PH-ILD Market Summary
Pulmonary Hypertension Associated with Interstitial Lung Disease (PH-ILD) Insights and Trends
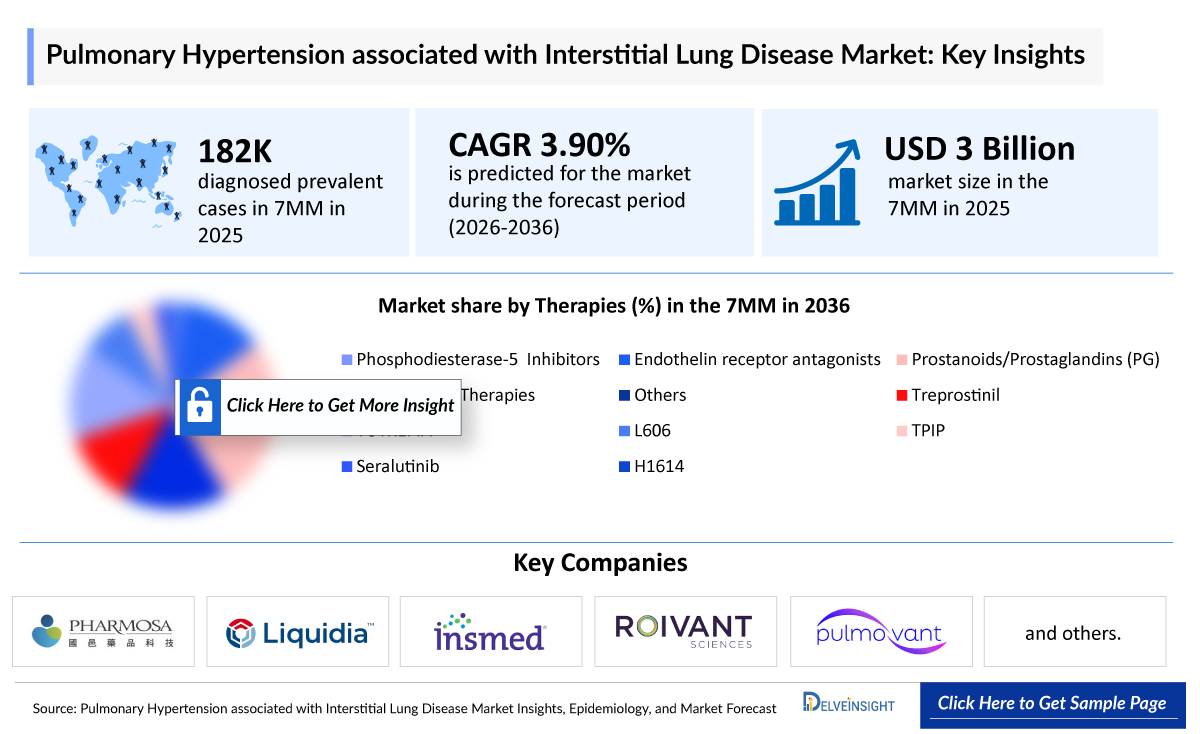
- According to DelveInsight’s analysis, the PH-ILD market across the 7MM was valued at approximately USD 3 billion in 2025 and is projected to expand at a Compounded Annual Growth Rate (CAGR) of 3.9% through 2036, driven by the anticipated entry of advanced therapies and improved clinical management.
- In 2025, the United States accounted for approximately 83,000 diagnosed prevalent cases of PH-ILD, representing the largest patient base in the 7MM, while EU4 and the UK contributed nearly 70,000 cases, driving significant demand for specialized therapeutic options.
- The current standard of care includes approved inhaled therapies such as treprostinil (YUTREPIA) and treprostinil (TYVASO/TYVASO DPI/ TREPROST), which focus on symptom management and improving the quality of life (QoL) for patients with severe pulmonary vascular complications.
- The PH-ILD pipeline is transitioning toward next-generation inhaled and oral modalities, with late-stage assets such as treprostinil liposomal (L606) and treprostinil palmitil inhalation powder (TPIP), alongside emerging candidates like mosliciguat, seralutinib, and H1614 (HB-1614), expected to reshape the treatment landscape and drive market expansion throughout the forecast period.
- Research in PH-ILD continues to evolve, emphasizing the need for continuous clinical trials to address high morbidity and mortality rates, ensuring better diagnostic precision and updated treatment guidelines for diverse patient populations.
- Significant treatment gaps in PH-ILD persist due to delayed diagnosis, as overlapping symptoms with Interstitial Lung Disease (ILD) often lead to misdiagnosis. The reliance on invasive procedures like Right Heart Catheterization further limits early detection, underscoring an urgent unmet need for non-invasive biomarkers and advanced imaging to improve patient prognosis and timely intervention.
