Malignant Pleural Effusion Market Summary
Malignant Pleural Effusion Market Insights and Trends
- According to DelveInsight analysis, the Malignant Pleural Effusion Market Size was found to be more than USD 2 billion in the leading markets (the United States, the EU4 (Germany, France, Italy, and Spain), the United Kingdom, and Japan) in 2025.
- Among the 7MM, the largest Malignant Pleural Effusion Market Share of more than USD 1 billion in the MPE market was captured by the US in 2025 and it is projected to continue with a positive growth trajectory.
- In 2025, the US had the highest Malignant Pleural Effusion Incident Cases among the 7MM which was approximately 180,000 cases. The rise in Malignant Pleural Effusion Cases in the US is attributed to increased cancer incidence and survival rates and improved healthcare access, resulting in increased and heightened awareness of the condition.
- The current Malignant Pleural Effusion Market predominantly depends on drainage procedures due to the absence of approved therapies and the severe nature of the disease, which necessitates often painful interventions. A range of palliative treatment options is available, including systemic cancer therapy, thoracentesis, tube thoracostomy, pleurodesis with sclerosants, pleural catheter placement, pleuroperitoneal shunts, and pleurectomy. They are all aimed at managing symptoms and improving patient comfort.
- It is recommended that talc pleurodesis, particularly via thoracoscopic poudrage with large-bore drains, be used as the preferred treatment for MPE due to its high success rates and effectiveness in symptom relief. Indwelling pleural catheters (IPC) can be considered as an alternative, as they provide similar symptom relief and shorter hospital stays, though they are associated with higher adverse event rates. Nonsteroidal analgesics should be used for pain management, as they do not impact pleurodesis success.
- Malignant Pleural Effusion Emerging Therapies are investigating diverse mechanisms to address multiple facets of the disease. Among them, PTS500, a small-molecule candidate from Gongwing Biopharma, is currently being evaluated in Phase II clinical trials.
- The lack of epidemiological studies on Malignant Pleural Effusion limits understanding of its incidence and risk factors, hindering early diagnosis and evidence-based treatment strategies. This gap results in inconsistent management, reduced clinical trial development, inadequate resource allocation, and poor healthcare planning, ultimately affecting patient outcomes and limiting advancements in Malignant Pleural Effusion therapy.
Malignant Pleural Effusion Market size and forecast
- 2025 Malignant Pleural Effusion Market Size in the 7MM: ~USD 2 Billion
- Malignant Pleural Effusion Growth Rate (2022–2036): 4.9% CAGR
Request for Unlocking the Sample Page of the "Malignant Pleural Effusion Treatment Market"
Factors Contributing to the Growth of the Malignant Pleural Effusion Market by DelveInsight
-
Rising Prevalence of Cancer-Associated Conditions
The increasing incidence of cancers such as lung cancer, breast cancer, and lymphoma is a major driver, as malignant pleural effusion (MPE) commonly occurs as a complication of advanced malignancies.
-
Growing Geriatric Population
The aging population is more susceptible to cancer and related complications like MPE, thereby increasing the overall patient pool.
-
Advancements in Diagnostic Techniques
Improved imaging technologies and minimally invasive diagnostic procedures are enabling early detection and better management of malignant pleural effusion.
-
Increasing Demand for Minimally Invasive Treatments
Rising preference for procedures such as thoracentesis, indwelling pleural catheters, and pleurodesis is enhancing patient comfort and driving treatment adoption.
-
Development of Novel Therapeutic Approaches
Ongoing research into targeted therapies, immunotherapies, and innovative treatment modalities is expected to expand the treatment landscape.
-
Improved Supportive Care and Palliative Treatments
Growing emphasis on palliative care to improve quality of life in advanced cancer patients is contributing to the demand for effective MPE management solutions.
-
Favorable Reimbursement and Healthcare Policies
Supportive reimbursement frameworks and healthcare initiatives in developed regions are encouraging treatment uptake.
-
Increasing Healthcare Expenditure
Rising global healthcare spending is facilitating better access to advanced diagnostics and treatment options.
