Necrotizing Enterocolitis Market Summary
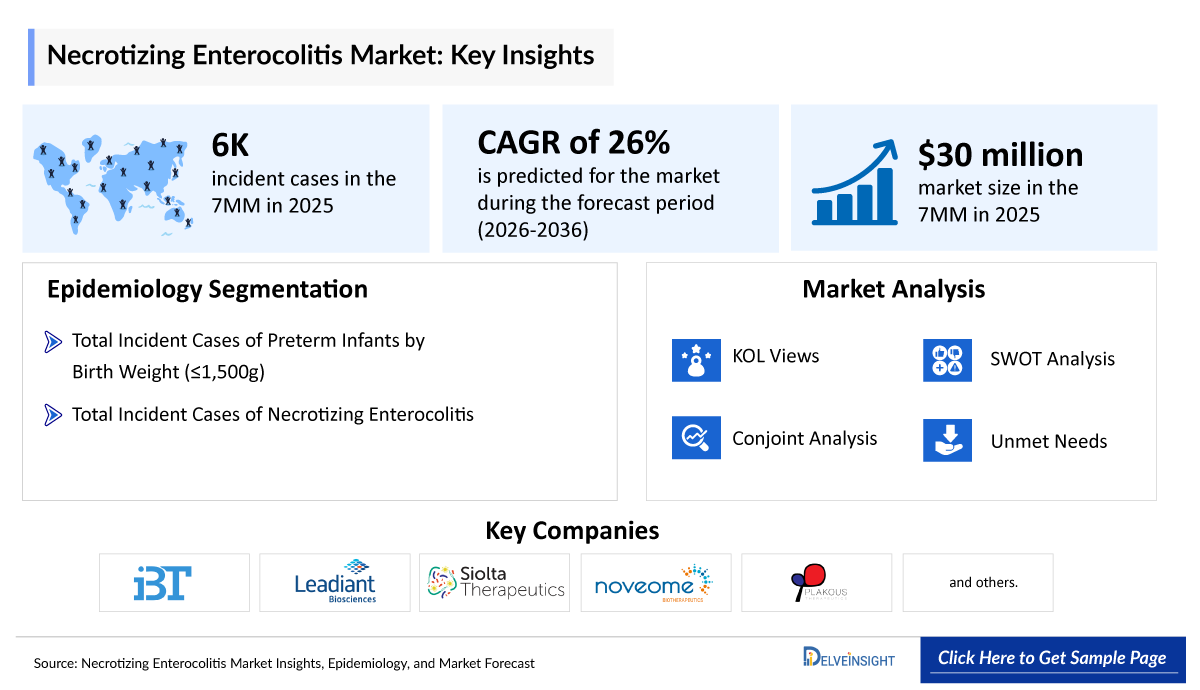
- According to DelveInsight’s analysis, necrotizing enterocolitis market size was found to be ~USD 30 million in the leading markets (the United States, the EU4 (Germany, France, Italy, and Spain), the United Kingdom, and Japan) in 2025.
- Necrotizing Enterocolitis Market is expected to grow at a CAGR of 26% by 2036.
Necrotizing Enterocolitis Market Insights and Trends
- Necrotizing Enterocolitis is a severe and life-threatening gastrointestinal emergency that primarily affects premature infants in neonatal intensive care units worldwide. Despite major advancements in neonatal care, its exact etiology remains incompletely understood, making it a key area of research focus.
- At birth, neonates are exposed to environmental bacteria; however, preterm infants experience delayed and abnormal gut colonization. This leads to exaggerated inflammatory responses and contributes to necrotizing enterocolitis development. Feeding practices further influence risk delayed enteral feeding and reliance on total parenteral nutrition increase susceptibility, whereas early enteral feeding may offer protective benefits.
- Nutritional practices play a crucial role in necrotizing enterocolitis risk, as delayed initiation of enteral feeding and dependence on total parenteral nutrition can lead to intestinal mucosal atrophy and increased susceptibility, whereas early enteral feeding may help reduce the risk of disease development.
