Endometriosis Market Summary
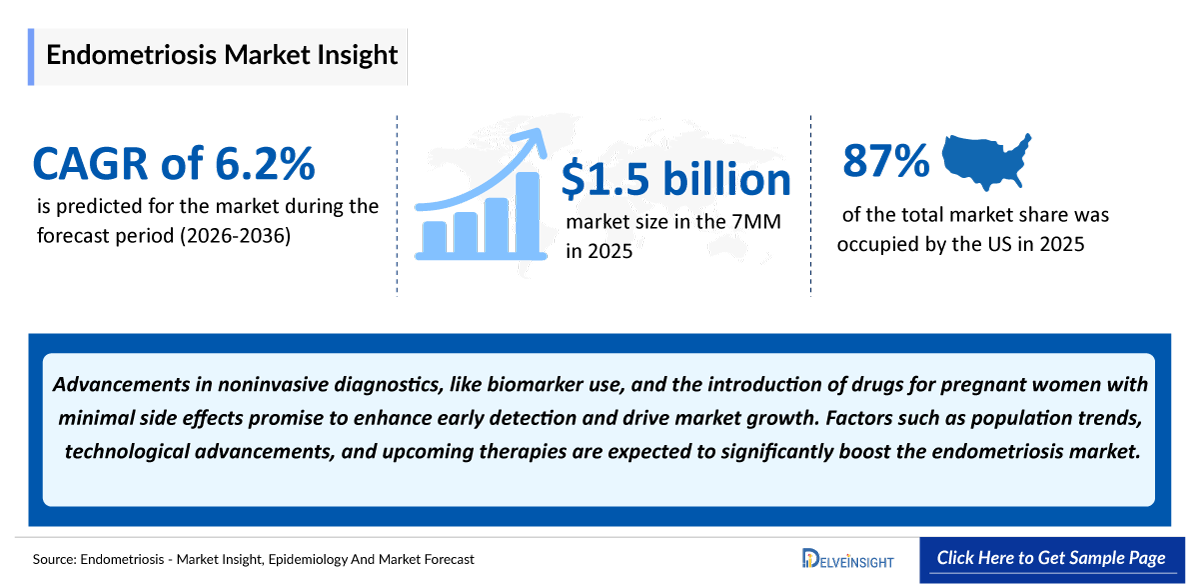
- According to DelveInsight’s analysis, endometriosis market size was found to be around USD 1,500 million in the leading markets (the United States, the EU4 (Germany, France, Italy, and Spain), the United Kingdom, and Japan) in 2025.
Endometriosis Market Insights and Trends
- Endometrial diseases reported approximately 7 million prevalent cases and around 4 million diagnosed prevalent cases in the US.
- Current treatment strategies remain largely suppressive, relying on hormonal therapies (Oral (combined) contraceptive pills, progestins, gonadotropin-releasing hormone analogs) and surgery. Surgical intervention provides short-term relief but limited durability, with high recurrence rates and diminishing returns following repeat procedures. This reinforces the unmet need for non-surgical, disease-modifying therapies.
- In the US, contraceptive-based therapies generated around USD 150 million in 2025. This segment is anticipated to experience gradual yet consistent growth, with revenues projected to rise steadily over the forecast period, reaching higher levels by 2036.
- The most transformative shift in endometriosis management has occurred with the introduction of oral GnRH antagonists. In the US, FDA approvals of elagolix (ORILISSA) by AbbVie and relugolix with estradiol and norethindrone acetate (MYFEMBREE) by Myovant/Pfizer marked a departure from injectable GnRH agonists by enabling rapid, dose-dependent estrogen suppression without an initial flare.
- Relugolix with estradiol and norethindrone acetate (MYFEMBREE) holds a competitive advantage due to its fixed-dose add-back composition, which improves bone protection and supports longer-term use.
- The emergence of next-generation pipeline therapies including HMI-115, and TU2670 (MERIGOLIX) are broadening the therapeutic landscape by targeting distinct hormonal and disease-modifying mechanisms. These innovations are expected to diversify treatment options, address limitations of current standards of care, and support sustained market growth and therapeutic innovation over the long term.
