Interstitial Lung Disease Market Summary
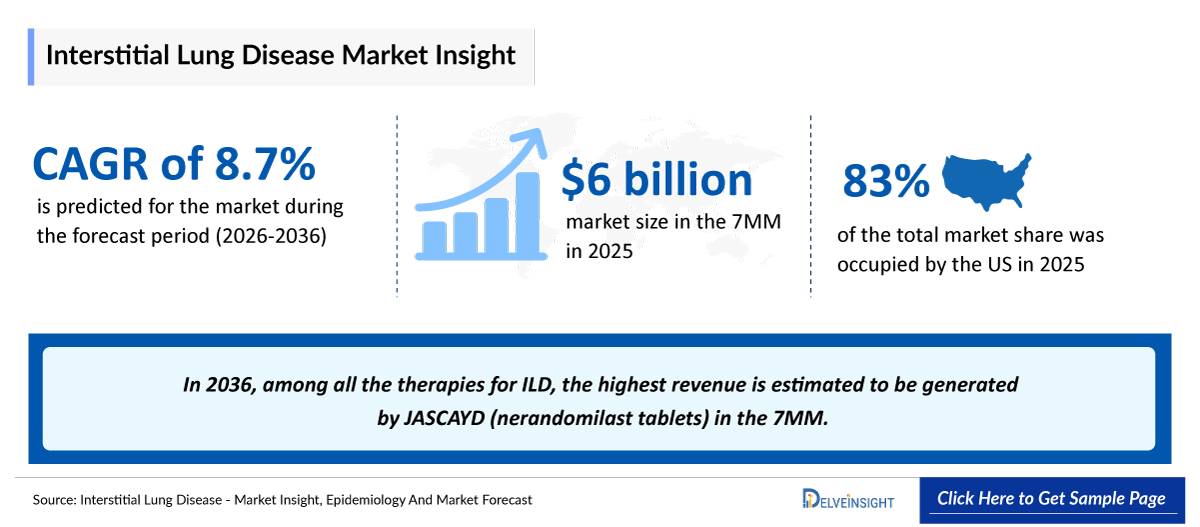
- According to DelveInsight’s analysis, the Interstitial Lung Disease Market Size was found to be more than USD 6 billion in the leading markets (the United States, the EU4 (Germany, France, Italy, and Spain), the United Kingdom, and Japan) in 2025.
- The leading Interstitial Lung Disease Companies such as United Therapeutics, Liquidia Corporation, Boehringer Ingelheim, Roche, GSK, Bristol-Myers Squibb, and others.
Interstitial Lung Disease Market Insights and Trends
- Interstitial Lung Disease reported approximately 500,000 Interstitial Lung Disease Diagnosed Prevalent Cases in the US in 2025 and this number is projected to increase by 2036.
- Limited disease-modifying options for several Interstitial Lung Disease subtypes, with current therapies largely slowing progression rather than reversing fibrosis, constrain clinical outcomes and long-term treatment effectiveness.
- Strong uptake of premium and differentiated therapies, particularly antifibrotics and pulmonary hypertension–targeted agents (e.g., OFEV, TYVASO/TYVASO DPI), is driving revenue growth beyond prevalence expansion.
- The emerging Interstitial Lung Disease Pipeline is robust and highly diversified, encompassing novel antifibrotic, immunomodulatory, and lung-targeted delivery approaches aimed at improving efficacy, tolerability, and phenotype specificity. Notable late- and mid-stage candidates include taladegib (ENV-101), admilparant (BMS-986278), and seralutinib, alongside immune-modulating agents such as belimumab (BENLYSTA) and Efzofitimod. Advanced inhaled and localized platforms—Treprostinil Palmitil Inhalation Powder (TPIP), L606, and AP01 (inhaled pirfenidone)—seek to maximize pulmonary exposure while limiting systemic toxicity, while innovative approaches such as N115 (nonsteroidal nasal spray) further expand mechanistic diversity. Interstitial Lung Disease is more prevalent in men than in women.
- Shift toward long-term disease management, with chronic therapy use, combination approaches, and extended treatment duration, is increasing per-patient lifetime value, especially in progressive fibrosing Interstitial Lung Disease market.
