Preeclampsia Market Summary
- The Preeclampsia Market Size in the 7MM is expected to grow from USD 87 Million in 2025 to USD 116 Million by 2036.
- The Preeclampsia Market is projected to grow at a CAGR of 3.30% by 2036 in leading countries like the US, EU4, UK, and Japan.
Preeclampsia Market Size and Epidemiological Analysis
- The total Preeclampsia Market Size in the 7MM, the United States accounted for the highest market share, i.e., approximately USD 70 million in 2024.
- Preeclampsia is a complex pregnancy disorder characterized by new-onset hypertension and proteinuria after 20 weeks of gestation. It is believed to stem from placental dysfunction, leading to systemic endothelial activation and inflammatory responses that affect maternal circulation. Preeclampsia is more prevalent in first-time mothers, women with a family history of the condition, and those with preexisting health issues such as hypertension or diabetes.
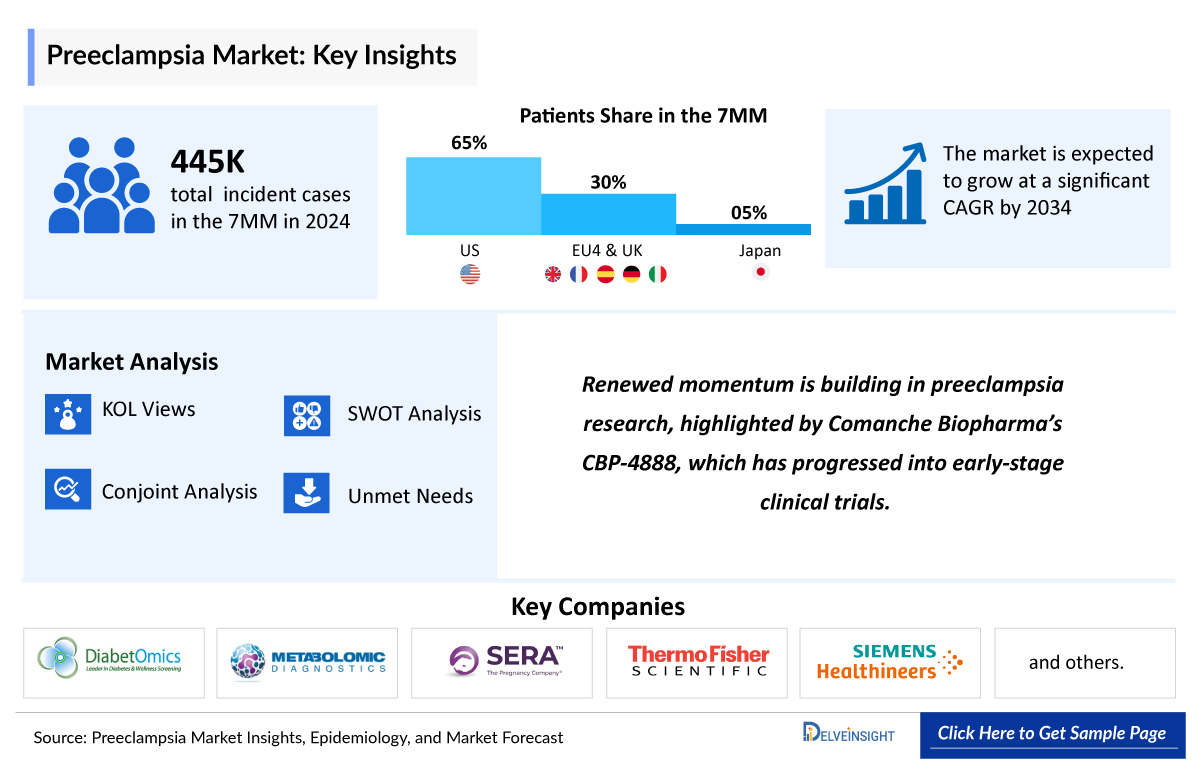
- In 2025, 7MM accounted for nearly 447,000 incident cases of preeclampsia.
- The US accounted for the highest number of diagnosed incident cases of preeclampsia accounting for more than half of the total cases of 7MM in 2025.
- In 2025, the 30–34-year age group accounted for the highest number of preeclampsia cases across EU4 and the UK, with approximately 53,000 reported cases.