- Inflammatory and immunological pathways are central to necrotizing enterocolitis pathogenesis, particularly the imbalance between Toll-like receptors TLR4 and TLR9, where increased TLR4 activation contributes to intestinal injury by promoting apoptosis and impairing enterocyte proliferation and migration.
- Clinical management of necrotizing enterocolitis is dependent on disease severity, with early-stage disease (Stages I and II) typically managed conservatively through bowel rest, broad-spectrum antibiotics, and supportive care, while advanced disease (Stage III) often requires surgical intervention, including procedures such as laparotomy or resection of necrotic bowel.
- Due to the absence of highly effective targeted therapies for necrotizing enterocolitis, current research and clinical focus have shifted toward preventive strategies, including early feeding with colostrum and mother’s milk, careful nutritional management, probiotic use, environmental control, and supportive interventions such as skin-to-skin care.
- Few companies are persistently working toward the development of new treatment therapies. Only Infant Bacterial Therapeutics has product at the advanced stage; however, companies like Noveome Biotherapeutics, Siolta Therapeutics, and Plakous Therapeutics have their assets in the early or preclinical stage.
Necrotizing Enterocolitis Market Size and Forecast in the 7MM
- 2025 Necrotizing Enterocolitis Market Size: ~USD 30 million
- 2036 Projected Necrotizing Enterocolitis Market Size: ~USD 800 million
- Necrotizing Enterocolitis Growth Rate (2026–2036): 26% CAGR
DelveInsight's ‘Necrotizing Enterocolitis– Market Insights, Epidemiology and Market Forecast – 2036’ report delivers an in-depth understanding of the Necrotizing Enterocolitis, historical and forecasted epidemiology, as well as the necrotizing enterocolitis market trends in the United States, EU4 (Germany, Spain, Italy, and France) and the United Kingdom, and Japan.
The Necrotizing Enterocolitis market report delivers a comprehensive analysis of the current treatment landscape, including standards of care, clinical practices, and evolving therapeutic algorithms. It evaluates, necrotizing enterocolitis patient burden trends, revenue & market share dynamics, peak patient share & therapy uptake analysis, and provides an in-depth market size assessment, and growth rate projections (Historical & Forecast 2022–2036) across global regions. The report highlights key unmet medical needs in necrotizing enterocolitis and maps the competitive and clinical landscape to uncover high‑value opportunities, providing a clear outlook on future market growth potential.
Scope of the Necrotizing Enterocolitis Market Report | |
|
Study Period |
2022–2036 |
|
Historical Year |
2022–2025 |
|
Forecast Period |
2026–2036 |
|
Base Year |
2026 |
|
Geographies Covered |
|
|
Necrotizing Enterocolitis Market CAGR (Forecast period) |
26% (2026–2036) |
|
Necrotizing Enterocolitis Epidemiology Segmentation Analysis |
Patient Burden Assessment
|
|
Necrotizing Enterocolitis Companies |
|
|
Necrotizing Enterocolitis Therapies |
|
|
Necrotizing Enterocolitis Market |
Segmented by
|
|
Analysis |
|
Key Factors Driving the Necrotizing Enterocolitis Market
High Disease Burden in Premature Infants
Necrotizing Enterocolitis remains one of the most serious and life-threatening gastrointestinal emergencies in preterm infants, particularly those in neonatal intensive care units. The increasing survival of extremely premature infants, driven by advancements in neonatal care, has inadvertently expanded the at-risk population, thereby sustaining the clinical and economic burden of necrotizing enterocolitis globally.
Significant Unmet Medical Need
Despite decades of research, there are no approved targeted pharmacological therapies for necrotizing enterocolitis, and current management relies primarily on supportive care and surgical intervention in advanced stages. This lack of effective treatment options highlights a substantial unmet need, creating strong opportunities for the development of disease-modifying therapies.