- Currently approved antifibrotic therapies are associated with significant gastrointestinal, hepatic, and tolerability-related adverse effects, often leading to dose reductions or discontinuation. Chronic daily oral dosing over years further compounds adherence challenges, particularly in elderly patients or those with comorbid autoimmune disease. Even inhaled prostacyclin therapies used in Interstitial Lung Disease-associated pulmonary hypertension, such as TYVASO, impose device-dependent administration burdens.
Interstitial Lung Disease Market Size and Forecast
- 2025 Interstitial Lung Disease Market Size: USD ~6 billion
- 2036 Projected Interstitial Lung Disease Market Size: USD ~13 billion
- Interstitial Lung Disease Growth Rate (2026–2036): 8.7% CAGR
Request for Unlocking the Sample Page of the "Interstitial Lung Disease Treatment Market"
Key Factors Driving the Interstitial Lung Disease Market Growth by DelveInsight
-
Rising Prevalence of Interstitial Lung Disease
The increasing incidence of pulmonary fibrosis, autoimmune-associated ILD, and occupational lung disorders is significantly driving market demand.
-
Growing Aging Population
The elderly population is more susceptible to chronic respiratory diseases, contributing to higher diagnosis and treatment rates of ILD globally.
-
Advancements in Diagnostic Technologies
Improved access to high-resolution CT scans, imaging techniques, pulmonary function tests, and biomarker-based diagnostics is enabling earlier and more accurate diagnosis.
-
Increasing Adoption of Antifibrotic Therapies
The growing use of antifibrotic drugs such as nintedanib and pirfenidone is supporting market expansion due to their ability to slow disease progression.
-
Strong Pipeline of Emerging Therapies
Several pharmaceutical and biotechnology companies are actively developing innovative therapies targeting fibrosis, inflammation, and immune dysregulation pathways.
-
Growing Awareness About Rare Pulmonary Diseases
Improved awareness among healthcare professionals and patients is leading to earlier diagnosis and better disease management.
-
Expanding Role of Precision Medicine
Personalized treatment approaches and phenotype-specific therapies are improving treatment efficacy and patient outcomes.
-
Increasing Healthcare Expenditure
Rising healthcare spending and improved access to advanced respiratory care are supporting the adoption of innovative ILD therapies.
-
High Unmet Medical Need
Existing therapies mainly slow disease progression rather than cure the disease, creating demand for more effective and disease-modifying treatments.
DelveInsight's ‘Interstitial Lung Disease Market Insights, Epidemiology and Market Forecast – 2036’ report delivers an in-depth understanding of Interstitial Lung Disease, historical and forecasted epidemiology, as well as the Interstitial Lung Disease market trends in the United States, EU4 (Germany, Spain, Italy, and France) and the United Kingdom, and Japan.
The Interstitial Lung Disease Treatment Market Report delivers a comprehensive analysis of the current treatment landscape, including standards of care, clinical practices, and evolving therapeutic algorithms. It evaluates, Interstitial Lung Disease patient burden trends, revenue & market share dynamics, peak patient share & therapy uptake analysis, and provides an in-depth market size assessment, and growth rate projections (Historical & Forecast 2026–2036) across global regions. The Interstitial Lung Disease Treatment Market Report highlights key unmet medical needs in Interstitial Lung Disease and maps the competitive and clinical landscape to uncover high‑value opportunities, providing a clear outlook on future market growth potential.
Scope of the Interstitial Lung Disease Market Report | |
|
Study Period |
2022–2036 |
|
Historical Year |
2022–2025 |
|
Forecast Period |
2026–2036 |
|
Base Year |
2025 |
|
Geographies Covered |
|
|
Interstitial Lung Disease Market CAGR (Forecast period) | |
|
Interstitial Lung Disease Epidemiology Segmentation Analysis |
Patient Burden Assessment
|
|
Interstitial Lung Disease Companies |
|
|
Interstitial Lung Disease Therapies |
|
|
Interstitial Lung Disease Market |
Segmented by
|
|
Analysis |
|
Interstitial Lung Disease Understanding
Interstitial Lung Disease Overview and Diagnosis
Interstitial Lung Disease, also known as diffuse parenchymal lung diseases, represents a heterogeneous group of lung disorders characterized by shared clinical, radiographic, physiologic, or pathologic features. Understanding Interstitial Lung Disease is challenging due to complex terminology and broad disease diversity. Pathologically, these conditions involve inflammatory and fibrotic processes that extend beyond the interstitium to affect the lung parenchyma, including alveoli, alveolar ducts, and bronchioles.
