million
- 2036 Projected Huntington’s Disease Market Size: USD XXX million
- Huntington’s Disease Growth Rate (2026–2036): 14% CAGR
Download the Sample PDF to Get More Insight @ Huntington’s Disease Market Forecast
Key Factors Driving the Huntington’s Disease Market
- Advancements in Gene and RNA-Based Therapies: Increasing research into gene-silencing and RNA-targeted treatments is creating significant growth opportunities in the Huntington's Disease market.
- Rising Awareness and Improved Diagnostics: Enhanced genetic testing capabilities and growing awareness among healthcare professionals are supporting earlier diagnosis and patient management.
- Strong Pipeline of Emerging Therapies: Pharmaceutical companies are actively developing innovative therapeutics, expanding the clinical pipeline and driving market expansion.
- Growing Investment in Rare Neurological Disorders: Increased funding from biotech firms, research organizations, and government initiatives is accelerating Huntington’s disease research and treatment development.
- Supportive Regulatory Incentives: Orphan drug designations, fast-track approvals, and regulatory support for rare disease therapies are encouraging innovation and commercial growth.
DelveInsight's ‘Huntington’s Disease Market Insights, Epidemiology and Market Forecast – 2036’ report delivers an in-depth understanding of Huntington’s disease, historical and forecasted epidemiology, as well as the Huntington’s Disease market trends in the United States, EU4 (Germany, Spain, Italy, and France) and the United Kingdom, and Japan.
The Huntington’s Disease market report delivers a comprehensive analysis of the current treatment landscape, including standards of care, clinical practices, and evolving therapeutic algorithms. It evaluates Huntington’s disease patient burden trends, revenue & market share dynamics, peak patient share & therapy uptake analysis, and provides an in-depth market size assessment, and growth rate projections (Historical & Forecast 2022–2036) across global regions. The report highlights key unmet medical needs in Huntington’s disease and maps the competitive and clinical landscape to uncover high‑value opportunities, providing a clear outlook on future market growth potential.
Scope of the Huntington's Disease Market Report | |
|
Study Period |
2022–2036 |
|
Historical Year |
2022–2025 |
|
Forecast Period |
2026–2036 |
|
Base Year |
2026 |
|
Geographies Covered |
|
|
Huntington’s Disease Market CAGR (Study period/Forecast period) |
14% (2026–2036) |
|
Huntington’s Disease Epidemiology Segmentation Analysis |
Patient Burden Assessment
|
|
Huntington’s Disease Companies |
|
|
Huntington’s Disease Therapies |
|
|
Huntington’s Disease Market |
Segmented by
|
|
Analysis |
|
Key Factors Driving the Huntington’s Disease Market
- Advancements in genetic testing and diagnosis: Widespread adoption of predictive genetic testing enables early identification of at-risk individuals, expanding the diagnosed patient pool and facilitating earlier intervention.
- Emergence of disease-modifying therapies: Ongoing development of gene-silencing approaches (e.g., antisense oligonucleotides) and targeted therapies is shifting the market from symptomatic management to potential disease-modifying treatments.
- Increasing research funding and awareness: Growing support from governments and patient advocacy organizations is accelerating clinical research and improving disease awareness, driving demand for innovative therapies.
- Improved understanding of disease biology: Deeper insights into the molecular and genetic basis of Huntington’s disease are enabling the development of more precise and targeted therapeutic strategies.
Huntington’s Disease Understanding
Huntington’s Disease Overview and Diagnosis
Huntington’s disease is a rare, inherited, progressive brain disorder caused by a mutation in the HTT gene on chromosome 4, leading to the production of abnormal huntingtin protein and gradual nerve cell damage. It follows an autosomal dominant pattern, meaning individuals who inherit the faulty gene will develop the disease. Symptoms typically appear in adulthood and include involuntary movements (chorea), cognitive decline, and psychiatric issues such as depression and irritability. Diagnosis is based on clinical evaluation, family history, genetic (blood) testing, and imaging techniques such as CT or MRI scans. While there is no cure, treatment focuses on managing symptoms and providing supportive care as the disease progresses.
Further details are provided in the report....