- A key market driver PH-ILD is the increasing recognition of the disease and improvement in diagnostic practices, enabling earlier identification and treatment initiation, which is expected to expand the treatable patient pool and drive demand for both existing therapies and emerging pipeline treatments.
Pulmonary Hypertension Associated with Interstitial Lung Disease (PH-ILD) Market size and forecast
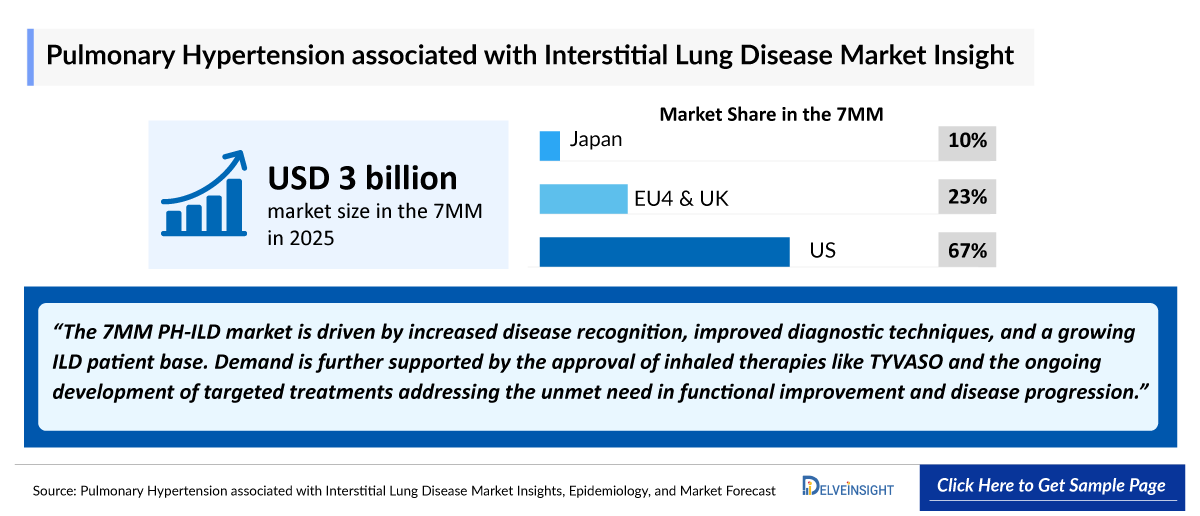
- 2025 PH-ILD Market Size in the 7MM: USD 3 billion
- PH-ILD Growth Rate (2026–2036) in the 7MM: 3.9% CAGR
DelveInsight's ‘Pulmonary Hypertension Associated with Interstitial Lung Disease (PH-ILD) – Market Insights, Epidemiology and Market Forecast – 2036’ report delivers an in-depth understanding of PH-ILD I, historical and forecasted epidemiology, as well as the PH-ILD market trends in the United States, EU4 (Germany, Spain, Italy, and France), the United Kingdom, and Japan.
The PH-ILD market report delivers a comprehensive analysis of the current treatment landscape, including standards of care, clinical practices, and evolving therapeutic algorithms. It evaluates, PH-ILD patient burden trends, revenue & market share dynamics, peak patient share & therapy uptake analysis, and provides an in-depth market size assessment, and growth rate projections (Historical & Forecast 2022–2036) across the 7MM regions. The report highlights key unmet medical needs in PH-ILD and maps the competitive and clinical landscape to uncover high‑value opportunities, providing a clear outlook on future market growth potential.
Scope of the Pulmonary Hypertension Associated with Interstitial Lung Disease (PH-ILD) Market Insights Report | |
|
Study Period |
2022–2036 |
|
Historical Year |
2022–2025 |
|
Forecast Period |
2026–2036 |
|
Base Year |
2025 |
|
Geographies Covered |
|
|
Pulmonary Hypertension Associated with Interstitial Lung Disease (PH-ILD) Market CAGR (Forecast period) |
3.9% (2026-2036) |
|
Pulmonary Hypertension Associated with Interstitial Lung Disease (PH-ILD) Epidemiology Segmentation Analysis |
Patient Burden Assessment
|
|
Pulmonary Hypertension Associated with Interstitial Lung Disease (PH-ILD) Companies |
|
|
Pulmonary Hypertension Associated with Interstitial Lung Disease (PH-ILD) Therapies |
|
|
Pulmonary Hypertension Associated with Interstitial Lung Disease (PH-ILD) Market |
Segmented by
|
|
Analysis |
|
Pulmonary Hypertension Associated with Interstitial Lung Disease (PH-ILD) Understanding and Treatment Algorithm
Pulmonary Hypertension Associated with Interstitial Lung Disease (PH-ILD) Overview and Diagnosis
PH-ILD refers to a wide range of lung conditions, many of which involve scarring or inflammation in the thin layer of tissue between the air sacs and nearby blood vessels. This delicate region, known as the interstitium, is just micrometers thick but plays a vital role by maintaining the structure of the alveoli and enabling smooth oxygen transfer into the bloodstream. Several factors can increase the risk of developing PH-ILD. Chronic smoking and long-term exposure to environmental or occupational toxins can damage the lung tissue, contributing to inflammation and fibrosis that elevate pulmonary artery pressure.