Growing Focus on Preventive Strategies
With limited treatment options available, there is an increasing shift toward prevention-based approaches. Strategies such as early feeding with colostrum and mother’s milk, optimized nutritional protocols, probiotic supplementation, and improved neonatal care practices are gaining importance in reducing necrotizing enterocolitis incidence.
Necrotizing Enterocolitis Understanding and Treatment Algorithm
Necrotizing Enterocolitis Overview and Diagnosis
Necrotizing Enterocolitis is a severe, life-threatening gastrointestinal disease primarily affecting premature infants, characterized by intestinal inflammation, bacterial invasion, and progressive necrosis. It may lead to intestinal perforation, resulting in peritonitis, sepsis, and death. Symptoms are often nonspecific (e.g., poor feeding, vomiting, lethargy, abdominal tenderness), requiring high clinical suspicion. The exact etiology remains unclear, but intestinal immaturity, abnormal bacterial colonization, and exaggerated inflammatory responses are key contributors, with progression from inflammation to ischemia, necrosis, and perforation. Management is primarily supportive, including stabilization (ABC), fluid resuscitation, gastric decompression, broad-spectrum antibiotics, and total parenteral nutrition, with enteral feeding resumed after improvement. Severe or non-responsive cases require surgical intervention (e.g., laparotomy, removal of necrotic bowel, ostomy), while peritoneal drainage may be used in critically ill infants.
After a premature baby is born, steps are taken to prevent the infant from getting infections. If these preventive measures are unsuccessful, the baby can experience symptoms like a swollen abdomen, tiredness, and breathing pauses. Doctors conduct tests such as a complete blood count, a C-reactive protein test, and x-rays to diagnose the condition. After these tests, it is confirmed that the baby has necrotizing enterocolitis.
Further details are provided in the report.
Necrotizing Enterocolitis Treatment Landscape
No pharmaceutical therapies are currently approved specifically for necrotizing enterocolitis. Due to the lack of effective treatments and the high mortality associated with the disease, research efforts have increasingly shifted toward preventive strategies, including early exposure to colostrum and mother’s milk, optimized nutritional management, probiotics, environmental protection, skin-to-skin care (SSC), and pharmacological approaches. While probiotics (dietary supplements containing live bacteria) are suggested to help prevent necrotizing enterocolitis, they are not subject to FDA premarket approval for safety and efficacy or the same manufacturing and quality standards required for regulated drugs. Safety concerns are further highlighted by a reported case in 2014, where a premature infant in a US NICU died from gastrointestinal mucormycosis linked to a contaminated probiotic supplement.
Further details related to country-based variations are provided in the report.
Necrotizing Enterocolitis Unmet Needs
The section “unmet needs of Necrotizing Enterocolitis” outlines the critical gaps between the current state of patient care, diagnosis, and the ideal & effective management of the disease. It highlights the obstacles experienced by patients, clinicians, and researchers and identifies potential solutions for future progress.
- Lack of noninvasive treatment
- Challenges in diagnosing necrotizing enterocolitis
- High mortality rate
- Reduced quality of life
- Unclear etiology
- Novel research strategies for prevention, and others…..
Note: Comprehensive unmet needs insights in Necrotizing Enterocolitis and their strategic implications are provided in the full report.
Necrotizing Enterocolitis Epidemiology
Key Findings from Necrotizing Enterocolitis Epidemiological Analysis and Forecast:
- In 2025, the United States continues to report the highest absolute number of very low birth weight (≤1,500 g) preterm infants among the 7MM, driven by higher total birth volume compared with European countries and Japan. However, rates of VLBW births have remained relatively stable in recent years, with minor fluctuations. As per the estimate, the incident cases of preterm infants by birth weight (≤1,500 g) in 7MM are anticipated to increase during the forecast period of 2026–2036.
- Across the 7MM, the population of preterm infants with birth weight ≤1,500 g is expected to remain stable to slightly increasing through 2030, largely due to improved neonatal survival rates rather than a significant rise in preterm birth incidence. This trend is consistently reported by World Health Organization and regional neonatal databases.