Current Huntington’s Disease Treatment Landscape
There is currently no cure or disease-modifying therapy for Huntington’s disease; treatment focuses on managing symptoms and improving quality of life. Medications are used to control movement disorders and psychiatric symptoms. Deutetrabenazine and Tetrabenazine are approved for treating chorea, while antidepressants, antipsychotics, and mood stabilizers help manage depression, irritability, and behavioral changes. Supportive care, including physical therapy, occupational therapy, and speech therapy, plays a key role in maintaining function and independence. As the disease progresses, patients often require increasing assistance, supervision, and comprehensive caregiving support.
Further details related to country-based variations are provided in the report...
Huntington’s Disease Unmet Needs
The section “unmet needs of Huntington’s Disease” outlines the critical gaps between the current state of patient care, diagnosis, and the ideal & effective management of the disease. It highlights the obstacles experienced by patients, clinicians, and researchers and identifies potential solutions for future progress.
-
Lack of Disease-modifying Therapies
-
Inadequate Treatment for Cognitive Decline
-
Psychiatric Burden Management Gaps
-
Delayed Diagnosis and Predictive Challenges and others…..
Note: Comprehensive unmet needs insights in Huntington’s disease and their strategic implications are provided in the full report...
Huntington’s Disease Epidemiology
The Huntington’s Disease epidemiology section provides insights about the historical and current Huntington’s Disease patient pool and forecasted trends for individual seven major countries. It helps to recognize the causes of current and forecasted trends by exploring numerous studies and views of key opinion leaders. This part of the Huntington’s Disease market report also provides the diagnosed patient pool and their trends along with assumptions undertaken.
Key Findings from Huntington’s Disease Epidemiological Analysis and Forecast
- In the United States, the total number of prevalent cases of Huntington’s disease was 44,000 in 2025, which is expected to increase during the forecast period, i.e., 2026–2036.
- In 2025, the total prevalent cases of Huntington’s disease were 37,000 in EU4 and the UK, which are projected to increase during the forecast period, i.e., 2026–2036.
- Findings from various secondary studies suggest that Huntington’s disease is more prevalent in the elderly population.
- In addition, secondary data suggest that fewer people are identified in stage IV and stage V with Huntington's disease. In 2025, 80 Stage IV cases of Huntington’s disease and 20 Stage V cases of Huntington’s disease were estimated for Japan.
- In the 7MM, Huntington’s Disease is more prevalent in individuals aged ≥60 years, accounting for approximately 50% of total cases.
Huntington’s Disease Epidemiology Segmentation
- Total Prevalent Cases of Huntington’s Disease
- Total Diagnosed Prevalent Cases of Huntington’s Disease
- Age-specific Cases of Huntington’s Disease
- Stage-specific Cases of Huntington’s Disease
- Total Treated Cases of Huntington’s Disease
Huntington’s Disease Drug Analysis & Competitive Landscape
The Huntington’s disease drug chapter provides a detailed, market-focused review of approved therapies and the emerging pipeline across Phase I–III Huntington’s Disease clinical trials. It covers the mechanism of action, clinical trial data, regulatory approvals, patents, collaborations, and strategic partnerships for each therapy, along with their advantages, limitations, and recent developments. This section offers critical insights into the Huntington’s disease treatment landscape, supporting market assessment, competitive analysis, and growth forecasting for the Huntington’s disease market.
Approved Therapies for Huntington’s Disease
Deutetrabenazine (AUSTEDO): Teva Pharmaceuticals
AUSTEDO is a deuterated form of a small molecule inhibitor of vesicular monoamine 2 transporter, or VMAT2, that is designed to regulate the levels of a specific neurotransmitter, dopamine, in the brain. It is indicated in adults for the treatment of Chorea associated with Huntington’s disease. The FDA granted AUSTEDO New Chemical Entity Exclusivity until April 2022 and Orphan Drug exclusivity for the treatment of chorea associated with Huntington disease until April 2024.
- In May 2024, Teva Pharmaceuticals announced that the US Food and Drug Administration (FDA) had approved AUSTEDO XR as a one-pill, once-daily treatment option, now with four new tablet strengths (30, 36, 42, 48 mg) indicated in adults for tardive dyskinesia and Huntington disease chorea.