Diagnosing patients with PH-ILD requires a combination of noninvasive and invasive methods. If a patient with ILD shows unexplained worsening of breathlessness or has symptoms that seem disproportionate to the extent of their lung disease, further evaluation for PH is recommended. Since ILD and PH share many overlapping signs, distinguishing between the two can be challenging. To aid diagnosis, healthcare providers often rely on noninvasive tools like pulmonary function tests, the 6MWD, echocardiography, and imaging such as chest X-rays or CT scans. However, for a definitive diagnosis of PH-ILD, RHC remains the most accurate and conclusive method.
Further details are provided in the report.
Pulmonary Hypertension Associated with Interstitial Lung Disease (PH-ILD) Treatment Landscape
The treatment of PH-ILD primarily focuses on managing the underlying lung disease, yet the scarcity of specifically approved therapies presents a significant challenge in standardizing care. Currently, the landscape is anchored by approved inhaled therapies such as treprostinil (YUTREPIA) and treprostinil (TYVASO/TYVASO DPI/ TREPROST), which focus on symptom management and improving quality of life. Despite these options, many clinicians still report prescribing Pulmonary Arterial Hypertension-approved medications off-label for nearly half of their PH-ILD patients due to persistent therapeutic gaps. However, the paradigm is shifting as late-stage pipeline assets like treprostinil liposomal (L606) and treprostinil palmitil inhalation powder (TPIP).
Further details related to country-based variations are provided in the report.
Pulmonary Hypertension Associated with Interstitial Lung Disease (PH-ILD) Unmet Needs
The section “unmet needs of PH-ILD” outlines the critical gaps between the current state of patient care, diagnosis, and the ideal & effective management of the disease. It highlights the obstacles experienced by patients, clinicians, and researchers and identifies potential solutions for future progress.
- Diagnostic challenges and risk of misdiagnosis
- Need of noninvasive biomarker development
- The critical need for diversified therapies in PH-ILD
- Expanding treprostinil routes to ease the daily burden
- Lack of standard diagnostic pathways leaves PH-ILD patients at risk
- and others…
Note: Comprehensive unmet needs insights in PH-ILD and their strategic implications are provided in the full report.
Pulmonary Hypertension Associated with Interstitial Lung Disease (PH-ILD) Epidemiology
Key Findings from Pulmonary Hypertension Associated with Interstitial Lung Disease (PH-ILD) Epidemiological Analysis and Forecast
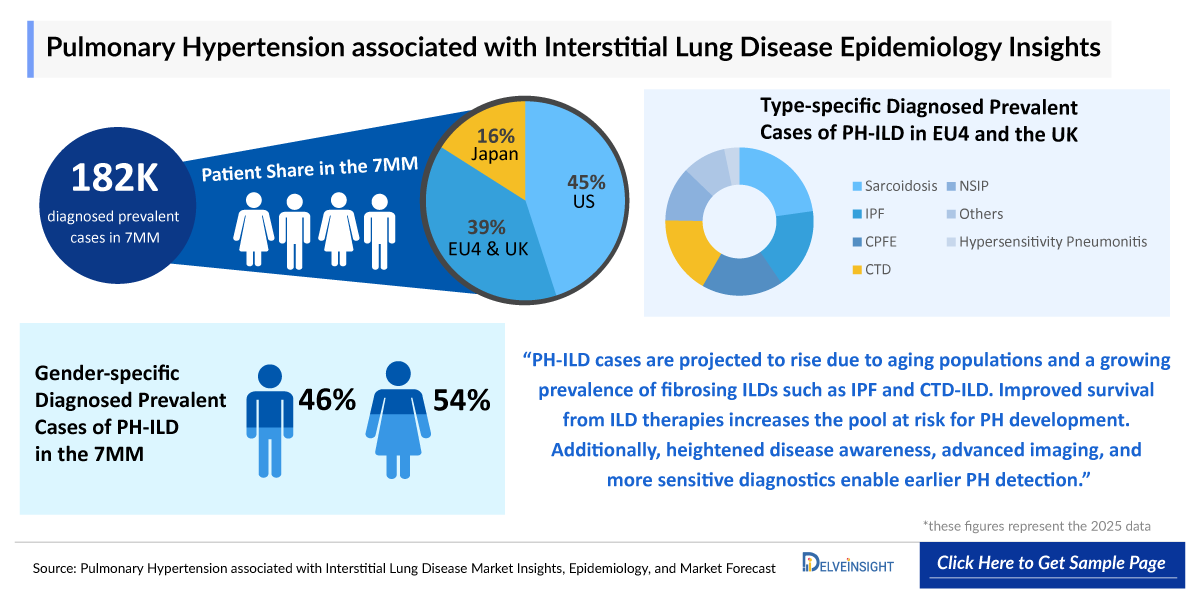
- As of 2025, the total diagnosed prevalent population of PH-ILD across the 7MM is estimated at nearly 182,000, reflecting the significant burden of the disease.