- Diagnosis of preeclampsia is based on routine prenatal screenings measuring blood pressure and urine protein, confirmed with readings of =140/90 mmHg and proteinuria =300 mg in a 24-h urine collection.
- Development of advanced diagnostic tools like BRAHMS PIGF plus KRYPTOR and BRAHMS sFlt-1 KRYPTOR by Thermo Fisher Scientific and blood tests allows for better prediction and management of preeclampsia.
- The differential diagnosis of preeclampsia includes antiphospholipid antibody syndrome, thrombotic microangiopathies, lupus nephritis, epilepsy or seizure disorder, chronic renal disease, chronic liver disease, gestational hypertension, chronic hypertension, and others.
- Among severity-specific incident cases, mild form of preeclampsia is the most common one in the 7MM.
- There is no approved therapies for preeclampsia, delivery is the only cure and management majorly relies on antihypertensive agents, anticonvulsants, and corticosteroids
- Currently emerging space for preeclampsia is not so active, only few companies are developing and they are also in preclinical stage. Recently Comanche biopharma’s CBP-4888 has become active with Fast Track Designation (FTD) for preeclampsia.
Request for unlocking the Sample Page of the "Preeclampsia Market Insights"
Key Factors Driving the Preeclampsia Market
- Rising Incidence of High-Risk Pregnancies: The increasing prevalence of maternal risk factors such as advanced maternal age, obesity, diabetes, hypertension, and assisted reproductive technologies is contributing to a higher incidence of preeclampsia, driving demand for early diagnosis and effective management solutions.
- Growing Awareness and Early Screening Initiatives: Improved awareness among healthcare providers and expectant mothers, along with wider adoption of prenatal screening and monitoring programs, is supporting earlier detection and timely intervention for preeclampsia.
- Advancements in Diagnostic Technologies: Innovations in biomarkers, blood-based tests, and non-invasive monitoring tools are enhancing diagnostic accuracy, enabling risk stratification and personalized pregnancy management.
- Unmet Need for Targeted Therapies: The lack of disease-modifying treatments beyond symptomatic management and early delivery is encouraging research and investment into novel therapeutics aimed at improving maternal and fetal outcomes.
- Supportive Government and Regulatory Initiatives: Public health programs, maternal health policies, and funding for women’s healthcare research are strengthening access to prenatal care and accelerating clinical development in the preeclampsia market.
DelveInsight's “Preeclampsia Market Insight, Epidemiology and Market Forecast – 2036” report delivers an in-depth analysis of Preeclampsia epidemiology, market, and clinical development understanding, Addition to this report provides historical and forecasted epidemiology and market data as well as a detailed analysis on the Preeclampsia therapeutics market trends in the United States, EU4 (Germany, Spain, Italy, and France) and the United Kingdom, and Japan.
The Preeclampsia Market report provides current treatment practices, emerging drugs, market share of individual therapies, and current and forecasted 7MM Preeclampsia Market Size from 2022 to 2036. The report also covers Preeclampsia treatment practices and unmet medical needs to curate the best opportunities and assess the market’s potential
Scope of the Preeclampsia Market Report | |
|
Study Period |
2022 to 2036 |
|
Forecast Period |
2025-2036 |
|
Geographies Covered |
|
|
Preeclampsia Market |
|
|
Preeclampsias Market Size | |
|
Preeclampsia Companies |
Diabetomics, Inc., Metabolomic Diagnostics Ltd., Sera Prognostics, Thermo Fisher Scientific Inc., Siemens Healthineers AG, Bayer AG, and others. |
|
Preeclampsia Epidemiology Segmentation |
|
Preeclampsia Disease Understanding
Preeclampsia Overview
Hypertensive disorders of pregnancy constitute a leading cause of maternal and perinatal mortality worldwide. Preeclampsia, with or without severe features, is a disorder of pregnancy associated with new-onset hypertension, usually with accompanying proteinuria, which occurs most often after 20 weeks of gestation and frequently near term. This disease represents a spectrum of hypertensive disease in pregnancy, beginning with gestational hypertension and progressing to develop severe features, ultimately leading to its more severe manifestations, such as eclampsia and HELLP (hemolysis, elevated liver enzymes, low platelet count) syndrome. Early diagnosis and prompt management are essential to preventing both maternal and neonatal complications through symptomatic management and delivery planning.