INGREZZA (valbenazine/NBI-98854): Neurocrine Biosciences
INGREZZA is a selective VMAT2 inhibitor, believed to work by reducing the amount of dopamine released in a region of the brain that controls movement and motor function. In August 2023, the FDA granted approval for INGREZZA capsules to treat chorea in adults with Huntington's disease. The FDA approval is supported by data from two clinical studies conducted in collaboration with the Huntington Study Group (HSG), including the KINECT-HD Phase III study and the ongoing KINECT-HD2 open-label extension trial.
Note: Detailed marketed therapies assessment will be provided in the final report....
Huntington’s Disease Pipeline Analysis
SKY-0515: Skyhawk Therapeutics
SKY-0515 is an orally-administered, investigational small molecule RNA modulator developed through the company's novel RNA-modulating platform, SKYSTAR. SKY-0515 therapeutically reduces both HTT protein and PMS1 protein. PMS1 is an additional key driver of somatic CAG repeat expansion and HD pathology and should complement the benefits of reducing mutant HTT. Skyhawk also announced that its SKY-0515 Phase II/III FALCON-HD trial, open at twelve sites in Australia and New Zealand, has expanded worldwide. Skyhawk has now treated more than 90 patients with SKY-0515.
-
In January 2026, Skyhawk Therapeutics reported positive results from a nine-month interim analysis of its Phase I trial evaluating SKY-0515 for Huntington’s Disease.
Votoplam (PTC518): Novartis Pharmaceuticals
PTC518 is a small molecule splicing modifier that acts via a unique mechanism to promote the inclusion of a novel pseudoexon containing a premature termination codon, thus triggering Huntingtin (HTT) mRNA degradation and subsequent reduction in HTT protein levels.
- In March 2025, PTC Therapeutics announced results from the Phase II PIVOT-HD study of PTC518 (votoplam) in Stage 2 and Stage 3 Huntington's disease patients. The study met its primary endpoint of reduction in blood Huntingtin (HTT) protein levels (p<0.0001) at Week 12 and favorable safety and tolerability. In addition, the 12-month data from the Stage 2 patients are consistent with the previously reported dose-dependent lowering of HTT protein and dose-dependent trends across clinical scales.
- In December 2024, Novartis announced that it had entered into a global license and collaboration agreement with PTC Therapeutics for PTC518, an HTT mRNA splice modulator with the potential to become the first oral disease-modifying therapy for Huntington's disease.
Competitive Landscape of Huntington’s Disease Pipeline Drugs | ||||||
|
Drug Name |
Company |
Highest Phase |
Indication |
RoA |
MoA |
Anticipated Launch in the US |
|
SKY-0515 |
Skyhawk Therapeutics |
III |
Huntington’s disease |
Oral |
RNA modulator |
Information is available in the full report |
|
Votoplam (PTC518) |
Novartis Pharmaceuticals |
III |
Huntington’s disease |
Oral |
RNA splicing modulators |
Information is available in the full report |
|
Note: Launch insights are provisional and may change with future report updates or the occurrence of major key catalysts. | ||||||
Note: Detailed emerging therapies assessment will be provided in the final report...
Huntington’s Disease Key Players, Market Leaders, and Emerging Companies
- Teva Pharmaceuticals
- Neurocrine Biosciences
- Novartis Pharmaceuticals
- Skyhawk Therapeutics
- Alnylam Pharmaceuticals
- Roche
- UniQure Biopharma, and others.
Huntington’s Disease Drug Updates
-
In September 2025, Skyhawk Therapeutics announced positive results from the first interim analysis of the Part C patient cohort in its Phase I clinical trial of SKY-0515, an investigational treatment for Huntington's disease. At Day 84, patients receiving SKY-0515 demonstrate dose-dependent reductions of mHTT protein in blood, including 62% at the 9mg dose. SKY-0515 has been generally well tolerated at both dose levels tested.
-
In March 2025, PTC Therapeutics announced results from the Phase II PIVOT-HD study of PTC518 (votoplam) in Stage 2 and Stage 3 Huntington's disease patients. The study met its primary endpoint of reduction in blood Huntingtin (HTT) protein levels (p<0.0001) at Week 12 and favorable safety and tolerability. In addition, the 12-month data from the Stage 2 patients are consistent with the previously reported dose-dependent lowering of HTT protein and dose-dependent trends across clinical scales.