The diagnosis of Interstitial Lung Disease requires a comprehensive and multidisciplinary approach, as Interstitial Lung Disease encompasses a broad and heterogeneous group of disorders with overlapping clinical, radiologic, and histopathologic features. Rather than relying on a single test, diagnosis is achieved through the integration of clinical history, physiologic assessment, imaging, laboratory evaluation, and, when necessary, tissue sampling, with the ultimate goal of identifying the underlying disease process and defining disease behavior.
Current Interstitial Lung Disease Treatment Landscape
The Interstitial Lung Disease treatment pipeline appears robust, with several therapies expected to launch during the forecast period. These include belimumab (BENLYSTA), treprostinil palmitil inhalation powder (TPIP), seralutinib, L606, admilparant (BMS-986278), N-115 and others. Long-term market growth will depend on payer recognition of clinical value, as reimbursement decisions increasingly rely on demonstrated reduction in proteinuria, delayed progression to end-stage kidney disease, and real-world safety outcomes.
Further details related to country-based variations are provided in the report.
Interstitial Lung Disease Unmet Needs
The section “unmet needs of Interstitial Lung Disease” outlines the critical gaps between the current state of patient care, diagnosis, and the ideal & effective management of the disease. It highlights the obstacles experienced by patients, clinicians, and researchers and identifies potential solutions for future progress.
- Limited Disease-Modifying Options Beyond Slowing Fibrosis
- High Treatment Burden and Poor Long-Term Tolerability
- Inadequate Stratification Across Heterogeneous Interstitial Lung Disease Subtypes
- Lack of Noninvasive Biomarkers for Early Diagnosis and Monitoring
Interstitial Lung Disease Epidemiology
Key Findings from Interstitial Lung Disease Epidemiological Analysis and Forecast
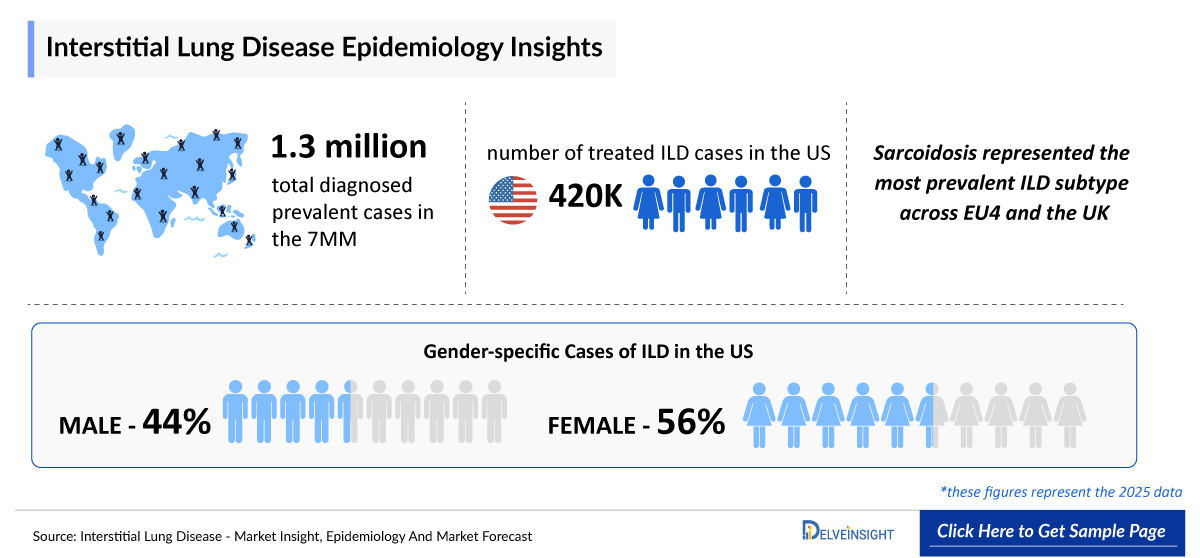
- The total number of Interstitial Lung Disease Diagnosed Prevalent Cases in the 7MM is ~1.3 million in 2025.
- According to DelveInsight’s estimates, in 2025, the US reported nearly 520,000 diagnosed prevalent cases of Interstitial Lung Disease.
- According to DelveInsight analysis, in 2025, males represented about 44% of Interstitial Lung Disease cases in the US, whereas females accounted for roughly 56%.