Further details are provided in the report…
Preeclampsia Diagnosis
Preeclampsia is diagnosed based on the presence of hypertension (systolic BP = 140 mmHg or diastolic BP = 90 mmHg) after 20 weeks of gestation, along with proteinuria (= 300 mg/24 hours or a protein-to-creatinine ratio = 0.3). In the absence of proteinuria, it can still be diagnosed if there are signs of organ dysfunction, such as renal impairment (creatinine > 1.1 mg/dL), elevated liver enzymes, neurological symptoms (e.g., headache, visual disturbances), or thrombocytopenia. Severe preeclampsia is characterized by significantly elevated BP (= 160/110 mmHg), marked proteinuria, or complications like pulmonary edema and organ damage.
Further details related to country-based variations are provided in the report…
Preeclampsia Treatment
To manage high blood pressure, medications like labetalol, nifedipine, or methyldopa may be prescribed. Labetalol is specifically licensed for pregnant women, while the others are used off-label when the benefits outweigh the risks. In severe cases, anticonvulsant medicine may be given to prevent or treat fits. Delivery is typically recommended around the 37th to 38th week, either through induced labor or cesarean section, to minimize complications. If the condition worsens before 37 weeks, earlier delivery may be necessary. Premature birth may pose additional risks for the baby. After delivery, preeclampsia generally improves, but complications can develop in the days following birth. Blood pressure will continue to be monitored, and additional medication may be prescribed if necessary. The baby may require monitoring in a neonatal intensive care unit if born prematurely. Postpartum, the patient will have follow-up appointments to check blood pressure and assess whether ongoing treatment is needed.
Further details related to treatment and management are provided in the report…
Preeclampsia Epidemiology
The Preeclampsia epidemiology chapter in the report provides historical as well as forecasted epidemiology segmented by Total Incidence Cases, Total Diagnosed Incidence Cases, Age-specific Cases, Sub-type specific Cases, Severity specific Cases, Treated Cases of Preeclampsia in the United States, EU4 countries (Germany, France, Italy, and Spain) and the United Kingdom, and Japan from 2022 to 2036.
Key Findings from the Preeclampsia Epidemiological Analyses and Forecasts
- In 2025, 7MM accounted for nearly 447,000 incident cases of preeclampsia.
- The US accounted for the highest number of diagnosed incident cases of preeclampsia accounting for more than half of the total cases of 7MM in 2025.
- In 2025, the 30–34-year age group accounted for the highest number of preeclampsia cases across EU4 and the UK, with approximately 53,000 reported cases.
- In 2025, late-onset preeclampsia accounted for approximately 260,000 cases in the US, making it the most prevalent subtype.
- In 2025, Japan reported 25,000 incident cases of preeclampsia.
- In the US, in 2025, preeclampsia is estimated to affect nearly 170,000 individuals with mild cases and 55,000 with severe cases, reflecting the considerable burden and severity-specific distribution of this hypertensive pregnancy disorder.
Preeclampsia Epidemiology Segmentation
- Total Incident Cases of Preeclampsia in the 7MM
- Treated Cases of Preeclampsia in the 7MM
- Total Diagnosed Incident Cases of Preeclampsia in the 7MM
- Severity-specific Incident Cases of Preeclampsia in the 7MM
- Age-specific Incident Cases of Preeclampsia in the 7MM
- Subtype-specific Incident Cases of Preeclampsia in the 7MM
Preeclampsia Market Recent Developments and Breakthroughs
- In December 2025,DiaMedica Therapeutics announced significant progress in its DM199 program for treating preeclampsia. Following a successful pre-IND meeting, the FDA requested an additional non-clinical study, with results expected by Q2 2026. Simultaneously, an ongoing Phase 2 investigator-sponsored trial in South Africa has demonstrated promising early safety and efficacy. DM199, a recombinant human tissue kallikrein-1, aims to improve blood flow and reduce blood pressure without crossing the placental barrier. These developments bolster the potential of DM199 as a novel therapeutic for a condition where delivery remains the only current treatment.
Preeclampsia Drug Analysis
The section dedicated to Preeclampsia drugs report provides an in-depth evaluation of pipeline drugs related to Preeclampsia. The drug chapters section provides valuable information on various aspects related to clinical trials of Preeclampsia, such as the pharmacological mechanisms of the drugs involved, designations, approval status, patent information, and a comprehensive analysis of the pros and cons associated with each drug. Furthermore, it presents the most recent news updates and press releases on drugs targeting Preeclampsia.