- The incidence of necrotizing enterocolitis in high-income regions, including the United States and EU4 and the UK, has stabilized or modestly declined by 2025, driven by advances in neonatal care such as human milk feeding, standardized NICU practices, and improved infection control. Although probiotic use is increasing, organizations like the American Academy of Pediatrics highlight inconsistent evidence and lack of standardized formulations, limiting widespread guideline adoption.
- In the United States, the annual incidence of necrotizing enterocolitis remains in the range of ~3,000–5,000 cases, with recent estimates (2024–2025) suggesting stabilization or modest decline, particularly in tertiary NICUs implementing preventive bundles. This aligns with surveillance data from Centers for Disease Control and Prevention and neonatal networks.
- Within the EU4 (Germany, France, Italy, Spain), Germany continues to report the highest number of VLBW preterm infants, followed by France, reflecting population size and birth rates, while Spain reports comparatively lower absolute numbers. This pattern remains consistent in 2025 neonatal registry updates.
- In Japan, the incidence of necrotizing enterocolitis remains low (~100–200 cases annually), driven by declining birth rates, advanced neonatal care, and strong breastfeeding practices. Recent data (2024–2025), including from the Japan Neonatal Research Network, indicate stable to slightly declining incidence, reflecting ongoing improvements in neonatal management and prevention.
Necrotizing Enterocolitis Drug Analysis & Competitive Landscape
The Necrotizing Enterocolitis drug chapter provides a detailed emerging pipeline across Phase I/II–III clinical trials. It covers mechanism of action, clinical trial data, patents, collaborations, strategic partnerships upcoming Key catalyst for each therapy, along with their advantages, limitations, and recent developments. This section offers critical insights into the Necrotizing Enterocolitis pipeline therapies, treatment landscape, supporting market assessment, competitive analysis, and growth forecasting for the necrotizing enterocolitis therapeutics market.
Necrotizing Enterocolitis Pipeline Analysis
IBP-9414: Infant Bacterial Therapeutics
IBT is currently developing the drug candidate IBP-9414. The ambition for IBP-9414 is to become the world’s first approved probiotic drug to prevent life-threatening diseases in premature infants, including necrotizing enterocolitis and sepsis, by promoting healthy stomach and bowel development in premature infants. IBP-9414 contains the active compound Lactobacillus reuteri, a human bacterial strain naturally present in breast milk. Lactobacillus reuteri is a live bacterium known to be anti-inflammatory, anti-pathogenic, and beneficial to gut motility and mucosa maturity. IBP-9414 is currently in an ongoing registration-enabling pivotal Phase III study to prevent necrotizing enterocolitis. The company is expected to submit an approval application in 2026, and results from the pivotal Phase III “Connection Study” in very low birth weight infants have been published in Pediatric Research (Nature).
STMC-106: Siolta Therapeutics
Siolta Therapeutics is developing the drug candidate STMC-106 as part of its microbiome-based live biotherapeutic pipeline. The program is focused on preventing and treating necrotizing enterocolitis in preterm infants by leveraging targeted microbial therapies to support gut health and reduce disease risk. STMC-106 is currently in preclinical development and has received grant support to advance its research, with the aim of progressing toward clinical evaluation.