- In 2025, sarcoidosis represented the most prevalent Interstitial Lung Disease subtype across EU4 and the UK, accounting for the largest share of diagnosed patients with approximately 180,000 cases, highlighting the significant contribution of inflammatory and immune-mediated lung disease to the regional Interstitial Lung Disease burden. This was followed by IPF with about 120,000 patients, underscoring the continued clinical and healthcare impact of progressive fibrotic Interstitial Lung Disease.
- DelveInsight’s 2025 assessment highlights a modest gender disparity in Interstitial Lung Disease in Japan, with males accounting for approximately 52% of cases and females comprising about 48%. This near-balanced distribution suggests that while Interstitial Lung Disease slightly predominates in men—potentially reflecting higher exposure to occupational or environmental risk factors—the disease burden remains substantial in women, underscoring the need for gender-inclusive awareness, diagnosis, and management strategies.
- The number of treated Interstitial Lung Disease cases in the United States was about 420,000 in 2025 and is projected to increase by 2036.This upward trend highlights a steadily expanding treated population, reflecting both a rising underlying disease burden and improvements in diagnosis, treatment initiation, and long-term disease management across the US healthcare system.
Interstitial Lung Disease Epidemiological Segmentation in the 7MM
- Interstitial Lung Disease Diagnosed Prevalent Cases
- Interstitial Lung Disease Gender-specific Cases
- Interstitial Lung Disease Type-specific Cases
- Interstitial Lung Disease Treated Cases
Interstitial Lung Disease Drug Analysis & Competitive Landscape
The Interstitial Lung Disease drug chapter provides a detailed, market-focused review of approved therapies and the emerging Interstitial Lung Disease Pipeline across Phase I-III Interstitial Lung Disease Clinical Trials. It covers mechanism of action, ILD Clinical Trials data, regulatory approvals, patents, collaborations, strategic partnerships upcoming Key catalyst for each therapy, along with their advantages, limitations, and recent developments. This section offers critical insights into the Interstitial Lung Disease treatment landscape, supporting market assessment, competitive analysis, and growth forecasting for the Interstitial Lung Disease therapeutics market.
Interstitial Lung Disease Approved Therapies
-
Treprostinil (TYVASO/TYVASO DPI/TREPROST): United Therapeutics
TYVASO and treprostinil (TYVASO DPI) are inhaled prescription therapies for adults with PAH (Group 1) and PH-Interstitial Lung Disease (Group 3). As a prostacyclin analogue, treprostinil promotes pulmonary vasodilation and inhibits platelet aggregation, improving exercise capacity. United Therapeutics reported in March 2026 that the Phase III TETON-1 trial assessing inhaled treprostinil achieved its primary endpoint in patients with idiopathic pulmonary fibrosis (IPF), indicating a notable improvement in lung function (FVC) and supporting the drug's potential to extend beyond PH-Interstitial Lung Disease into more general Interstitial Lung Disease indications.
Positive outcomes from the Phase III TETON-2 trial in 2025 further demonstrated the safety and effectiveness of inhaled treprostinil in Interstitial Lung Disease, with steady improvements in lung function and no new safety signals, setting up the treatment for possible regulatory submission in populations with IPF and progressive fibrosing Interstitial Lung Disease Market.
Interstitial Lung Disease Marketed/Approved Therapies | ||||||
|
Drug/Therapy |
Company |
Indication |
Molecule Type |
MoA |
RoA |
Marketed Region |
|
Treprostinil (TYVASO/TYVASO DPI/TREPROST) |
United Therapeutics |
PH-Interstitial Lung Disease |
Small molecule |
Prostacyclin agonist |
Inhalation |
US:2021;2022 JP: 2024 |
|
Treprostinil (YUTREPIA) |
Liquidia Corporation |
PH-Interstitial Lung Disease |
Small molecule |
Prostacyclin agonist |
Oral inhalation |
US: 2025 |
|
Drug C |
XX |
XX |
XX |
XX |
XX |
XX |
Interstitial Lung Disease Pipeline Analysis
-
Belimumab (BENLYSTA): GSK
Belimumab (BENLYSTA) is a B-lymphocyte Stimulator (BLyS)-specific inhibitor that binds to soluble BLyS, a key survival factor for B cells. As a fully human monoclonal antibody (mAb), belimumab inhibits BLyS-mediated prolonged B-cell survival, including autoreactive B cells, and reduces the differentiation of B cells into immunoglobulin-producing plasma cells. Belimumab (BENLYSTA) is currently under evaluation in late-stage (Phase II/III and Phase III) clinical trials for the treatment of SSc-Interstitial Lung Disease , and CTD-Interstitial Lung Disease reflecting increasing interest in B-cell-targeted immunomodulation to preserve lung function and slow fibrotic disease progression.