-
Growing Awareness and Clinical Research Activities
Enhanced awareness among clinicians and increased clinical trials are supporting innovation and improving treatment outcomes in the malignant pleural effusion market.
DelveInsight's ‘Malignant Pleural Effusion Market Insights, Epidemiology and Market Forecast – 2036’ report delivers an in-depth understanding of MPE, historical and forecasted epidemiology, as well as the MPE market trends in the United States, EU4 (Germany, Spain, Italy, and France), the United Kingdom, and Japan.
The Malignant Pleural Effusion Treatment Market Report delivers a comprehensive analysis of the current treatment landscape, including standards of care, clinical practices, and evolving therapeutic algorithms. It evaluates Malignant Pleural Effusion patient burden trends, revenue & market share dynamics, peak patient share & therapy uptake analysis, and provides an in-depth market size assessment, and growth rate projections (Historical & Forecast 2022–2036) across the 7MM regions. The Malignant Pleural Effusion Market Report highlights key unmet medical needs in Malignant Pleural Effusion and maps the competitive and clinical landscape to uncover high‑value opportunities, providing a clear outlook on future market growth potential.
Scope of the Malignant Pleural Effusion Market Report | |
|
Study Period |
2022–2036 |
|
Historical Year |
2022–2025 |
|
Forecast Period |
2026–2036 |
|
Base Year |
2025 |
|
Geographies Covered |
|
|
Malignant Pleural Effusion Market CAGR |
4.9% (2026-2036) |
|
Malignant Pleural Effusion Epidemiology Segmentation Analysis |
Patient Burden Assessment
|
|
Malignant Pleural Effusion Companies |
|
|
Malignant Pleural Effusion Therapies |
|
|
Malignant Pleural Effusion Market |
Segmented by
|
|
Analysis |
|
Malignant Pleural Effusion Understanding
Malignant Pleural Effusion Overview and Diagnosis
MPE is an effusion characterized by the presence of malignant cells. The fluid and cancer cells build up between the chest wall and the lung. It is a common but serious condition related to poor quality of life, morbidity, and mortality. MPE is a common manifestation in patients with metastatic disease. It is most common in lung cancer, followed by Breast Cancer, lymphoma, gynecological cancers, and malignant mesothelioma.
Chest ultrasonography, Computed Tomography, Positron emission tomography-computed tomography (PET-CT), and pleural biopsy are key tools for diagnosing MPE. Ultrasound is effective in assessing pleural effusion characteristics and guiding procedures, while CT can identify pleural abnormalities, although it has lower sensitivity. PET-CT offers high specificity for staging pleural disease, and cytology, while commonly used, has limited sensitivity. Pleural biopsy, particularly when guided by imaging, is the gold standard with high sensitivity and specificity, and medical thoracoscopy provides both diagnostic and therapeutic benefits.
Current Malignant Pleural Effusion Treatment Landscape
Currently, Malignant Pleural Effusion Treatment is primarily driven by drainage procedures such as thoracentesis, pleurodesis, and indwelling pleural catheters, which are used for symptom management. The Malignant Pleural Effusion Market faces substantial challenges due to the lack of curative treatments and dependence on symptomatic procedures like thoracentesis, pleurodesis, and indwelling pleural catheters. While these methods offer short-term relief, high recurrence rates and complications underscore the need for pharmaceutical innovations.
Malignant Pleural Effusion Market growth is anticipated to rely on epidemiological studies identifying patient populations most likely to benefit from new therapies. Understanding incidence and survival rates is key for drug development. While emerging treatments may address these gaps, their success will depend on robust clinical results and timely regulatory approvals. To proclaim Malignant Pleural Effusion Treatment Market rule in the US, some major pharma players are proactive with their emerging candidates, including PTS500 by Gongwing Biopharma, RSO-021 by RS Oncology, M701 by Wuhan YZY Biopharma, and others are expected to revolutionize the Malignant Pleural Effusion Treatment in the forecast period.
Malignant Pleural Effusion Unmet Needs
The section “Malignant Pleural Effusion Unmet Needs” outlines the critical gaps between the current state of patient care, diagnosis, and the ideal & effective management of the disease. It highlights the obstacles experienced by patients, clinicians, and researchers and identifies potential solutions for future progress.