Necrotizing Enterocolitis Competitive Landscape of Pipeline Drugs | ||||||
|
Drug Name |
Company |
Highest Phase |
Indication |
RoA |
MoA |
Anticipated Launch in the US |
|
IBP-9414 |
Infant Bacterial Therapeutics |
Registar |
Prevention of NEC |
Oral |
Probiotic |
Information is available in the full report |
|
STMC-106 |
Siolta Therapeutics |
Pre-clinical |
Prevention of NEC |
NA |
Mixed-species LBP reduces pathobiont colonization and restores intestinal epithelial barrier function |
Information is available in the full report |
|
ST266 |
Noveome Biotherapeutics |
Pre-clinical |
Prevention of NEC |
NA |
Potential neuroprotective cultured amnion-derived multipotent progenitor cells |
Information is available in the full report |
|
Protego-PD |
Plakous Therapeutics |
Pre-clinical |
Prevention of NEC |
NA |
Human placental extract with anti-inflammatory and immunomodulatory capabilities |
Information is available in the full report |
|
Note: Launch insights are provisional and may change with future report updates or the occurrence of major key catalysts. | ||||||
Note: Detailed emerging therapies assessment will be provided in the final report.
Necrotizing Enterocolitis Key Players, Market Leaders and Emerging Companies
- Infant Bacterial Therapeutics
- Leadiant Biosciences
- Siolta Therapeutics
- Noveome Biotherapeutics
- Plakous Therapeutics, and others
Recent Developments in Necrotizing Enterocolitis Clinical Trial
- In March 2026, Noveome Biotherapeutics announced the independent Data Safety Monitoring Board (DSMB) has completed its review of safety data from Cohort 1 of the ongoing clinical trial evaluating ST266 for the treatment of necrotizing enterocolitis in neonates.
- In March 2026, Infant Bacterial Therapeutics announced announce that results from its pivotal Phase III trial, with IBP-9414 in very low birth weight infants “The Connection Study” have now been published in Pediatric Research (a Nature Publication).
- As of December 2025, Infant Bacterial Therapeutics announced that it had entered into a partnership with BioConnection for the production of IBP-9414 drug product.In November 2025, Infant Bacterial Therapeutics announced that company has decided to pursue an accelerated approval pathway for IBP-9414.
Necrotizing Enterocolitis Market Outlook
The necrotizing enterocolitis market remains largely unchanged and continues to rely on supportive care approaches, including bowel rest, broad-spectrum antibiotics, parenteral nutrition, and surgical intervention in severe cases. Despite being the leading gastrointestinal cause of mortality and the primary surgical emergency in neonates, there are no approved targeted pharmacological therapies for necrotizing enterocolitis. The current treatment paradigm focuses on stabilizing the patient and limiting disease progression through intensive supportive measures such as bowel decompression, fluid resuscitation, respiratory and cardiovascular support, and close clinical monitoring. However, outcomes remain suboptimal, particularly in advanced stages requiring surgical resection, which is associated with significant morbidity and long-term complications.
Given the high mortality rate, clinical complexity, and substantial economic burden, the necrotizing enterocolitis landscape is increasingly shifting toward preventive strategies rather than treatment. Approaches such as early feeding with colostrum and mother’s milk, optimized nutritional practices, and microbiome modulation through probiotics have gained considerable attention. Although probiotics have demonstrated potential in reducing necrotizing enterocolitis incidence in multiple meta-analyses, their adoption remains inconsistent due to lack of standardized guidelines, safety concerns, and absence of FDA-approved pharmaceutical-grade products. Regional variability is evident, with higher adoption rates in Europe compared to relatively limited but gradually increasing use in the United States.
The pipeline landscape is gradually evolving, with microbiome-based therapies and novel preventive approaches gaining traction. Among these, IBP-9414, a pharmaceutical-grade probiotic currently in Phase III clinical trials, represents a potential first-in-class therapy targeting necrotizing enterocolitis prevention. If approved, it could establish a regulatory and clinical precedent for microbiome-based interventions in neonatal care. Overall, increasing disease awareness, ongoing clinical research, and the emergence of late-stage pipeline candidates are expected to drive gradual transformation in the necrotizing enterocolitis market, with a strong focus on prevention, early intervention, and mechanism-based therapies over the forecast period.
Key findings from Necrotizing Enterocolitis Market Report
- Among the 7MM, the United States accounts for the largest potential market, driven by high NICU admissions and healthcare expenditure, although treatment options remain limited.