- Beyond PH-ILD, the broader diagnosed prevalent population of ILD in the 7MM was significantly higher, reaching approximately more than 1 million cases in 2025, underscoring the large pool of patients at risk of developing associated pulmonary hypertension.
- In the year 2025, the United States represented the largest share of the PH-ILD market, with nearly 83,000 diagnosed prevalent cases, supported by advanced diagnostic infrastructure and higher clinical awareness.
- In 2025, EU4 and the UK together contributed approximately 70,000 cases of PH-ILD with the United Kingdom representing the largest individual European segment with nearly 18,000 of PH-ILD diagnosed cases.
- Japan accounted for approximately 29,000 diagnosed prevalent cases of PH-ILD in 2025, representing a stable and clinically significant patient base in the Asian market.
- Gender-specific analysis in the United States for 2025 shows a slightly higher prevalence in males, with approximately 43,000 cases, compared to nearly 40,000 cases in females.
Pulmonary Hypertension Associated with Interstitial Lung Disease (PH-ILD) Drug Analysis & Competitive Landscape
The PH-ILD drug chapter provides a detailed, market-focused review of approved therapies and the emerging pipeline across mid and late Phase clinical trials. It covers mechanism of action, clinical trial data, regulatory approvals, patents, collaborations, strategic partnerships upcoming Key catalyst for each therapy, along with their advantages, limitations, and recent developments. This section offers critical insights into the PH-ILD treatment landscape, supporting market assessment, competitive analysis, and growth forecasting for the PH-ILD therapeutics market.
Approved Therapies for Pulmonary Hypertension Associated with Interstitial Lung Disease (PH-ILD)
Treprostinil (YUTREPIA): Liquidia Corporation
YUTREPIA is a dry powder inhalation formulation of treprostinil sodium, a prostacyclin mimetic designed for the treatment of PAH. Delivered through a single-use, handheld inhaler, each capsule contains treprostinil sodium blended with carefully selected excipients L-leucine, polysorbate 80, sodium chloride, sodium citrate, and trehalose.
- In May 2025, YUTREPIA (treprostinil) inhalation powder received full FDA approval for improving exercise capacity in adults with PAH and PH-ILD. As the first dry powder prostacyclin therapy enabled by Liquidia’s PRINT technology, YUTREPIA delivers uniform particles to the deep lung via a low-resistance, breath-actuated device, offering enhanced usability and potential adherence benefits.
- In August 2024, Liquidia received tentative FDA approval to market YUTREPIA, its inhaled dry powder formulation of treprostinil, for the treatment of PAH and PH-ILD. This approval allows commercialization once the existing regulatory exclusivity expires in May 2025. The New Drug Application (NDA) for YUTREPIA was submitted via the 505(b) (2) pathway, using nebulized TYVASO as the reference listed drug.
Note: Detailed marketed therapies assessment will be provided in the final report.
Pulmonary Hypertension Associated with Interstitial Lung Disease (PH-ILD) Marketed/Approved Therapies | ||||||
|
Drug/Therapy |
Company |
Indication |
Molecule Type |
MoA |
RoA |
Marketed Region |
|
Treprostinil (YUTREPIA) |
Liquidia Corporation |
PH-ILD |
Small molecule |
Prostacyclin agonist |
Oral inhalation |
US: 2025 |
|
Treprostinil (TYVASO/TYVASO DPI/ TREPROST) |
United Therapeutics |
PH-ILD |
Small molecule |
Prostacyclin agonist |
Inhalation |
US: 2022, JP: 2024 |
|
Note: To be continued in the final report | ||||||
Pulmonary Hypertension Associated with Interstitial Lung Disease (PH-ILD) Pipeline Analysis
Treprostinil liposomal (L606): Pharmosa BioPharm/Liquidia Corporation
L606 is a liposomal formulation of treprostinil, with liposome particle sizes ranging from 110–140 nm, suspended in a sodium citrate–bicarbonate buffer. This advanced formulation is engineered to enhance drug stability and prolong systemic circulation by protecting treprostinil from rapid degradation and clearance. The liposomal encapsulation enables a sustained pulmonary release, allowing for more consistent drug exposure over time, while minimizing peaks and troughs that may lead to side effects.