- Improved early detection and diagnosis
- Lack of epidemiological studies
- Prognostic and therapeutic markers
- Lack of targeted therapies and others…..
Note: Comprehensive unmet needs insights in MPE and their strategic implications are provided in the full report.
Malignant Pleural Effusion Epidemiology
Key Findings from MPE Epidemiological Analysis and Forecast
- The total number of Malignant Pleural Effusion Incident Cases in the 7MM is approximately 480,000 in 2025.
- According to DelveInsight’s estimates, in the US, females accounted for approximately 100,000 incident cases and males for nearly 70,000 incident cases of Malignant Pleural Effusion in 2025. Malignant Pleural Effusion Incident cases in the US are expected to rise in lung cancer patients due to genetic predispositions and environmental factors, which make individuals with lung cancer more susceptible to developing MPE.
- In 2025, there were approximately 50,000 incident cases of Malignant Pleural Effusion due to breast cancer and more than 25,000 cases due to lymphoma across EU4 and the UK. According to DelveInsight’s analysis, the overall incident in both types is projected to rise during the forecast period.
- Japan recorded nearly 50,000 symptomatic cases of Malignant Pleural Effusion in 2025 and around 12,000 cases were asymptomatic. Projections indicate a rise in symptom-specific incidence in Malignant Pleural Effusion.
Malignant Pleural Effusion Epidemiology Segmentation in the 7MM
- Malignant Pleural Effusion Incident Cases
- Malignant Pleural Effusion Tumor Type-specific Incident Cases
- Malignant Pleural Effusion Gender-specific Incident Cases
- Malignant Pleural Effusion Symptom-specific Incident Cases
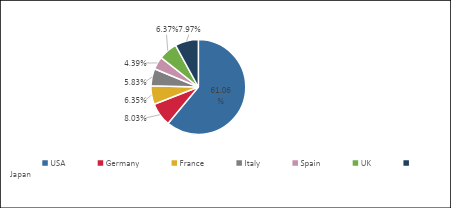
Market Share (%) Distribution of MPE by Countries in the 7MM in 2025
Malignant Pleural Effusion Analysis & Competitive Landscape
The Malignant Pleural Effusion drug chapter provides a detailed, market-focused review of the emerging pipeline across Phase I-III Malignant Pleural Effusion Clinical Trials. It covers Malignant Pleural Effusion Mechanism of Action, clinical trial data, regulatory approvals, patents, collaborations, strategic partnerships upcoming Key catalyst for each therapy, along with their advantages, limitations, and recent developments. This section offers critical insights into the Malignant Pleural Effusion Treatment landscape, supporting market assessment, competitive analysis, and growth forecasting for the Malignant Pleural Effusion Therapeutics Market.
Malignant Pleural Effusion Pipeline Analysis
-
M701: Wuhan YZY Biopharma
M701 is a new category I biological drug independently developed by YZY Biopharma, which is intended to be developed for the treatment of MPE and malignant ascites caused by tumors. M701 can target both EpCAM (as the target on tumor cells) and CD3 (as the immune T cell activation target), which bridges both tumor cells and immune T cells by binding these targets, thereby activating T cells to kill the tumor cells. Accordingly, intraperitoneal/intrapleural infusion of M701 can activate immune cells to selectively eliminate and suppress tumor cells in the abdominal/chest cavity. The drug is currently in Phase II trials for MPE caused by NSCLC.
-
- In February 2026, Wuhan YZY Biopharma received FDA approval for its IND application for M701, a bispecific antibody targeting MPE in patients with advanced lung and breast cancers.
Competitive Landscape of Pipeline Drugs | ||||||
|
Drug Name |
Company |
Highest Phase |
Indication |
RoA |
MoA |
Anticipated Launch in the US |
|
PTS500 |
Gongwing Biopharm |
II |
MPE |
Intratumoral injection |
Cell death stimulant |
2030 |
|
M701 |
Wuhan YZY Biopharma |
II |
MPE caused by NSCLC |
Intrapleural infusion |
T cell activator |
Information is available in the full report |
|
RSO-021 |
RS Oncology |
I/II |
MPE |
Pleural infusion |
Peroxiredoxin III inhibitor |
Information is available in the full report |
|
Note: Launch insights are provisional and may change with future report updates or the occurrence of major key catalysts. | ||||||
Note: Detailed emerging therapies assessment will be provided in the final report.