-
- In February 2023, GSK announced that the US FDA has granted orphan drug designation (ODD) to Belimumab (BENLYSTA), a B-cell–inhibiting monoclonal antibody, for the potential treatment of systemic sclerosis.
Competitive Landscape of Pipeline Drugs | ||||||
|
Drug Name |
Company |
Highest Phase |
Indication |
RoA |
MoA |
Anticipated Launch in the US |
|
Belimumab (BENLYSTA) |
GSK |
III |
SSc-Interstitial Lung Disease; CTD-Interstitial Lung Disease |
SC |
BLyS |
Information is available in the full report |
|
Treprostinil palmitil inhalation powder (TPIP) |
Insmed |
III |
PH-Interstitial Lung Disease |
Oral inhalation |
Prostacyclin receptor agonist |
Information is available in the full report |
|
Seralutinib |
Gossamer Bio/ Chiesi Farmaceutici |
III |
PH-Interstitial Lung Disease |
Oral inhalation |
PDGFR, CSF1R, c-KIT inhibitor |
2030 |
|
L606 |
Liquidia Corporation |
III |
PH-Interstitial Lung Disease |
Inhalation |
Prostacyclin (IP receptor) agonist |
Information is available in the full report |
|
Drug E |
XX |
XX |
XX |
XX |
XX |
XX |
|
Note: Launch insights are provisional and may change with future report updates or the occurrence of major key catalysts. | ||||||
Interstitial Lung Disease Companies, Market Leaders and Emerging Companies
- United Therapeutics
- Liquidia Corporation
- Boehringer Ingelheim
- Roche
- Insmed
- GSK and others
Interstitial Lung Disease Drug Updates
- In December 2025, PureTech Health announced the successful completion of the End-of-Phase II (EOP2) meeting with the US FDA regarding the development of deupirfenidone (LYT-100) for the treatment of IPF.
- In December 2025, US FDA approved nerandomilast (JASCAYD) tablets for the treatment of PPF in adults.
- In November 2025, Endeavor BioMedicines reported that the EMA granted PRIME designation to its investigational therapy taladegib for the treatment of IPF.
Interstitial Lung Disease Market Outlook
The expanding Interstitial Lung Disease treatment market is complemented by widely used supportive and background therapies, including corticosteroids, immunosuppressants, biologics, and adjunctive agents such as cough suppressants. In parallel, the emergence of next-generation pipeline therapies—including seralutinib, L606, N-115, and efzofitimod—is broadening the therapeutic landscape by targeting diverse disease mechanisms, sustaining long-term market growth and treatment innovation.
Key Interstitial Lung Disease Marketed Therapies Shaping Current Management
Nerandomilast (JASCAYD): An oral, small-molecule therapy developed by Boehringer Ingelheim, is an oral phosphodiesterase-4 inhibitor approved in the US in December 2025, expanding the anti-fibrotic treatment landscape. By modulating intracellular inflammatory and fibrotic signaling pathways, nerandomilast provides an additional oral disease-modifying option for patients with chronic fibrosing Interstitial Lung Disease. Its introduction reflects ongoing efforts to diversify therapeutic mechanisms beyond tyrosine kinase inhibitior.
Nintedanib capsules (OFEV): An oral tyrosine kinase inhibitor (TKI) that targets key pathways driving Lung Fibrosis, helping slow lung function decline. It is approved for IPF, Systemic Sclerosis-associated Interstitial Lung Disease, and other chronic fibrosing Interstitial Lung Diseases with a progressive phenotype. In March 2025, Sandoz reported that it reached a federal court consent judgment with Boehringer Ingelheim allowing its proposed generic nintedanib capsules (100 mg and 150 mg) to proceed without infringing key OFEV patents, potentially accelerating US generic competition.
Overall, the launch of first-in-class therapies, improved testing, and rising disease awareness are expected to drive steady growth in the 7MM Interstitial Lung Disease Treatment Market from 2022–2036, with strong commercial implications for both marketed products and emerging pipelines. As diagnostic sophistication improves — driven by HRCT, multidisciplinary consensus, and broader awareness — the pool of patients recognized with PF-Interstitial Lung Disease has expanded beyond traditional IPF. This expansion, coupled with unmet needs in disease modification and quality-of-life preservation, makes Interstitial Lung Disease a strategically important specialty market with evolving treatment paradigms and high per-patient value.