Huntington’s Disease Market Outlook
The treatment landscape for Huntington’s disease remains highly limited and largely symptomatic, with no approved disease-modifying therapies currently available. Existing pharmacological options primarily focus on managing motor symptoms such as chorea and addressing psychiatric manifestations, with agents like Deutetrabenazine and Tetrabenazine forming the cornerstone of therapy. As a result, the market size remains relatively small compared to other neurodegenerative disorders, driven by the disease’s rare prevalence and limited therapeutic options.
However, the Huntington’s disease pipeline is evolving, with increasing focus on disease-modifying approaches such as Gene silencing therapies, Antisense oligonucleotides, RNA-targeting strategies, and cell-based therapies aimed at reducing mutant huntingtin protein expression. These emerging modalities have the potential to transform the treatment paradigm if proven effective, shifting the market from symptomatic management to targeted, mechanism-based interventions.
Despite this progress, several challenges continue to restrain market growth, including high clinical trial failure rates, complexities in targeting the central nervous system, limited patient populations for large-scale studies, and regulatory uncertainties. Additionally, the lack of validated biomarkers and standardized endpoints complicates drug development and approval pathways.
Looking ahead, the market is expected to witness gradual growth driven by advancements in precision medicine, increasing research investments, and improved understanding of disease biology. The successful launch of disease-modifying therapies, along with enhanced diagnostic capabilities and patient identification, is anticipated to significantly expand the market potential and improve long-term outcomes for patients with Huntington’s disease.
- In 2025, the total market size of Huntington’s disease by therapies was USD 320 million in the 7MM. The highest revenue was captured by AUSTEDO, accounting for USD 167 million in 2025.
- During the forecast period (2026–2036), pipeline candidates such as SKY-0515, PTC518, and others are expected to drive the growth of the Huntington’s disease market size.
Further details will be provided in the report….
Drug Class/Insights into Leading Emerging and Marketed Therapies in Huntington’s Disease (2022–2036 Forecast)
The treatment landscape of Huntington’s disease remains limited and is primarily focused on symptomatic management; however, emerging therapeutic approaches are targeting the underlying genetic cause and disease progression.
- Gene-targeting therapies: Advanced approaches such as antisense oligonucleotides (ASOs) and RNA interference (RNAi) therapies aim to reduce the production of mutant huntingtin protein, which is the root cause of neuronal damage in Huntington’s disease. These therapies work by selectively silencing or modifying the expression of the faulty gene and are currently being evaluated in clinical trials as potential disease-modifying options.
- Small molecules: Existing pharmacological treatments mainly address symptoms rather than disease progression. Agents such as Deutetrabenazine and Tetrabenazine are used to manage chorea, while other small molecules, including antidepressants, antipsychotics, and mood stabilizers, are used to control psychiatric and behavioral symptoms.
Huntington’s Disease Drug Uptake
This section focuses on the uptake rate of potential Huntington’s disease drugs expected to be launched in the market during the forecast period (2026–2036). The analysis covers the Huntington’s disease drug’s uptake, performance at peak, factors affecting performance during prime years of growth, patient uptake by therapy, and anticipated sales generated by each drug.
Among the therapies, INGREZZA and ANX005 (tanruprubart) are expected to capture the largest market share.
Detailed insights into emerging therapies' drug uptake are included in the report...
Huntington’s Disease Market Access and Reimbursement of Approved Therapies
The report further provides detailed insights on the country-wise accessibility and reimbursement scenarios, cost-effectiveness scenario of approved therapies, programs making accessibility easier and out-of-pocket costs more affordable, insights on patients insured under federal or state government prescription drug programs, etc.
The United States
Teva TOTAL SUPPORT
More than 90% of people pay USD 10 or less per month for AUSTEDO XR. Cost shouldn't stand in the way of starting treatment. Teva Total Support is designed to assist you every step of the way.