- In the forecast period, pipeline therapies such as IBP-9414 are expected to capture significant attention and potential market share, given the absence of approved treatments.
- The entry of microbiome-based and preventive therapies is anticipated to reshape the necrotizing enterocolitis treatment landscape, intensifying competition and innovation in the coming years.
Further details will be provided in the report….
Drug Class/Insights into Leading Emerging and Marketed Therapies in Necrotizing Enterocolitis (2022–2036 Forecast)
The necrotizing enterocolitis treatment landscape remains highly underserved, with no approved pharmacological therapies currently available, and management primarily reliant on supportive care and surgical intervention. Emerging pipeline approaches are beginning to reshape the space, including microbiome-based therapies and regenerative biologics aimed at addressing underlying disease mechanisms such as intestinal immaturity, dysbiosis, and inflammation. For instance, investigational assets like STMC-106 from Siolta Therapeutics and Protego-PD from Plakous Therapeutics highlight innovative strategies targeting early-life gut health and immune modulation. While these candidates remain in preclinical stages, they reflect growing scientific and clinical interest in preventive interventions for high-risk preterm infants. The advancement of such therapies, alongside increasing focus on standardized neonatal care practices, is expected to gradually transform the necrotizing enterocolitis landscape over the coming decade, although significant unmet needs persist in early diagnosis, effective prevention, and targeted treatment options.
Necrotizing Enterocolitis Drug Uptake
This section focuses on the uptake rate of potential Necrotizing Enterocolitis drugs expected to be launched in the market during the forecast period (2026–2036). The analysis covers the Necrotizing Enterocolitis drug’s uptake, performance at peak, factors affecting performance during prime years of growth, patient uptake by therapy, and anticipated sales generated by each drug.
Pipeline candidate IBP-9414 is expected to follow a moderate uptake trajectory, reflecting its investigational status and gradual adoption as clinical evidence emerges. Meanwhile, STP206 is anticipated to show gradual uptake.
Detailed insights of emerging therapies' drug uptake is included in the report
Market Access and Reimbursement of Approved therapies in Necrotizing Enterocolitis
Reimbursement of rare disease therapies can be limited due to lack of supporting policies and funding, challenges of high prices, lack of specific approaches to evaluating rare disease drugs given limited evidence, and payers’ concerns about budget impact. The high cost of rare disease drugs usually has a limited effect on the budget due to the small number of eligible patients being prescribed the drug. The US FDA has approved several rare disease therapies recently. From a patient perspective, health insurance and payer coverage guidelines surrounding rare disease treatments restrict broad access to these treatments, leaving only a small number of patients who can bypass insurance and pay for products independently.
The report further provides detailed insights on the country-wise accessibility and reimbursement scenarios, cost-effectiveness scenario of currently used therapies, programs making accessibility easier and out-of-pocket costs more affordable, insights on patients insured under federal or state government prescription drug programs, etc.
Probiotics: Requires a Letter of Medical Necessity (LMN) to be eligible with a Flexible SpendingAccount (FSA).
Probiotics may be eligible with a Letter of Medical Necessity (LMN) with a flexible spending account(FSA), health savings account (HSA), or health reimbursement arrangement (HRA). Probiotics are noteligible for reimbursement with limited-purpose flexible spending accounts (LPFSA) and dependentcare flexible spending accounts (DCFSA).
NOTE: Further Details are provided in the final report….
Necrotizing Enterocolitis Therapies Price Scenario & Trends
Pricing and analogue assessment of necrotizing enterocolitis therapies highlights evolving price dynamics structures. This section summarizes the cost of approved treatments, closest and most appropriate analogue selection for emerging therapies, and understanding of how pricing influences market access, adherence, and long-term uptake.