- Among the 7MM, the US accounted for the largest Interstitial Lung Disease Market Size. i.e., USD ~ 5 billion in 2025.
- Japan’s Interstitial Lung Disease market exceeded USD 350 million in 2025 and is projected to grow further through 2036, driven by rising treatment adoption and the expected introduction of emerging therapies.
- In 2036, among all the therapies for Interstitial Lung Disease, the highest revenue is estimated to be generated by JASCAYD (nerandomilast tablets) in the 7MM.
- The Interstitial Lung Disease therapeutic pipeline is undergoing a decisive shift from conventional anti-fibrotic and broad immunosuppressive strategies toward targeted biologics, novel small molecules, and advanced inhaled delivery platforms. Several late- and mid-stage programs now aim to address immune dysregulation, fibrotic signaling, and pulmonary vascular remodeling in a more mechanistically precise manner. BENLYSTA (Belimumab), developed by GSK, represents a shift toward B-cell-targeted therapy in autoimmune-driven interstitial lung disease, particularly systemic sclerosis–associated and connective tissue disease–associated Interstitial Lung Disease.
Interstitial Lung Disease Drug Class/Insights into Leading Emerging and Marketed Therapies (2026–2036 Forecast)
Corticosteroids and immunosuppressive agents: Many patients are initially treated with corticosteroids, such as prednisone. In selected cases—particularly when Interstitial Lung Disease is associated with inflammatory or autoimmune processes—additional immunosuppressive agents may be used. These therapies can help slow disease progression in certain Interstitial Lung Disease subtypes. Antifibrotic therapies: Pirfenidone and nintedanib have demonstrated the ability to slow functional decline in IPF. Nintedanib is also approved for use in progressive fibrosing Interstitial Lung Diseases of other etiologies. Both agents are commonly associated with adverse effects, making individualized risk–benefit assessment essential.
Acid suppression therapies: Gastroesophageal reflux disease is frequently observed in IPF and has been associated with increased lung injury. In patients with symptomatic reflux, acid-suppressive medications may be considered to mitigate potential pulmonary impact.
Interstitial Lung Disease Drug Uptake
This section focuses on the uptake rate of potential Interstitial Lung Disease drugs expected to be launched in the market during the forecast period (2026–2036). The analysis covers the Interstitial Lung Disease drug’s uptake, performance at peak, factors affecting performance during prime years of growth, patient uptake by therapy, and anticipated sales generated by each drug. The Interstitial Lung Disease Treatment Market Size is anticipated to experience growth during the forecast period due to promising emerging treatment options that include admilparant (BMS-986278), seralutinib, L606, treprostinil palmitil inhalation powder (TPIP) and others.
Interstitial Lung Disease Emerging Clinical and Market Trends
Broadening the spectrum of diagnosed diseases
Progressive fibrosing Interstitial Lung Disease Market across multiple subtypes is expanding treatment beyond IPF, with agents such as nintedanib now used in PF-Interstitial Lung Disease and CTD-Interstitial Lung Disease.
Earlier and more precise diagnosis
HRCT and AI-supported imaging are enabling earlier detection, supporting the timely initiation of anti-fibrotics and immunomodulators.
Phenotype-driven therapeutics
Autoimmune-predominant Interstitial Lung Disease is advancing toward targeted immunotherapies such as belimumab, while fibrotic-predominant disease remains anchored by nintedanib and pirfenidone.
Inhaled delivery renaissance
Localized lung delivery is expanding with inhaled treprostinil and next-generation inhaled anti-fibrotic candidates.
Interstitial Lung DiseaseMarket Access and Reimbursement of Approved Therapies
The Interstitial Lung Disease Treatment Market Report further provides detailed insights on the country-wise accessibility and reimbursement scenarios, cost-effectiveness scenario of approved therapies, programs making accessibility easier and out-of-pocket costs more affordable, insights on patients insured under federal or state government prescription drug programs, etc.