Preeclampsia Market Outlook
Currently, there are no effective pharmacological treatments or preventive strategies for preeclampsia. Available therapies primarily focus on controlling hypertension, a secondary consequence of placental dysfunction, rather than addressing the underlying pathophysiology. The optimal management strategy hinges on gestational age and disease severity, balancing maternal safety with fetal viability. As delivery remains the only definitive cure, clinicians must carefully time interventions to minimize risks while ensuring fetal maturation.
Aspirin is the most widely used preventive measure, with evidence suggesting that early initiation before 16 weeks of gestation may mitigate the risk of preeclampsia. However, its efficacy is significantly diminished when started later, underscoring the importance of early risk stratification and intervention.
- A few key players are leading the Preeclampsia treatment landscape, such as Comanche Biopharma, and others. The details of the country-wise and therapy-wise market size have been provided below.
- In the total market size of Preeclampsia in the 7MM, the United States accounted for the highest market share, i.e. approximately USD 70 million in 2024.
- In 2024, Germany and France generated the highest revenue among the EU4 and the UK, reaching nearly USD 4 million.
- There is no approved therapies for preeclampsia, delivery is the only cure and management majorly relies on antihypertensive agents, anticonvulsants, and corticosteroids.
- Antihypertensive therapy remains the cornerstone of preeclampsia management. In 2024, antihypertensive agents accounted for ~USD 50 million of the US market valuation.
Preeclampsia Competitive Landscape
The preeclampsia market features a mix of established diagnostic and pharmaceutical companies alongside emerging biotech innovators, all striving to enhance early detection and treatment of this pregnancy-related condition. In the diagnostics segment, major players such as Roche, Thermo Fisher Scientific, Revvity (PerkinElmer), Sera Prognostics, Siemens Healthineers, Bayer AG and Quidel Corp dominate, offering biomarker-based blood tests and screening tools that help assess risk and stratification in prenatal care. These firms focus on regulatory approvals, performance consistency, and expanding global reach to maintain competitive advantage.
In the therapeutics and drugs market, traditional pharmaceutical giants like Bayer AG, Pfizer Inc., Novartis AG, Merck & Co., and GlaxoSmithKline are key competitors, leveraging extensive R&D and broad portfolios of antihypertensives and supportive therapies. Meanwhile, biotech companies such as Comanche Biopharma, Gmax Biopharm, and Vicore Pharma are advancing novel investigational therapies including siRNA and molecular approaches that target underlying mechanisms of preeclampsia. Strategic collaborations, pipeline advancements, and innovative diagnostic-therapeutic combinations characterize the competitive dynamics, with companies seeking differentiation through product innovation and expanded clinical utility.
Key Preeclampsia Companies
The Key Preeclampsia companies actively involved in the Preeclampsia treatment landscape include -
- Diabetomics, Inc.
- Metabolomic Diagnostics Ltd.
- Sera Prognostics
- Thermo Fisher Scientific Inc.
- Siemens Healthineers AG
- Bayer AG, and others
Preeclampsia Drugs Uptake
This section focuses on the uptake rate of potential Preeclampsia drugs expected to be launched in the market during 2022–2036, which depends on the competitive landscape, safety, efficacy data along with order of entry. It is important to understand that the key players evaluating their novel therapies in the pivotal and confirmatory trials should remain vigilant when selecting appropriate comparators to stand the greatest chance of a positive opinion from regulatory bodies, leading to approval, smooth launch, and rapid uptake.
Preeclampsia Clinical Trials Analysis
The Preeclampsia pipeline report provides insights into Preeclampsia clinical trials within the Phase III and Phase II stages. It also analyzes key players involved in developing targeted therapeutics.
Preeclampsia Pipeline Development Activities
The Preeclampsia clinical trial analysis report covers information on collaborations, acquisitions and mergers, licensing, and patent details for Preeclampsia emerging therapies.
Latest KOL Views on Preeclampsia Market Report
To stay abreast of the latest trends in the market, we conduct primary research by seeking the opinions of Key Opinion Leaders (KOLs) and Subject Matter Experts (SMEs) who work in the relevant field. This helps us fill any gaps in data and validate our secondary research. We have reached out to industry experts to gather insights on various aspects of Preeclampsia, including the evolving treatment landscape, patients’ reliance on conventional therapies, their acceptance of therapy switching, drug uptake, and challenges related to accessibility. The experts we contacted included medical/scientific writers, professors, and researchers from prestigious universities in the US, Europe, the UK, and Japan.