- In January 2025, at the 43rd J.P. Morgan Healthcare Conference, Liquidia Corporation presented advancements in treprostinil (L606), highlighting its twice-daily dosing, improved tolerability, and rapid delivery via a next-generation breath-actuated handheld nebulizer with smart technology. The company also outlined plans to expand its clinical potential by exploring additional indications such as Idiopathic Pulmonary Fibrosis (IPF), Pulmonary Hypertension associated with Chronic Obstructive Pulmonary Disease (PH-COPD), and Systemic Sclerosis-related Raynaud’s phenomenon (SSc-RP), while also targeting inadequate responders in Pulmonary Arterial Hypertension (PAH) and Pulmonary Hypertension associated with Interstitial Lung Disease (PH-ILD).
Note: Detailed emerging therapies assessment will be provided in the final report.
Competitive Landscape of Pipeline Drugs | ||||||
|
Drug Name |
Company |
Highest Phase |
Indication |
RoA |
MoA |
Anticipated Launch in the US |
|
Treprostinil liposomal (L606) |
Pharmosa BioPharm/ Liquidia Corporation |
III |
PH-ILD |
Inhalation |
Prostacyclin agonist |
2030 |
|
Treprostinil Palmitil Inhalation Powder (TPIP) |
Insmed |
IIII |
PH-ILD |
Oral Inhalation |
Prostacyclin agonist |
Information is available in the full report |
|
Mosliciguat |
Roivant Sciences/ Pulmovant |
II |
PH-ILD |
Inhalation |
Guanylate cyclase stimulants |
Information is available in the full report |
|
H1614 (HB-1614) |
Halo Biosciences |
II |
PH-ILD |
Oral |
Hyaluronan-synthesis inhibitors |
Information is available in the full report |
|
Note: Launch insights are provisional and may change with future report updates or the occurrence of major key catalysts. To be continued in the final report | ||||||
Pulmonary Hypertension Associated with Interstitial Lung Disease (PH-ILD), Market Leaders and Emerging Companies
- Liquidia Corporation
- United Therapeutics
- Pharmosa BioPharm
- Halo Biosciences
- Roivant Sciences/Pulmovant
- Insmed and others
Pulmonary Hypertension Associated with Interstitial Lung Disease (PH-ILD) Drug Updates
- In May 2025, Gossamer Bio reported the completion of enrollment for the ongoing global Phase III PROSERA study, which is evaluating seralutinib in patients with Functional Class II and III PAH.
- In June 2025, Halo Biosciences reported the publication of results from its Phase IIa SATURN study in Thorax, marking a pivotal milestone in the development of HB-1614 for PH-ILD.
- In June 2025, Insmed reported positive topline results from its Phase IIb study of TPIP as an once‑daily treatment for PAH.
- In September 2024, TREPROST (treprostinil) inhalation solution received approval in Japan for a new indication targeting adults at risk of disease progression, marking a significant regulatory milestone in expanding access to treprostinil therapy for patients in the Japanese markets.
Pulmonary Hypertension Associated with Interstitial Lung Disease (PH-ILD) Market Outlook
The market outlook for PH-ILD is evolving, driven by high unmet clinical need, increasing disease recognition, and the progressive nature of cardiopulmonary impairment associated with the condition. While no curative therapies currently exist, growing adoption of supportive care, oxygen therapy, and targeted pulmonary vasodilators, along with advancements in understanding disease pathophysiology, are expected to support market growth and improve long-term patient management outcomes.
Key marketed therapies shaping current management
- Treprostinil (YUTREPIA) - Liquidia Corporation: YUTREPIA is an inhaled dry powder formulation of treprostinil sodium developed for the management of Pulmonary Arterial Hypertension. It is administered using a disposable handheld inhaler, where each dose capsule combines treprostinil with functional excipients including L-leucine, polysorbate 80, sodium chloride, sodium citrate, and trehalose to ensure efficient pulmonary delivery and formulation stability.
- Treprostinil (TYVASO/TYVASO DPI/ TREPROST) - United Therapeutics: Treprostinil (TYVASO) inhalation solution and treprostinil (TYVASO DPI) inhalation powder are prescription medicines used in adults to treat pulmonary arterial hypertension (PAH) (PAH; Group 1), a condition involving high blood pressure in the arteries of the lungs. They are also used to treat PH-ILD disease (PH-ILD; Group 3), which involves elevated pressure in the lungs due to inflammation.