Malignant Pleural Effusion Companies, Market Leaders and Emerging Companies
- Gongwing Biopharm
- Wuhan YZY Biopharma
- RS Oncology
- Genelux Corporation
Malignant Pleural Effusion Drug Updates
- In March 2024, RS Oncology reported two abstracts with comprehensive preclinical and translational data in support of RSO-021, accepted for poster presentations at the Annual Meeting of the American Association for Cancer Research.
- In June 2024, RS Oncology reported positive data from the Phase I trial of RSO-021.
Malignant Pleural Effusion Market Outlook
The current Malignant Pleural Effusion Therapeutics Market lacks FDA approved curative treatments, relying instead on palliative interventions such as thoracentesis, pleurodesis, and indwelling pleural catheters. While these approaches help manage symptoms by reducing fluid buildup and alleviating dyspnea, their effectiveness is temporary, with frequent recurrence. Additionally, some procedures can be invasive or cause complications such as infection and pain, further challenging long-term disease management and emphasizing the need for more advanced therapeutic options.
- Among the 7MM, EU4 and the UK accounted for the second largest Malignant Pleural Effusion Market Size i.e., approximately USD 750 million in 2025 which is expected to rise with the anticipated launch of the emerging therapies.
- EU4 and the UK Malignant Pleural Effusion Market also lacks EMA-approved treatments, relying on palliative interventions such as thoracentesis, pleurodesis, and indwelling pleural catheters. While these treatments provide temporary relief, they are associated with high recurrence rates, complications, and limited long-term effectiveness, highlighting the need for more effective, approved therapies in these regions.
- In Japan, the Malignant Pleural Effusion Market Size was nearly USD 200 million in 2025. The Malignant Pleural Effusion Market in Japan is influenced not only by pharmacological treatments but also by drainage procedures, which play a significant role in disease management.
Drug Class/Insights into Leading Emerging Therapies in MPE (2022–2036 Forecast)
-
Anti-angiogenic agents
In recent years, intracavitary biotherapy has become a prominent area of research in the treatment of MPE. Studies have shown that recombinant human vascular endothelial growth factor inhibitors, when combined with intracavitary chemotherapy, can control malignant pleural effusion with a control rate of approximately 60%. This suggests a promising application of anti-angiogenic drugs in treating malignant thoracic and abdominal effusions. Combinations with other thoracic drugs, such as cisplatin, have also demonstrated improved efficiency compared to single-agent treatments. Bevacizumab, a recombinant humanized monoclonal antibody that binds to VEGF, prevents neovascularization and has shown positive results when used in combination with chemotherapy.
-
Biological agent treatment
The commonly used biological agents in the clinic for treating MPE are primarily Interleukin 2 (IL-2) and Tumor Necrosis Factor (TNF). IL-2 is a thymus-dependent lymphocyte growth factor that stimulates the secretion of interferon and various cytokines. It promotes long-term survival and enhances the killing activity of T cells, as well as the proliferation and activation of natural killer cells, lymphokine-activated killer cells, and tumor-infiltrating lymphocytes. IL-2 is used as an adjuvant therapy for tumors and the treatment of cancerous pleural effusion. Studies have shown that IL-2, whether used alone or in combination with other anti-cancer therapies, can improve survival outcomes for patients with advanced cancer.
Malignant Pleural Effusion Drug Uptake
This section focuses on the uptake rate of potential Malignant Pleural Effusion drugs expected to be launched in the market during the forecast period (2026–2036). The analysis covers the MPE drug’s uptake, performance at peak, factors affecting performance during prime years of growth, patient uptake by therapy, and anticipated sales generated by each drug.