Our team of analysts at Delveinsight connected with more than 10 KOLs across the 7MM. We contacted institutions such as the National Institute of Child Health and Human Development, St. Joseph's Health Care, University of Milan, Nagoya City University, etc., among others. By obtaining the opinions of these experts, we gained a better understanding of the current and emerging treatment patterns in the Preeclampsia market, which will assist our clients in analyzing the overall epidemiology and market scenario.
Preeclampsia Report Qualitative Analysis
We perform Qualitative and Market Intelligence analysis using various approaches, such as SWOT analysis and Conjoint Analysis. In the SWOT analysis, strengths, weaknesses, opportunities, and threats in terms of disease diagnosis, patient awareness, patient burden, competitive landscape, cost-effectiveness, and geographical accessibility of therapies are provided. These pointers are based on the Analyst’s discretion and assessment of the patient burden, cost analysis, and existing and evolving treatment landscape.
Conjoint Analysis analyzes multiple approved and emerging therapies based on relevant attributes such as safety, efficacy, frequency of administration, designation, route of administration, and order of entry. Scoring is given based on these parameters to analyze the effectiveness of therapy. Further, the therapies’ safety is evaluated wherein the acceptability, tolerability, and adverse events are majorly observed, and it sets a clear understanding of the side effects posed by the drug in the trials. In addition, the scoring is also based on the route of administration, order of entry and designation, probability of success, and the addressable patient pool for each therapy. According to these parameters, the final weightage score and the ranking of the emerging therapies are decided.
Preeclampsia Market Access and Reimbursement
Because newly authorized drugs are often expensive, some patients escape receiving proper treatment or use off-label, less expensive prescriptions. Reimbursement plays a critical role in how innovative treatments can enter the market. The cost of the medicine, compared to the benefit it provides to patients who are being treated, sometimes determines whether or not it will be reimbursed. Regulatory status, target population size, the setting of treatment, unmet needs, the number of incremental benefit claims, and prices can all affect market access and reimbursement possibilities.
The report further provides detailed insights on the country-wise accessibility and reimbursement scenarios, cost-effectiveness scenario of approved therapies, programs making accessibility easier and out-of-pocket costs more affordable, insights on patients insured under federal or state government prescription drug programs, etc.
Scope of the Preeclampsia Market Report
- The Preeclampsia treatment market report covers the descriptive overview of Preeclampsia, explaining its causes, signs and symptoms, pathophysiology, diagnosis and currently available Preeclampsia therapies
- Comprehensive insight has been provided into the Preeclampsia epidemiology and treatment in the 7MM
- Additionally, an all-inclusive account of both the current and emerging therapies for Preeclampsia is provided, along with the assessment of new therapies, which will have an impact on the current treatment landscape
- A detailed review of the Preeclampsia treatment market; historical and forecasted is included in the report, covering drug outreach in the 7MM
- The Preeclampsia treatment market report provides an edge while developing business strategies, by understanding trends shaping and driving the Preeclampsia market
Preeclampsia Market Report Highlights
- In the coming years, the Preeclampsia market is set to change due to the rising awareness of the disease, and incremental healthcare spending across the world; which would expand the size of the market to enable the Preeclampsia drug manufacturers to penetrate more into the Preeclampsia market
- The Preeclampsia companies and academics are working to assess challenges and seek opportunities that could influence Preeclampsia R&D. The therapies under development are focused on novel approaches to treat/improve the disease condition
- Major Preeclampsia companies are involved in developing therapies for Preeclampsia. The launch of emerging therapies will significantly impact the Preeclampsia market
- A better understanding of disease pathogenesis will also contribute to the development of novel therapeutics for Preeclampsia
- Our in-depth analysis of the pipeline assets across different stages of development (Phase III and Phase II), different emerging trends and comparative analysis of pipeline products with detailed clinical profiles, key cross-competition, launch date along with product development activities will support the clients in the decision-making process regarding their therapeutic portfolio by identifying the overall scenario of the research and development activities
Preeclampsia Market Report Insights
- Preeclampsia Patient Population
- Preeclampsia Therapeutic Approaches
- Preeclampsia Market Size and Trends
- Existing Market Opportunity
- Preeclampsia Drug Market
- Preeclampsia Emerging Drugs
Preeclampsia Market Report Key Strengths
- 11-year Forecast
- The 7MM Coverage
- Preeclampsia Epidemiology Segmentation
- Key Cross Competition
- Preeclampsia Drugs Uptake
Preeclampsia Market Report Assessment
- Current Preeclampsia Treatment Practices
- Preeclampsia Market Reimbursements
- Preeclampsia Market Attractiveness
- Qualitative Analysis (SWOT, Conjoint Analysis, Unmet needs)
- Preeclampsia Market Drivers
- Preeclampsia Market Barriers
Key Questions Answered In The Preeclampsia Market Forecast Report:
Preeclampsia Market Insights:
- What was the Preeclampsia drug class share (%) distribution in 2024 and how it would look like in 2036?