Reimbursement is a crucial factor that affects the drug’s access to the market. Often, the decision to reimburse comes down to the price of the drug relative to the benefit it produces in treated patients. To reduce the healthcare burden of these high-cost therapies, many payment models are being considered by payers and other industry insiders.
NOTE: Further Details are provided in the final report….
Huntington’s Disease Therapies Price Scenario & Trends
Pricing and analogue assessment of Huntington’s disease therapies highlights evolving price dynamics structures. This section summarizes the cost of approved treatments, the closest and most appropriate analogue selection for emerging therapies, and the understanding of how pricing influences market access, adherence, and long-term uptake.
Pricing of Huntington’s Disease Approved Drugs
AUSTEDO is administered orally titrate at weekly intervals by 6 mg per day based on reduction of chorea and tolerability, up to a maximum recommended daily dosage of 48 mg. The estimated annual treatment cost is approximately USD 60,000.
Industry Experts and Physician Views for Huntington’s Disease
To keep up with Huntington’s Disease market trends, we take Key Opinion Leaders (KOLs) and Subject Matter Experts (SMEs) opinions working in the domain through primary research to fill the data gaps and validate our secondary research. Industry Experts were contacted for insights on the Huntington’s disease emerging therapies, evolving treatment landscape, patient adherence to conventional therapies, therapy switching trends, drug adoption and uptake, accessibility challenges, and epidemiology and real-world prescription patterns in Huntington’s Disease, including MD, PhD, Instructor, Postdoctoral Researcher, Professor, Researcher, and others.
DelveInsight’s analysts connected with 10+ KOLs to gather insights at the country level. Centers such as the Huntington’s Disease Research Foundation (MMRF), University of Texas MD Anderson Cancer Center, and Iwate Medical University, etc. were contacted. Their opinion helps understand and validate current and emerging Huntington’s disease therapies, highlight unmet medical needs, provide epidemiological context, and support strategic decisions for market access, therapy adoption, and pipeline prioritization in Huntington’s disease.
|
Region |
Key Opinion Leaders (KOLs) and Subject Matter Experts (SMEs) |
|
United States |
“Children of a parent with Huntington’s disease have a 50% probability of inheriting the mutated HTT gene and, consequently, developing the disease later in life. With the advent of PCR-based genetic testing techniques, at-risk individuals can now determine their genetic status, although the decision to undergo such testing remains a personal choice. With the growing number of research foundations focused on Huntington’s disease and the expanding understanding of its molecular basis, continued collaborative efforts are expected to drive progress toward identifying effective treatments and, ultimately, a cure.” |
|
Spain |
“Although Huntington’s disease is rare, it does receive a great deal of research attention. One reason is that Huntington’s disease has some features that make it more likely to be a tractable problem than other neurodegenerative conditions. First, the autosomal dominant nature of the condition means that the diagnosis is almost definitive and can be made before death. Thus, it is possible to accurately model and study the disease in vitro and in vivo.” |
Huntington’s Disease Report Qualitative Analysis: SWOT and Conjoint Analysis
We perform qualitative and market Intelligence analysis using various approaches, such as SWOT analysis and conjoint analysis.
In the SWOT analysis of Huntington’s Disease, strengths, weaknesses, opportunities, and threats in terms of disease diagnosis, patient awareness, patient burden, competitive landscape, cost-effectiveness, and geographical accessibility of therapies are provided.
Conjoint analysis analyzes emerging therapies based on relevant attributes such as safety, efficacy, frequency of administration, route of administration, and order of entry. Scoring is given based on these parameters to analyze the effectiveness of therapy. The team of analysts analyzes promising emerging therapies based on relevant attributes such as safety, efficacy, frequency of administration, route of administration, and order of entry. In efficacy, the trial’s primary and secondary outcome measures are evaluated, whereas the therapies’ safety is evaluated, wherein the acceptability, tolerability, and adverse events are mainly observed.
In addition, the scoring is also based on the route of administration, order of entry, probability of success, and the addressable patient pool for each therapy. According to these parameters, the final weightage score and the ranking of the emerging therapies are decided.
Scope of the Huntington’s Disease Market Report
- The report covers a segment of key events, an executive summary, a descriptive overview of Huntington’s disease, explaining their causes, signs and symptoms, pathogenesis, and currently available treatments.