According to DelveInsight estimates, the pipeline for MPE treatments appears moderate, with some therapies expected to launch during the forecast period. These include Gongwing Biopharma’s PTS500, Wuhan YZY Biopharma’s M701, and RS Oncology’s Thiostrepton (RSO-021).
M701, a bispecific antibody developed by Wuhan YZY Biopharma, is under investigation for MPE treatment. At ESMO 2024, intrapleural infusion of M701 showed promising results, including a trend toward preventing pleural effusion buildup, achieving a 61.5% pleurodesis success rate, and extending puncture-free survival to 237 days. M701 is anticipated to launch in EU4 and the UK market by 2029, with peak sales expected to occur within 7 years of its release.
Malignant Pleural Effusion Market Access and Reimbursement of Emerging Therapies
The Malignant Pleural Effusion Therapeutics Market Report further provides detailed insights on the country-wise accessibility and reimbursement scenarios, cost-effectiveness scenario of approved therapies, programs making accessibility easier and out-of-pocket costs more affordable, insights on patients insured under federal or state government prescription drug programs, etc. Reimbursement is a crucial factor that affects the drug’s access to the market. Often, the decision to reimburse comes down to the price of the drug relative to the benefit it produces in treated patients. To reduce the healthcare burden of these high-cost therapies, many payment models are being considered by payers and other industry insiders.
Malignant Pleural Effusion Standard of Care
Once the Malignant Pleural Effusion Diagnosis is made palliation of symptoms and improving the quality of life becomes the priority. The treatment approach varies depending on performance status, type of tumor, and expected survival. In a patient who is asymptomatic and has limited survival, an approach of best supportive care without any intervention is reasonable.
- Therapeutic thoracentesis: It is typically the first step in management and should be performed in most patients with dyspnea. It allows for the assessment of improvement in clinical symptoms, lung re-expansion, and the rate of recurrence. Thoracentesis is a safe procedure, and complication rates have decreased significantly with the routine use of pre-procedural ultrasound.
- Pleurodesis: Pleurodesis is the process of fusing the parietal and visceral pleura, leading to the obliteration of the pleural space and preventing the accumulation of pleural effusion. While the exact mechanism remains unclear, it is believed to involve inflammation and fibrosis, potentially mediated by transforming growth factor beta. Various mechanical and chemical agents, such as talc, bleomycin, tetracycline, corynebacterium parvum, and doxycycline, have been used to achieve pleurodesis.
- Talc slurry versus poudrage: Talc can be administered during thoracoscopy via an atomizer (talc poudrage) or in a suspension form (talc slurry) through a chest tube. A 2004 Cochrane review suggested that talc was the most effective sclerosant and found talc poudrage at thoracoscopy to be more effective than talc slurry in preventing recurrence.
- Tunneled pleural catheter (TPC): The use of TPC is becoming increasingly common for managing MPE. A TPC is a silicone tube placed into the pleural cavity, tunneled under the skin with a small cuff, and exits the body with a one-way valve, allowing patients or caregivers to drain pleural fluid at home easily. TPCs have been shown to be safe and effective in improving symptoms and quality of life.
- Indwelling pleural catheter (IPC): The IPC is a silicone catheter placed in the pleural cavity and tunneled under the skin. It has a cuff at the distal end that forms fibrous adhesions in the subcutaneous tissue over time, securing the IPC in place and preventing ascending infections.
- Anti-angiogenic agents: In recent years, intracavitary biotherapy has become a prominent area of research in the treatment of MPE. Studies have shown that recombinant human VEGF inhibitors, when combined with intracavitary chemotherapy, can control malignant pleural effusion with a control rate of approximately 60%.
- The thoracoperitoneal shunt: For patients whose lungs cannot be expanded or trapped due to MPE, a thoracoabdominal shunt tube can be placed, which is a subcutaneous tunnel from the chest to the abdomen. The liquid is pumped manually, but due to the limited capacity of the pump chamber, it needs to be pumped frequently.
- Immunotherapy: Immunotherapy primarily involves immune cell therapy and immune checkpoint inhibitor therapy (such as PD-1/PD-L1 and CTLA-4 inhibitors). Intrathoracic autoimmune cell infusion therapy is an alternative approach to control MPE, with its effectiveness correlating with a higher frequency of peripheral blood effector T cells. Although its efficacy may be limited, it is generally well tolerated, causes fewer adverse effects, and is not restricted by the patient’s liver or kidney function or physical status.