- And more
Overall, in PH-ILD, market growth across the 7MM from 2022–2036 is expected to be driven by advancements in diagnostic approaches, including noninvasive tools and confirmatory methods like Right Heart Catheterization, along with increasing disease awareness and evolving treatment options, creating strong commercial potential for both existing therapies and emerging pipeline candidates.
- Among the 7MM, the United States accounted for the largest market size of PH-ILD, valued at approximately USD 2,500 million in 2025.
- EU4 and the UK combined represented a significant market segment, with a total market size of approximately USD 500 million in 2025, driven by the increasing adoption of specialized inhaled therapies.
- Japan accounted for a market size of approximately USD 300 million in 2025, representing a stable and growing portion of the total 7MM market.
- The most meaningful recent shift in the treatment landscape has been the focus on addressing the limitations of traditional vasodilators and off-label PAH medications. Currently, the landscape is anchored by approved inhaled therapies like treprostinil (TYVASO/TYVASO DPI/ TREPROST) which generated over ~USD 1,100 million in the 7MM in 2025 and the emergence of treprostinil (YUTREPIA).
- The transition toward next-generation therapies, such as treprostinil liposomal (L606) and treprostinil palmitil (TPIP), represents a significant leap in precision delivery. These advanced modalities aim to optimize drug concentration in the lungs and provide more comprehensive disease control, significantly improving the long-term quality of life and functional outcomes for patients with severe PH-ILD phenotypes.
Further details will be provided in the report….
Drug Class/Insights into Leading Emerging and Marketed Therapies in Pulmonary Hypertension Associated with Interstitial Lung Disease (PH-ILD) (2022–2036 Forecast)
The current treatment landscape for PH-ILD is structured across several therapeutic categories, reflecting disease severity, underlying ILD subtype, and patient-specific response. Supplemental oxygen therapy and pulmonary rehabilitation remain foundational options for managing chronic hypoxemia and improving exercise capacity. For patients with more advanced disease, the treatment paradigm is evolving to include targeted pulmonary vasodilators such as inhaled prostacyclins or PDE5 inhibitors which aim to reduce pulmonary vascular resistance (PVR) and alleviate right heart strain.
- Inhaled Therapy: Products such as TYVASO/ TREPROST/TYVASO DPI and YUTREPIA provide patients with targeted inhaled delivery options that help reduce PVR and improve exercise capacity. These therapies, developed by United Therapeutics and Liquidia Corporation, have played a central role in addressing this significant unmet need. inhalation solution (TYVASO ) and dry powder inhaler (TYVASO DPI) are the first inhaled treprostinil therapies approved specifically for PH-ILD, addressing an important unmet need for patients with IIP, IPF, CPFE, and WHO Group 3 connective tissue diseases.
- Small Molecule: YUTREPIA is Liquidia Corporation’s DPI formulation of treprostinil, approved for PH-ILD in the US. Targeting patients with idiopathic interstitial pneumonia (IIP), idiopathic pulmonary fibrosis (IPF), combined pulmonary fibrosis and emphysema (CPFE), and WHO Group 3 connective tissue diseases, YUTREPIA offers an alternative to TYVASO DPI with potentially improved delivery and patient experience.
Pulmonary Hypertension Associated with Interstitial Lung Disease (PH-ILD) Drug Uptake
This section focuses on the uptake rate of potential drugs expected to be launched in the market during the forecast period (2026–2036). The analysis covers the PH-ILD drug’s uptake, performance at peak, factors affecting performance during prime years of growth, patient uptake by therapy, and anticipated sales generated by each drug.
The emergence of next-generation inhaled therapies is reshaping the treatment landscape of PH-ILD, with a focus on improving drug delivery, efficacy, and patient convenience, treprostinil (YUTREPIA), developed by Liquidia Corporation, is an advanced dry powder inhalation formulation of treprostinil sodium, a prostacyclin mimetic, designed for efficient pulmonary delivery via a handheld device. Engineered with optimized excipients, it enables precise drug deposition in the lungs, enhancing therapeutic outcomes, and is expected to witness a medium-to-fast uptake, reflecting its potential to significantly improve patient-friendly disease management.
Detailed insights of emerging therapies' drug uptake is included in the report.
Market Access and Reimbursement of Approved therapies in Pulmonary Hypertension Associated with Interstitial Lung Disease (PH-ILD)
The report further provides detailed insights on the country-wise accessibility and reimbursement scenarios, cost-effectiveness scenario of approved therapies, programs making accessibility easier and out-of-pocket costs more affordable, insights on patients insured under federal or state government prescription drug programs, etc.