Endometriosis Market size and forecast
- 2025 Endometriosis Market Size in the 7MM: USD ~1,500 million
- 2036 Projected Endometriosis Market Size in the 7MM: USD ~2,700 million
- Endometriosis Growth Rate (2026–2036) in the 7MM: 6.2% CAGR
DelveInsight's ‘Endometriosis Market Insights, Epidemiology and Market Forecast – 2036’ report delivers an in-depth understanding of endometriosis, historical and forecasted epidemiology, as well as the endometriosis market trends in the United States, EU4 (Germany, Spain, Italy, and France), the United Kingdom, and Japan
The endometriosis market report delivers a comprehensive analysis of the current treatment landscape, including standards of care, clinical practices, and evolving therapeutic algorithms. It evaluates, endometriosis patient burden trends, revenue & market share dynamics, peak patient share & therapy uptake analysis, and provides an in-depth market size assessment, and growth rate projections (Historical & Forecast 2022–2036) across the 7MM regions. The report highlights key unmet medical needs in endometriosis and maps the competitive and clinical landscape to uncover high‑value opportunities, providing a clear outlook on future market growth potential.
|
Study Period |
2022–2036 |
|
Historical Year |
2022–2025 |
|
Forecast Period |
2026–2036 |
|
Base Year |
2025 |
|
Geographies Covered |
|
|
Endometriosis Market CAGR (Study period/Forecast period) |
6.2% (2026-2036) |
|
Endometriosis Epidemiology Segmentation Analysis |
Patient Burden Assesment
|
|
Endometriosis Companies |
|
|
Endometriosis Therapies |
|
|
Endometriosis Market |
Segmented by
|
|
Analysis |
|
Endometriosis Understanding and Treatment Algorithm
Endometriosis Overview and Diagnosis
Endometriosis, a chronic estrogen-dependent inflammatory disease, involves the growth of endometrial-like tissue outside the uterus, commonly affecting the ovaries, fallopian tubes, and pelvic peritoneum, leading to pain and infertility.The range of symptoms varies by lesion location but commonly includes dysmenorrhea, chronic pelvic pain, dyspareunia, heavy periods, and infertility.
Laparoscopy with histological confirmation remains the gold standard, revealing ectopic endometrial glands/stroma. Noninvasive tools like transvaginal ultrasound and MRI aid in detecting ovarian endometriomas or deep lesions, while biomarkers and endometrial nerve fiber analysis are emerging but not definitive. First-line management for pain targets hormonal suppression with NSAIDs, combined oral contraceptives, or progestins. GnRH agonists/antagonists (e.g., with add-back therapy) are second-line for persistent symptoms, while laparoscopic excision of lesions improves pain and fertility in select cases. Multidisciplinary care emphasizes shared decision-making, considering fertility goals.
Further details are provided in the report.
Current Endometriosis Treatment Landscape
The overall endometriosis market is expected to expand over the forecast period due to the advancement of late- and mid-stage pipeline therapies, including HMI-115, a first-in-class prolactin receptor–targeting antibody; TU2670 (MERIGOLIX), an oral GnRH antagonist; and linzagolix choline (YSELTY), emerging in Japan. Collectively, these agents are expected to diversify the treatment landscape, improve tolerability and durability of response, and sustain long-term market growth.
Further details related to country-based variations are provided in the report.
Endometriosis Unmet Needs
The section “unmet needs of endometriosis” outlines the critical gaps between the current state of patient care, diagnosis, and the ideal & effective management of the disease. It highlights the obstacles experienced by patients, clinicians, and researchers and identifies potential solutions for future progress.
- Lack of Disease-modifying Therapies
- Limited Nonhormonal and Fertility-sparing Therapeutic Choices
- High Recurrence Rates Following Surgical Intervention
- Insufficient Long-term Medical Options Compatible With Chronic use
- Inadequate Solutions for Persistent Pain Independent of Lesion Burden
and others…
Note: Comprehensive unmet needs insights in endometriosis and their strategic implications are provided in the full report.
Endometriosis Epidemiology
Key Findings from Endometriosis Epidemiological Analysis and Forecast
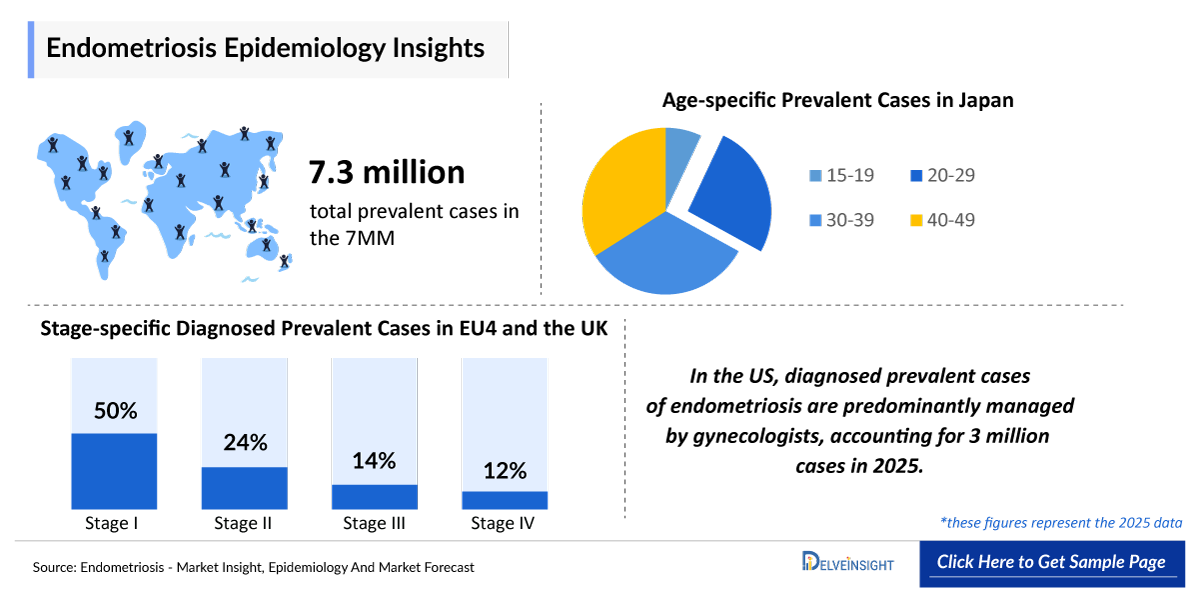
- The total number of prevalent cases of endometriosis in the 7MM is ~12 million in 2025, whereas the total number of diagnosed prevalent cases of endometriosis in the 7MM is around ~ 6 million in 2025.