Industry Experts and Physician Views for Malignant Pleural Effusion
To keep up with MPE market trends, we take Key Opinion Leaders (KOLs) and Subject Matter Experts (SMEs) opinions working in the domain through primary research to fill the data gaps and validate our secondary research. Industry Experts were contacted for insights on the MPE emerging therapies, evolving treatment landscape, patient adherence to conventional therapies, therapy switching trends, drug adoption and uptake, accessibility challenges, and epidemiology and real-world prescription patterns in MPE, including MD, PhD, Instructor, Postdoctoral Researcher, Professor, Researcher, and others.
DelveInsight’s analysts connected with 8+ KOLs to gather insights at country level. Centers such as the Johns Hopkins Hospital, City of Hope National Medical Center, American Thoracic Society, University Medicine Essen etc. were contacted. Their opinion helps understand and validate current and emerging MPE therapies, highlight unmet medical needs, provide epidemiological context, and support strategic decisions for market access, therapy adoption, and pipeline prioritization in MPE.
Region |
Key Opinion Leaders (KOLs) and Subject Matter Experts (SMEs) |
|
United States |
“MPE signals advanced metastatic cancer with a poor prognosis, often leading to only 4–9 months of survival. It severely impacts quality of life through breathlessness, pain, and weight loss. However, Anti-EpCAM x Anti-CD3X therapies, which enhance cancer immunotherapy, are emerging as a promising treatment approach for MPE.” |
|
Germany |
“Advancements in immunocytochemistry and biomarker research have significantly improved MPE diagnosis, particularly in cancer of unknown primary (CUP) cases. Using key markers like EGFR, BRAF, KRAS, and ALK enhances diagnostic accuracy and helps determine the tumor’s origin, ultimately guiding more precise and effective treatment strategies.” |
Malignant Pleural Effusion Qualitative Analysis: SWOT and Conjoint Analysis
We perform qualitative and market Intelligence analysis using various approaches, such as SWOT analysis and conjoint analysis.
In the SWOT analysis of MPE, strengths, weaknesses, opportunities, and threats in terms of disease diagnosis, patient awareness, patient burden, competitive landscape, cost-effectiveness, and geographical accessibility of therapies are provide conjoint analysis analyzes emerging therapies based on relevant attributes such as safety, efficacy, frequency of administration, route of administration, and order of entry. Scoring is given based on these parameters to analyze the effectiveness of therapy.
The team of analysts analyzes promising emerging therapies based on relevant attributes such as safety, efficacy, frequency of administration, route of administration, and order of entry. In efficacy, the trial’s primary and secondary outcome measures are evaluated, whereas the therapies’ safety is evaluated, wherein the acceptability, tolerability, and adverse events are majorly observed. In addition, the scoring is also based on the route of administration, order of entry, probability of success, and the addressable patient pool for each therapy. According to these parameters, the final weightage score and the ranking of the emerging therapies are decided.
Scope of the Malignant Pleural Effusion Market Report
- The Malignant Pleural Effusion Therapeutics Market Report covers a segment of key events, an executive summary, a descriptive overview, explaining their causes, signs and symptoms, pathogenesis, and currently available treatments.
- Comprehensive insight has been provided into the Malignant Pleural Effusion Epidemiology segments and forecasts, the future growth potential of the diagnosis rate, and disease progression along Malignant Pleural Effusion Treatment guidelines.
- Additionally, an all-inclusive account of the emerging treatments, along with the elaborative profiles of late-stage and prominent therapies, will have an impact on the current Malignant Pleural Effusion Treatment Landscape.
- A detailed review of the Malignant Pleural Effusion Market, historical and forecasted Malignant Pleural Effusion Treatment Market Size, Malignant Pleural Effusion Market Share by therapies, detailed assumptions, and rationale behind our approach is included in the report, covering the 7MM drug outreach.