United States
|
United States Reimbursement of Therapies Approved for PH-ILD | |
|
Drug/Therapy |
Program |
|
Treprostinil (YUTREPIA) |
Voucher Program |
Reimbursement is a crucial factor that affects the drug’s access to the market. Often, the decision to reimburse comes down to the price of the drug relative to the benefit it produces in treated patients. To reduce the healthcare burden of these high-cost therapies, many payment models are being considered by payers and other industry insiders.
NOTE: Further Details are provided in the final report…
Pulmonary Hypertension Associated with Interstitial Lung Disease (PH-ILD) Price Scenario & Trends
Pricing and analogue assessment of PH-ILD therapies highlights evolving price dynamics structures. This section summarizes the cost of approved treatments, closest and most appropriate analogue selection for emerging therapies, and understanding of how pricing influences market access, adherence, and long-term uptake.
- Pricing of Pulmonary Hypertension Associated with Interstitial Lung Disease (PH-ILD) Approved Drugs
Treprostinil (YUTREPIA): The annual treatment cost of YUTREPIA in the United States is approximately USD 300,000, reflecting its role as an advanced inhaled therapy for managing PH-ILD associated with ILD.
Industry Experts and Physician Views for Pulmonary Hypertension Associated with Interstitial Lung Disease (PH-ILD)
To keep up with PH-ILD market trends, we take Key Opinion Leaders (KOLs) and Subject Matter Experts (SMEs) opinions working in the domain through primary research to fill the data gaps and validate our secondary research. Industry Experts were contacted for insights on the PH-ILD emerging therapies, evolving treatment landscape, patient adherence to conventional therapies, therapy switching trends, drug adoption and uptake, accessibility challenges, and epidemiology and real-world prescription patterns including MD, PhD, Instructor, Postdoctoral Researcher, Professor, Researcher, and others.
DelveInsight’s analysts engaged with 10+ key opinion leaders (KOLs) across major markets to capture country-level insights in PH-ILD Leading centers such as University of California and Royal College of Physicians, among others, were consulted to validate clinical practices, treatment patterns, and emerging therapeutic perspectives.
Their opinion helps understand and validate current and emerging PH-ILD, therapies, highlight unmet medical needs, provide epidemiological context, and support strategic decisions for Market access, therapy adoption, and pipeline prioritization in PH-ILD.
|
Region |
Key Opinion Leaders (KOLs) and Subject Matter Experts (SMEs) |
|
United States |
“PH-ILD represents a challenging clinical overlap, where two distinct diseases converge—each with its own diagnostic hurdles. Diagnosis is frequently delayed due to a combination of low clinical suspicion, overlapping symptoms that mask one another, and limited access to specialized diagnostic resources. This complexity often obscures timely identification and intervention.” |
|
Japan |
“PH-ILD frequently surface in diverse specialty settings, yet ambiguity persists regarding clear ownership of management. This highlights an important opportunity for cross-disciplinary collaboration, providing crucial perspective on how various clinicians approach, interpret, and address the same complex patient scenarios to ensure cohesive, comprehensive care.” |
Qualitative Analysis: SWOT and Attribute Analysis
We perform qualitative and market Intelligence analysis using various approaches, such as SWOT analysis and attribute analysis.
In the SWOT analysis of PH-ILD, strengths, weaknesses, opportunities, and threats in terms of disease diagnosis, patient awareness, patient burden, competitive landscape, cost-effectiveness, and geographical accessibility of therapies are provided. Attribute analysis analyzes emerging therapies based on relevant attributes such as safety, efficacy, frequency of administration, route of administration, and order of entry. Scoring is given based on these parameters to analyze the effectiveness of therapy.
The team of analysts analyzes promising emerging therapies based on relevant attributes such as safety, efficacy, frequency of administration, route of administration, and order of entry. In efficacy, the trial’s primary and secondary outcome measures are evaluated, whereas the therapies’ safety is evaluated, wherein the acceptability, tolerability, and adverse events are majorly observed. In addition, the scoring is also based on the route of administration, order of entry, probability of success, and the addressable patient pool for each therapy. According to these parameters, the final weightage score and the ranking of the emerging therapies are decided.
Scope of the Pulmonary Hypertension Associated with Interstitial Lung Disease Market Report
- The report covers a segment of key events, an executive summary, a descriptive overview of PH-ILD, explaining their causes, signs and symptoms, pathogenesis, and currently available treatments.
- Comprehensive insight has been provided into the epidemiology segments and forecasts, the future growth potential of the diagnosis rate, and disease progression along treatment guidelines.
- Additionally, an all-inclusive account of both the current and emerging treatments, along with the elaborative profiles of late-stage and prominent therapies, will have an impact on the current treatment landscape.
- A detailed review of the PH-ILD market, historical and forecasted market size, market share by therapies, detailed assumptions, and rationale behind our approach is included in the report, covering the 7MM drug outreach.