- What would be the Preeclampsia market size as well as market size by therapies across the 7MM during the forecast period (2024-2036)?
- What are the key findings pertaining to the market across 7MM and which country will have the largest Preeclampsia market size during the forecast period (2024-2036)?
- At what CAGR, the Preeclampsia market is expected to grow by 7MM during the forecast period (2024-2036)?
- What would be the Preeclampsia market outlook across the 7MM during the forecast period (2024-2036)?
- What would be the Preeclampsia market growth till 2036, and what will be the resultant market Size in the year 2036?
- How would the unmet needs affect the Preeclampsia market dynamics and subsequent analysis of the associated trends?
Preeclampsia Epidemiology Insights:
- What are the disease risk, burden, and regional/ethnic differences of the Preeclampsia?
- What are the key factors driving the epidemiology trend for seven major markets covering the United States, EU5 (Germany, Spain, France, Italy, UK), and Japan?
- What is the historical Preeclampsia patient pool in seven major markets covering the United States, EU5 (Germany, Spain, France, Italy, UK), and Japan?
- What would be the forecasted patient pool of Preeclampsia in seven major markets covering the United States, EU5 (Germany, Spain, France, Italy, UK), and Japan?
- Where will be the growth opportunities in the 7MM with respect to the patient population pertaining to Preeclampsia?
- Out of all 7MM countries, which country would have the highest prevalent population of Preeclampsia during the forecast period (2024-2036)?
- At what CAGR the patient population is expected to grow in 7MM during the forecast period (2024-2036)?
Preeclampsia Current Treatment Scenario, Marketed Drugs and Emerging Therapies
- What are the current options for the Preeclampsia treatment in addition to the approved therapies?
- What are the current treatment guidelines for the treatment of Preeclampsia in the USA, Europe, and Japan?
- What are the Preeclampsia marketed drugs and their respective MOA, regulatory milestones, product development activities, advantages, disadvantages, safety and efficacy, etc.?
- How many Preeclampsia companies are developing therapies for the treatment of Preeclampsia?
- How many Preeclampsia therapies are in-development by each company for Preeclampsia treatment?
- How many are emerging Preeclampsia therapies in mid-stage, and late stage of development for Preeclampsia treatment?
- What are the key collaborations (Industry - Industry, Industry - Academia), Mergers and acquisitions, licensing activities related to the Preeclampsia therapies?
- What are the recent novel therapies, targets, mechanisms of action and technologies being developed to overcome the limitation of existing therapies?
- What are the clinical studies going on for Preeclampsia and their status?
- What are the current challenges faced in Preeclampsia drug development?
- What are the key designations that have been granted for the emerging therapies for Preeclampsia?
- What are the global historical and forecasted Preeclampsia treatment market?
Reasons to buy Preeclampsia Market Report
- The Preeclampsia treatment market report will help in developing business strategies by understanding trends shaping and driving the Preeclampsia market
- To understand the future market competition in the Preeclampsia market and Insightful review of the key market drivers and barriers
- Organize sales and marketing efforts by identifying the best opportunities for Preeclampsia in the US, Europe (Germany, Spain, Italy, France, and the United Kingdom) and Japan
- Identification of strong upcoming Preeclampsia companies in the Preeclampsia drugs market will help in devising strategies that will help in getting ahead of competitors
- Organize sales and marketing efforts by identifying the best opportunities for Preeclampsia market
- To understand the future market competition in the Preeclampsia market