- According to DelveInsight’s estimates, in 2025, in the United States, diagnosed prevalent cases of endometriosis are predominantly managed by gynecologists, accounting for 3 million cases in 2025.
- In Japan, the age-specific prevalent cases of endometriosis is of 7% for ages 15–19, 26% for 20–29, 33% for 30–39, and 34% for 40–49 years.
- In 2025, Stage I (minimal) endometriosis accounted for the largest share of diagnosed prevalent cases across EU4 and the UK (50%), followed by Stage II (mild) disease (24%) and Stage III (moderate) disease (14%). Stage IV (severe) endometriosis represented the smallest proportion of the overall case burden (13%).
- Diagnosed prevalent cases of endometriosis in Japan were predominantly managed by gynecologists (600,000 cases), followed by generalists (270,000 cases) and other prescribers (27,000 cases).
Endometriosis Drug Analysis & Competitive Landscape
The endometriosis drug chapter provides a detailed, market-focused review of approved therapies and the emerging pipeline across Phase II clinical trials. It covers mechanism of action, clinical trial data, regulatory approvals, patents, collaborations, strategic partnerships upcoming Key catalyst for each therapy, along with their advantages, limitations, and recent developments. This section offers critical insights into the endometriosis treatment landscape, supporting market assessment, competitive analysis, and growth forecasting for the endometrial therapeutics market.
Approved Therapies for Endometriosis
Linzagolix choline (YSELTY): Kissei Pharmaceutical/Theramex
Linzagolix choline (YSELTY) is well-positioned as a next-generation oral GnRH antagonist in the endometriosis market, combining flexible, dose-dependent estrogen suppression with the convenience of oral administration and add-back therapy. It is an oral, small-molecule GnRH receptor antagonist for endometriosis-associated pain. It provides dose-dependent suppression of estrogen, enabling both partial and full hormonal suppression with flexible dosing. The oral, non-peptide profile allows rapid onset and avoids injectable therapy.
In May 2025, Theramex reported that the National Institute for Health and Care Excellence (NICE) recommended NHS reimbursement of linzagolix (YSELTY) for the symptomatic treatment of endometriosis in women with a history of prior medical or surgical treatment.
|
Endometriosis Marketed/Approved Therapies | ||||||
|
Drug/Therapy |
Company |
Indication |
Molecule Type |
MoA |
RoA |
Marketed Region |
|
Linzagolix choline (YSELTY ) |
Kissei Pharmaceutical/Theramex |
Endometriosis |
Small molecule |
GnRH receptor antagonist |
Oral |
EU: 2024 UK: 2025 |
|
Elagolix (ORILISSA) |
AbbVie |
Endometriosis |
Small molecule |
GnRH receptor antagonist |
Oral |
US: 2018 |
Endometriosis Pipeline Analysis
HMI-115 : Hope Medicine
HMI-115 is a humanized monoclonal antibody against the Prolactin Receptor (PRLR) developed by Hope Medicine for endometriosis and other prolactin-driven gynecologic disorders. It blocks prolactin signaling, a pathway linked to pain sensitization, inflammation, and lesion persistence in endometriosis.