- Comprehensive insight has been provided into the epidemiology segments and forecasts, the future growth potential of the diagnosis rate, and disease progression along treatment guidelines.
- Additionally, an all-inclusive account of both the current and emerging treatments, along with the elaborate profiles of late-stage and prominent therapies, will have an impact on the current treatment landscape.
- A detailed review of the Huntington’s disease market, historical and forecasted market size, market share by therapies, detailed assumptions, and rationale behind our approach is included in the report, covering the 7MM drug outreach.
- The report provides an edge while developing business strategies by understanding trends through SWOT analysis and expert insights/KOL views, patient journey, and treatment preferences that help in shaping and driving the 7MM Huntington’s disease market.
Huntington’s Disease Market Report Insights
- Huntington’s Disease Patient Population Forecast
- Huntington’s Disease Market Opportunity (Current and Forecasted)
- Huntington’s Disease Pipeline Analysis
- Huntington’s Disease Market Size and Trends
- Huntington’s Disease Market Opportunity (Current and Forecasted)
Huntington’s Disease Market Report Key Strengths
- Epidemiology‑based (Epi‑based) bottom‑up Forecasting
- Artificial Intelligence (AI)-enabled Market Research Report
- 11-year Forecast
- Huntington’s Disease Market Outlook (North America, Europe, Asia-Pacific)
- Patient Burden Trends (by geography)
- Huntington’s Disease Treatment Addressable Market (TAM)
- Huntington’s Disease Competitive Landscape
- Huntington’s Disease Major Companies Insights
- Huntington’s Disease Price Trends and Analogue Assessment
- Huntington’s Disease Therapies and Drug Adoption/Uptake
- Huntington’s Disease Therapies Peak Patient Share Analysis
Huntington’s Disease Market Report Assessment
- Huntington’s Disease Current Treatment Practices
- Huntington’s Disease Unmet Needs
- Huntington’s Disease Clinical Development Analysis
- Huntington’s Disease Emerging Drugs Product Profiles
- Huntington’s Disease Market Attractiveness
- Huntington’s Disease Qualitative Analysis (SWOT and Conjoint Analysis)
- Huntington’s Disease Market Drivers
- Huntington’s Disease Market Barriers
FAQs Related to the Huntington’s Disease Market Report:
Huntington’s Disease Market Insights
- What was the Huntington’s disease market size, the market size by therapies, market share (%) distribution in 2025, and what would it look like by 2036? What are the contributing factors for this growth?
- What are the anticipated pricing variations among different geographies for the emerging therapies in the future?
- What can be the future treatment paradigm of Huntington’s disease?
- What are the disease risks, burdens, and unmet needs of Huntington’s disease? What will be the growth opportunities across the 7MM concerning the Huntington’s disease patient population?
- Who is the major future competitor in the market, and how will the competitors affect their market share?
- What are the current options for the treatment of Huntington’s disease? What are the current guidelines for treating Huntington’s disease in the US, Europe, and Japan?
Reasons to Buy Huntington’s Disease Market Forecast Report
- The report will help in developing business strategies by understanding the latest trends and changing treatment dynamics driving the Huntington’s disease market.
- Bottom-up forecasting builds from the affected population to product forecasts, delivering a robust, data-driven approach ideal for new therapies and novel classes.
- Insights on patient burden/disease incidence, evolution in diagnosis, and factors contributing to the change in the epidemiology of the disease during the forecast years.
- Understand the existing market opportunities in varying geographies and the growth potential over the coming years.
- Identifying strong upcoming players in the market will help devise strategies to help get ahead of competitors.
- Detailed analysis and ranking of class-wise potential current and emerging therapies under the conjoint analysis section to provide visibility around leading classes.
- To understand KOLs’ perspectives on the accessibility, acceptability, and compliance-related challenges of existing treatment to overcome barriers in the future.
- Detailed insights into the unmet needs of the existing market so that the upcoming players can strengthen their development and launch strategy.
- This Artificial Intelligence (AI)-enabled report summarize and simplify complex datasets within the report into clear, actionable insights for stakeholders, investors, and healthcare providers, enabling faster, data-driven decisions.