EU4 and the UK
EU4 and the UK Reimbursement of Therapies Approved for Interstitial Lung Disease | |
|
Drug/Therapy |
AEMPS Assessment for Interstitial Lung Disease |
|
OFEV |
Nintedanib was approved in April 2020 for treating Systemic Sclerosis-associated Interstitial Lung Disease in adults, based on the SENSCIS trial, a randomized double-blind study in 580 patients showing reduced annual FVC decline over one year. |
Reimbursement is a crucial factor that affects the drug’s access to the market. Often, the decision to reimburse comes down to the price of the drug relative to the benefit it produces in treated patients. To reduce the healthcare burden of these high-cost therapies, many payment models are being considered by payers and other industry insiders.
Interstitial Lung Disease Therapies Price Scenario & Trends
Pricing and analogue assessment of Interstitial Lung Disease Therapies highlights evolving price dynamics structures. This section summarizes the cost of approved treatments, closest and most appropriate analogue selection for emerging therapies, and understanding of how pricing influences market access, adherence, and long-term uptake.
Pricing of Interstitial Lung Disease-Approved Drugs
OFEV treatment cost was evaluated using the approved dosing regimen of 150 mg administered orally twice daily with food. Under this standardized assumption, the annual treatment cost in the US was estimated nearly USD 160,000, reflecting prevailing list pricing. In Germany, pricing based on the Gemeinsamer Bundesausschuss (G-BA) framework yielded an annual cost around USD 41,938, while in the United Kingdom, NICE-aligned pricing resulted in an annual treatment cost nearly USD 35,000.
Interstitial Lung Disease Industry Experts and Physician Views
To keep up with Interstitial Lung Disease market trends, we take Key Opinion Leaders (KOLs) and Subject Matter Experts (SMEs) opinions working in the domain through primary research to fill the data gaps and validate our secondary research. Industry Experts were contacted for insights on the Interstitial Lung Disease’s emerging therapies, evolving treatment landscape, patient adherence to conventional therapies, therapy switching trends, drug adoption and uptake, accessibility challenges, and epidemiology and real-world prescription patterns in Interstitial Lung Disease, including MD, PhD, Instructor, Postdoctoral Researcher, Professor, Researcher, and others.
DelveInsight’s analysts connected with 8+ KOLs to gather insights at country level. Centers such as the University of Alabama, University Hospital Heidelberg, Keio University Hospital, and Hannover Medical School etc. were contacted. Their opinion helps understand and validate current and emerging Interstitial Lung Disease therapies, highlight unmet medical needs, provide epidemiological context, and support strategic decisions for market access, therapy adoption, and pipeline prioritization in Interstitial Lung Disease.
Region |
Key Opinion Leaders (KOLs) and Subject Matter Experts |
|
United States |
“Patients with PH-Interstitial Lung Disease often face a decline in functional capacity and commonly depend on supplemental oxygen. They also exhibit elevated healthcare resource utilization (HCRU) and face significantly high mortality rates. Within a few years of diagnosis, mortality can reach alarming levels.” |
|
Germany |
“The absence of reliable biomarkers to predict disease progression continues to limit early intervention in Interstitial Lung Disease, resulting in treatment initiation only after irreversible functional decline has occurred.” |
Interstitial Lung Disease Qualitative Analysis: SWOT and Conjoint Analysis
We perform qualitative and Interstitial Lung Disease Treatment Market Intelligence analysis using various approaches, such as SWOT analysis and conjoint analysis. In the SWOT analysis of Interstitial Lung Disease, strengths, weaknesses, opportunities, and threats in terms of disease diagnosis, patient awareness, patient burden, competitive landscape, cost-effectiveness, and geographical accessibility of therapies are provided. Conjoint analysis analyzes emerging therapies based on relevant attributes such as safety, efficacy, frequency of administration, route of administration, and order of entry. Scoring is given based on these parameters to analyze the effectiveness of therapy.
The team of analysts analyzes promising emerging therapies based on relevant attributes such as safety, efficacy, frequency of administration, route of administration, and order of entry. In efficacy, the trial’s primary and secondary outcome measures are evaluated, whereas the therapies’ safety is evaluated, wherein the acceptability, tolerability, and adverse events are majorly observed. In addition, the scoring is also based on the route of administration, order of entry, probability of success, and the addressable patient pool for each therapy. According to these parameters, the final weightage score and the ranking of the emerging therapies are decided.
Scope of the Interstitial Lung Disease Market Report
- The Interstitial Lung Disease Therapeutics Market Report covers a segment of key events, an executive summary, a descriptive overview, explaining their causes, signs and symptoms, pathogenesis, and currently available treatments.