Industry Experts and Physician Views for Necrotizing Enterocolitis
To keep up with Necrotizing Enterocolitis market trends, we take Key Opinion Leaders (KOLs) and Subject Matter Experts (SMEs) opinions working in the domain through primary research to fill the data gaps and validate our secondary research. Industry experts were contacted for insights on the Necrotizing Enterocolitis emerging therapies, evolving treatment landscape, patient adherence to conventional therapies, therapy switching trends, drug adoption and uptake, accessibility challenges, and epidemiology and real-world prescription patterns in Necrotizing Enterocolitis, including MD, PhD, Instructor, Postdoctoral Researcher, Professor, Researcher, and others.
DelveInsight’s analysts connected with 10+ KOLs to gather insights at country level. Centers such as the Nationwide Children’s Hospital, Columbus, OH, the United States, University Hospitals Dorset NHS Foundation Trust, the United Kingdom, Department of Pediatrics, North Shore University Health System, Evanston, Illinois, etc., were contacted.Their opinion helps understand and validate current and emerging Necrotizing Enterocolitis therapies, highlight unmet medical needs, provide epidemiological context, and support strategic decisions for market access, therapy adoption, and pipeline prioritization in Necrotizing Enterocolitis.
What are the latest KOL Views on Necrotizing Enterocolitis Market? | |
|
Region |
Key Opinion Leaders (KOLs) and Subject Matter Experts (SMEs) |
|
United States |
“Given the significant mortality and morbidity, there is an urgent need to develop improved prophylactic measures against the NEC. One potential preventative strategy for NEC is the use of probiotics. Although there has been significant interest for decades in probiotics in neonatal care, no clear guidelines exist regarding which probiotic to use or for which patients, and no FDA-approved products exist on the market for NEC.” |
|
United Kingdom |
“Since the term was first introduced, the general medical approach to NEC has mostly remained unchanged, involving actions such as holding back feeds, administering antibiotics, and performing surgery when necessary. Presently, NEC is the leading surgical emergency in newborns and the primary cause of gastrointestinal mortality in this delicate patient group.” |
Necrotizing Enterocolitis Qualitative Analysis: SWOT and Conjoint Analysis
We perform qualitative and market Intelligence analysis using various approaches, such as SWOT analysis and conjoint analysis.
In the SWOT analysis of Necrotizing Enterocolitis, strengths, weaknesses, opportunities, and threats in terms of disease diagnosis, patient awareness, patient burden, competitive landscape, cost-effectiveness, and geographical accessibility of therapies are provided.
Conjoint analysis analyzes emerging therapies based on relevant attributes such as safety, efficacy, frequency of administration, route of administration, and order of entry. Scoring is given based on these parameters to analyze the effectiveness of therapy.
The team of analysts analyzes promising emerging therapies based on relevant attributes such as safety, efficacy, frequency of administration, route of administration, and order of entry. In efficacy, the trial’s primary and secondary outcome measures are evaluated, whereas the therapies’ safety is evaluated, wherein the acceptability, tolerability, and adverse events are majorly observed. In addition, the scoring is also based on the route of administration, order of entry, probability of success, and the addressable patient pool for each therapy. According to these parameters, the final weightage score and the ranking of the emerging therapies are decided.
Scope of the Necrotizing Enterocolitis Market Report
- The Necrotizing Enterocolitis Market report covers a segment of key events, an executive summary, a descriptive overview of Necrotizing Enterocolitis, explaining their causes, signs and symptoms, pathogenesis, and currently available treatments.
- Comprehensive insight has been provided into the epidemiology segments and forecasts, the future growth potential of the diagnosis rate, and disease progression along treatment guidelines.
- Additionally, an all-inclusive account of both the current and emerging treatments, along with the elaborative profiles of late-stage and prominent therapies, will have an impact on the current treatment landscape.
- A detailed review of the Necrotizing Enterocolitis market, historical and forecasted market size, market share by therapies, detailed assumptions, and rationale behind our approach is included in the report, covering the 7MM drug outreach.
- The Necrotizing Enterocolitis Market report provides an edge while developing business strategies by understanding trends through SWOT analysis and expert insights/KOL views, patient journey, and treatment preferences that help in shaping and driving the 7MM Necrotizing Enterocolitis market.