- The Malignant Pleural Effusion Therapeutics Market Report provides an edge while developing business strategies by understanding trends through SWOT analysis and expert insights/KOL views, patient journey, and treatment preferences that help in shaping and driving the 7MM Malignant Pleural Effusion Drugs Market.
Malignant Pleural Effusion Market Report Insights
- Malignant Pleural Effusion Patient Population Forecast
- Malignant Pleural Effusion Therapeutics Market Size
- Malignant Pleural Effusion Pipeline Analysis
- Malignant Pleural Effusion Market Size and Trends
- Malignant Pleural Effusion Market Opportunity (Current and Forecasted)
Malignant Pleural Effusion Market Report Key Strengths
- Epidemiology‑based (Epi‑based) bottom‑up forecasting
- Artificial Intelligence (AI) - enabled market research report
- 11-year Malignant Pleural Effusion Market Forecast
- MPE Market Outlook (North America, Europe, Asia-Pacific)
- Patient Burden Trends (by geography)
- Malignant Pleural Effusion Treatment Addressable Market (TAM)
- Malignant Pleural Effusion Competitive Landscape
- Malignant Pleural Effusion Major Companies Insights
- Malignant Pleural Effusion Price Trends and Analogue Assessment
- Malignant Pleural Effusion Therapies Drug Adoption/Uptake
- Malignant Pleural Effusion Therapies Peak Patient Share analysis
Malignant Pleural Effusion Market Report Assessment
- Malignant Pleural Effusion Current Treatment Practices
- Malignant Pleural Effusion Unmet Needs
- Malignant Pleural Effusion Clinical Development Analysis
- Malignant Pleural Effusion Emerging Drugs Product Profiles
- Malignant Pleural Effusion Market Attractiveness
- Malignant Pleural Effusion Qualitative Analysis (SWOT and conjoint analysis)
Key Questions Answered in the Malignant Pleural Effusion Market Report
Malignant Pleural Effusion Market Insights
- What was the Malignant Pleural Effusion Treatment Market Size, the Malignant Pleural Effusion Market Size by therapies, Malignant Pleural Effusion Market Share (%) distribution in 2025, and what would it look like by 2036? What are the contributing factors for this growth?
- What are the anticipated pricing variations among different geographies for the emerging therapies in the future?
- What can be the future treatment paradigm of Malignant Pleural Effusion?
- What are the disease risks, burdens, and Malignant Pleural Effusion Unmet Needs? What will be the growth opportunities across the 7MM concerning the patient population with Malignant Pleural Effusion?
- Who is the major future competitor in the market, and how will the competitors affect their Malignant Pleural Effusion Market Share?
- What are the current options for the treatment of MPE? What are the current guidelines for treating MPE in the US, Europe, and Japan?
Reasons to Buy the Malignant Pleural Effusion Market Report
- The Malignant Pleural Effusion Therapeutics Market Report will help in developing business strategies by understanding the latest trends and changing treatment dynamics driving the Malignant Pleural Effusion Drugs Market.
- Bottom-up forecasting builds from the affected population to product forecasts, delivering a robust, data-driven approach ideal for new therapies and novel classes.
- Insights on patient burden/disease Malignant Pleural Effusion Incidence, evolution in diagnosis, and factors contributing to the change in the epidemiology of the disease during the forecast years.
- Understand the existing market opportunities in varying geographies and the growth potential over the coming years.
- Identifying strong upcoming players in the Malignant Pleural Effusion Drugs Market will help devise strategies to help get ahead of competitors.
- Detailed analysis and ranking of class-wise potential current and emerging therapies under the conjoint analysis section to provide visibility around leading classes.
- To understand KOLs’ perspectives on the accessibility, acceptability, and compliance-related challenges of existing treatment to overcome barriers in the future.
- Detailed insights on the unmet needs of the existing Malignant Pleural Effusion Drugs Market so that the upcoming players can strengthen their development and launch strategy.
- This Artificial Intelligence (AI) - enabled report summarize and simplify complex datasets within the report into clear, actionable insights for stakeholders, investors, and healthcare providers, enabling faster, data-driven decisions.
Stay updated with us for Recent Articles