- Comprehensive insight has been provided into the Interstitial Lung Disease epidemiology segments and forecasts, the future growth potential of the diagnosis rate, and disease progression along treatment guidelines.
- Additionally, an all-inclusive account of both the current and emerging treatments, along with the elaborative profiles of late-stage and prominent therapies, will have an impact on the current Interstitial Lung Disease Treatment landscape.
- A detailed review of the Interstitial Lung Disease Treatment Market, historical and forecasted ILD Market Size, ILD Market Share by therapies, detailed assumptions, and rationale behind our approach is included in the report, covering the 7MM drug outreach.
- The Interstitial Lung Disease Therapeutics Market Report provides an edge while developing business strategies by understanding trends through SWOT analysis and expert insights/KOL views, patient journey, and treatment preferences that help in shaping and driving the 7MM ILD Market.
Interstitial Lung Disease Market Report Insights
- Interstitial Lung Disease Patient Population Forecast
- Interstitial Lung Disease Therapeutics Market Size
- Interstitial Lung Disease pipeline analysis
- Interstitial Lung Disease market size and trends
- Interstitial Lung Disease market opportunity (Current and forecasted)
Interstitial Lung Disease Market Report Key Strengths
- Epidemiology‑based (Epi‑based) bottom‑up forecasting
- Artificial Intelligence (AI)-enabled market research report
- 11-year forecast
- ILD Market Outlook (North America, Europe, Asia-Pacific)
- Patient Burden trends (by geography)
- Interstitial Lung Disease Treatment addressable Market (TAM)
- Interstitial Lung Disease Competitive Landscape
- Interstitial Lung Disease major companies Insights
- Interstitial Lung Disease Price trends and analogue assessment
- Interstitial Lung Disease Therapies Drug Adoption/Uptake
- Interstitial Lung Disease Therapies Peak Patient Share analysis
Interstitial Lung Disease Market Report Assessment
- Current Interstitial Lung Disease Treatment Practices
- Interstitial Lung Disease Unmet needs
- Interstitial Lung Disease Clinical Development Analysis
- Interstitial Lung Disease emerging drugs product profiles
- Interstitial Lung Disease Market Attractiveness
- Interstitial Lung Disease Qualitative analysis (SWOT and conjoint analysis)
Key Questions Answered in the Interstitial Lung Disease Market Report
ILD Market Insights
- What was the Interstitial Lung Disease Treatment Market Size, the ILD Market Size by therapies, ILD Market Share (%) distribution in 2026, and what would it look like by 2036? What are the contributing factors for this growth?
- What are the anticipated pricing variations among different geographies for the emerging therapies in the future?
- What can be the future treatment paradigm of Interstitial Lung Disease?
- What are the disease risks, burdens, and ILD Unmet Needs? What will be the growth opportunities across the 7MM concerning the patient population with Interstitial Lung Disease?
- Who is the major future competitor in the market, and how will the competitors affect their market share?
- What are the current options for the treatment of Interstitial Lung Disease? What are the current guidelines for treating Interstitial Lung Disease in the US, Europe, and Japan?
Reasons to Buy the ILD Market Report
- The Interstitial Lung Disease Therapeutics Market Report will help in developing business strategies by understanding the latest trends and changing treatment dynamics driving the ILD Market.
- Bottom-up forecasting Interstitial Lung Diseases from the affected population to product forecasts, delivering a robust, data driven approach ideal for new therapies and novel classes.
- Insights on patient burden/disease Interstitial Lung Disease Prevalence, evolution in diagnosis, and factors contributing to the change in the epidemiology of the disease during the forecast years.
- Understand the existing ILD Market opportunities in varying geographies and the growth potential over the coming years.
- Identifying strong upcoming players in the ILD Market will help devise strategies to help get ahead of competitors.
- Detailed analysis and ranking of class-wise potential current and emerging therapies under the conjoint analysis section to provide visibility around leading classes.
- To understand KOLs’ perspectives on the accessibility, acceptability, and compliance-related challenges of existing treatment to overcome barriers in the future.
- Detailed insights on the unmet needs of the existing ILD Market so that the upcoming players can strengthen their development and launch strategy.
- This Artificial Intelligence (AI) enabled report summarize and simplify complex datasets within the report into clear, actionable insights for stakeholders, investors, and healthcare providers, enabling faster, data driven decisions.
Stay updated with us for Recent Articles