Necrotizing Enterocolitis Market Report Insights
- Necrotizing Enterocolitis Patient Population Forecast
- Necrotizing Enterocolitis Therapeutics Market Size
- Necrotizing Enterocolitis Pipeline Analysis
- Necrotizing Enterocolitis Market Size and Trends
- Necrotizing Enterocolitis Market Opportunity (Current and forecasted)
Necrotizing Enterocolitis Market Report Key Strengths
- Epidemiology‑based (Epi‑based) Bottom‑up Forecasting
- Artificial Intelligence (AI)-Enabled Market Research Report
- 11-Year Forecast
- Necrotizing Enterocolitis Market Outlook (North America, Europe, Asia-Pacific)
- Patient Burden Trends (By Geography)
- Necrotizing Enterocolitis Treatment Addressable Market (TAM)
- Necrotizing Enterocolitis Competitve Landscape
- Necrotizing Enterocolitis (NEC)) Major Companies Insights
- Necrotizing Enterocolitis Price Trends and Analogue Assessment
- Necrotizing Enterocolitis Therapies Drug Adoption/Uptake
- Necrotizing Enterocolitis Therapies Peak Patient Share Analysis
Necrotizing Enterocolitis Market Report Assessment
- Necrotizing Enterocolitis Current Treatment Practices
- Necrotizing Enterocolitis Unmet Needs
- Necrotizing Enterocolitis Clinical Development Analysis
- Necrotizing Enterocolitis Emerging Drugs Product Profiles
- Necrotizing Enterocolitis Market attractiveness
- Necrotizing Enterocolitis Qualitative Analysis (SWOT and conjoint analysis)
Key Questions Answered in the Necrotizing Enterocolitis Market
Necrotizing Enterocolitis Market Insights
- What was the Necrotizing Enterocolitis market size, the market size by therapies, market share (%) distribution in 2025, and what would it look like by 2036? What are the contributing factors for this growth?
- What are the anticipated pricing variations among different geographies for the emerging therapies in the future?
- What can be the future treatment paradigm of Necrotizing Enterocolitis?
- What are the disease risks, burdens, and unmet needs of Necrotizing Enterocolitis? What will be the growth opportunities across the 7MM concerning the patient population with Necrotizing Enterocolitis?
- Who is the major future competitor in the market, and how will the competitors affect their market share?
- What are the current options for the treatment of Necrotizing Enterocolitis? What are the current guidelines for treating Necrotizing Enterocolitis in the US, Europe, and Japan?
Reasons to Buy the Necrotizing Enterocolitis Market Report
- The Necrotizing Enterocolitis Market report will help in developing business strategies by understanding the latest trends and changing treatment dynamics driving the Necrotizing Enterocolitis market.
- Bottom‑up forecasting builds from the affected population to product forecasts, delivering a robust, data‑driven approach ideal for new therapies and novel classes.
- Insights on patient burden/disease incidence, evolution in diagnosis, and factors contributing to the change in the epidemiology of the disease during the forecast years.
- Understand the existing Necrotizing Enterocolitis Market opportunities in varying geographies and the growth potential over the coming years.
- Identifying strong upcoming players in the market will help devise strategies to help get ahead of competitors.
- Detailed analysis and ranking of class-wise potential current and emerging therapies under the conjoint analysis section to provide visibility around leading classes.
- To understand KOLs’ perspectives on the accessibility, acceptability, and compliance-related challenges of existing treatment to overcome barriers in the future.
- Detailed insights on the unmet needs of the existing Necrotizing Enterocolitis Market so that the upcoming Necrotizing Enterocolitis companies can strengthen their development and launch strategy.
- This Artificial Intelligence (AI)‑enabled report summarize and simplify complex datasets with in the report into clear, actionable insights for stakeholders, investors, and healthcare providers, enabling faster, data‑driven decisions.

-epidemiology.png&w=256&q=75)