- The report provides an edge while developing business strategies by understanding trends through SWOT analysis and expert insights/KOL views, patient journey, and treatment preferences that help in shaping and driving the 7MM PH-ILD market.
Pulmonary Hypertension Associated with Interstitial Lung Disease Market Report Insights
- Pulmonary Hypertension Associated with Interstitial Lung Disease (PH-ILD) Patient Population Forecast
- Pulmonary Hypertension Associated with Interstitial Lung Disease (PH-ILD) Therapeutics Market Size
- Pulmonary Hypertension Associated with Interstitial Lung Disease (PH-ILD) Pipeline Analysis
- Pulmonary Hypertension Associated with Interstitial Lung Disease (PH-ILD) Market Size and Trends
- Pulmonary Hypertension Associated with Interstitial Lung Disease (PH-ILD) Market Opportunity (Current and forecasted)
Pulmonary Hypertension Associated with Interstitial Lung Disease Market Report Key Strengths
- Epidemiology‑Based (Epi‑based) Bottom‑up Forecasting
- Artificial Intelligence (AI) - enabled Market Research Report
- 11-year forecast
- Pulmonary Hypertension Associated with Interstitial Lung Disease (PH-ILD) Market Outlook (North America, Europe, Asia-Pacific)
- Patient Burden Trends (by geography)
- Pulmonary Hypertension Associated with Interstitial Lung Disease (PH-ILD) Treatment Addressable Market (TAM)
- Pulmonary Hypertension Associated with Interstitial Lung Disease (PH-ILD) Competitive Landscape
- Pulmonary Hypertension Associated with Interstitial Lung Disease (PH-ILD) Major Companies Insights
- Pulmonary Hypertension Associated with Interstitial Lung Disease (PH-ILD) Price trends and Analogue Assessment
- Pulmonary Hypertension Associated with Interstitial Lung Disease (PH-ILD) Therapies Drug Adoption/Uptake
- Pulmonary Hypertension Associated with Interstitial Lung Disease (PH-ILD) Therapies Peak Patient Share analysis
Report Assessment
- Pulmonary Hypertension Associated with Interstitial Lung Disease (PH-ILD) Current Treatment Practices
- Pulmonary Hypertension Associated with Interstitial Lung Disease (PH-ILD) Unmet Needs
- Pulmonary Hypertension Associated with Interstitial Lung Disease (PH-ILD) Clinical Development Analysis
- Pulmonary Hypertension Associated with Interstitial Lung Disease (PH-ILD) Emerging Drugs Product Profiles
- Pulmonary Hypertension Associated with Interstitial Lung Disease (PH-ILD) Market Attractiveness
- Pulmonary Hypertension Associated with Interstitial Lung Disease (PH-ILD) Qualitative Analysis (SWOT and Attribute analysis)
Key Questions Answered in the Pulmonary Hypertension Associated with Interstitial Lung Disease Market Report
Market Insights
- What was the PH-ILD market size, the market size by therapies, market share (%) distribution in 2025, and what would it look like by 2036? What are the contributing factors for this growth?
- What are the anticipated pricing variations among different geographies for the emerging therapies in the future?
- What can be the future treatment paradigm of PH-ILD?
- What are the disease risks, burdens, and unmet needs of PH-ILD? What will be the growth opportunities across the 7MM concerning the patient population with PH-ILD ?
- Who is the major future competitor in the market, and how will the competitors affect their market share?
- What are the current options for the treatment of PH-ILD?
- What are the current guidelines for treating PH-ILD in the US, Europe, and Japan?
Reasons to Buy Pulmonary Hypertension Associated with Interstitial Lung Disease Market Report
- The report will help in developing business strategies by understanding the latest trends and changing treatment dynamics driving the PH-ILD market.
- Bottom‑up forecasting builds from the affected population to product forecasts, delivering a robust, data‑driven approach ideal for new therapies and novel classes.
- Insights on patient burden/prevalence, evolution in diagnosis, and factors contributing to the change in the epidemiology of the disease during the forecast years.
- Understand the existing market opportunities in varying geographies and the growth potential over the coming years.
- Identifying strong upcoming players in the market will help devise strategies to help get ahead of competitors.
- Detailed analysis and ranking of class-wise potential current and emerging therapies under the attribute analysis section to provide visibility around leading classes.
- To understand KOLs’ perspectives on the accessibility, acceptability, and compliance-related challenges of existing treatment to overcome barriers in the future.
- Detailed insights on the unmet needs of the existing market so that the upcoming players can strengthen their development and launch strategy.
- This Artificial Intelligence (AI) ‑enabled report summarize and simplify complex datasets within the report into clear, actionable insights for stakeholders, investors, and healthcare providers, enabling faster, data‑driven decisions.


-pipeline.png&w=256&q=75)