In December 2025, HMI-115 reported that its lead pipeline product, a monoclonal antibody targeting the prolactin receptor, received Fast Track Designation (FTD) from the US FDA for the treatment of moderate to severe pain associated with endometriosis. The drug is currently in Phase II and data demonstrate dose-dependent, clinically meaningful reductions in dysmenorrhea and nonmenstrual pelvic pain, with durable effects beyond treatment and reduced reliance on NSAIDs.
|
Competitive Landscape of Pipeline Drugs | ||||||
|
Drug Name |
Company |
Highest Phase |
Indication |
RoA |
MoA |
Anticipated Launch in the US |
|
Vipoglanstat (GS-248) |
Gesynta Pharma |
II |
Endometriosis |
Oral |
HPGDS inhibition |
Information is available in the full report |
|
HMI-115 |
Hope Medicine |
II |
Endometriosis |
SC |
PRLR antagonism |
2030 |
|
TU2670 (Merigolix) |
TiumBio |
II |
Endometriosis |
Oral |
GnRH antagonist |
Information is available in the full report |
|
VML-0501 |
Viramal |
II |
Endometriosis |
Locally |
sEH inhibitor |
Information is available in the full report |
|
Note: Launch insights are provisional and may change with future report updates or the occurrence of major key catalysts. | ||||||
Endometriosis Key Players, Market Leaders and Emerging Companies
- AbbVie
- Kissei Pharmaceutical/Theramex
- ASKA Pharmaceutical
- Myovant Sciences/Pfizer
- Hope Medicine
- TiumBio
- Gesynta Pharma and others
Endometriosis Drug Updates
- As of March 2026, Gesynta Pharma is administering vipoglanstat (GS-248) to the first patient in a new Phase II trial of a revolutionary, non-hormonal treatment for endometriosis. The NOVA trial advances the candidate to mid-stage testing in an area where novel therapeutic approaches that circumvent the adverse effects of hormone-based therapy.
- In June 2025, NICE reported a positive recommendation for linzagolix choline (YSELTY) for NHS reimbursement for the symptomatic treatment of endometriosis in women with a history of prior medical or surgical intervention.
Endometriosis Market Outlook
Current care in endometriosis is largely symptomatic, focused on suppressing ovarian hormone activity to reduce pain, bleeding, and lesion activity rather than reversing underlying disease biology.
Key marketed therapies shaping current management
Elagolix (ORILISSA); AbbVie: A first-in-class oral GnRH antagonist approved for moderate to severe endometriosis-associated pain. ORILISSA provides rapid, dose-dependent suppression of ovarian hormone production, enabling flexibility between partial and near-complete estrogen suppression.
Relugolix, estradiol, and norethindrone acetate (MYFEMBREE/RYEQO); Myovant Sciences/Pfizer: An oral GnRH antagonist combination designed to balance effective gonadotropin suppression with hormonal add-back to mitigate hypoestrogenic adverse effects. The convenience of once-daily oral dosing and a more favorable long-term safety profile have driven strong uptake across the US, EU4 and the UK.
Linzagolix choline (YSELTY); Kissei Pharmaceutical/Theramex: A selective oral GnRH antagonist developed with both dose-flexible monotherapy and combination (add-back) strategies. YSELTY is positioned to offer individualized estrogen suppression, aiming to optimize pain reduction.
And more
US administrative claims data show that endometriosis care is surgery-dominant with variable use of medical therapy. Overall, 70.9–72.1% of patients underwent surgery, while 42.1–44.9% received pharmacologic therapy (excluding opioids). Surgery was frequently first-line, used in 37.6% (OPTUM) and 41.8% (MarketScan) of patients; among these, laparoscopy accounted for 53.4–57.6% and hysterectomy for 42.4–46.6%.
- Overall, the launch of first-in-class therapies, improved testing, and rising disease awareness are expected to drive steady growth in the 7MM endometriosis market from 2022–2036, with strong commercial implications for both marketed products and emerging pipelines.
- Among the 7MM, the US accounted for the largest market size of endometriosis. i.e., USD ~ 1,300 million in 2025.
- In 2036, among all the therapies for endometriosis, the highest revenue is estimated to be generated by HMI-115 in the 7MM.
- The most meaningful recent shift in Europe has been the approval of oral GnRH antagonists with improved usability profiles. Relugolix, estradiol, and norethindrone acetate (MYFEMBREE/RYEQO) has been approved across EU markets, offering fixed-dose add-back therapy that supports longer treatment duration and improved bone protection.
Further details will be provided in the report….
Drug Class/Insights into Leading Emerging and Marketed Therapies in Endometriosis (2022–2036 Forecast)
The endometriosis market comprises Oral (combined) Contraceptive Pills, Progestin-only Pills GnRH agonists, GnRH antagonists , Opioids each targeting different aspects of endometriosis.
Oral (combined) Contraceptive Pills (OCPs): Widely used first-line hormonal option that suppresses ovulation and stabilizes hormonal fluctuations. OCPs can reduce dysmenorrhea and pelvic pain but often provide incomplete relief and are associated with breakthrough bleeding and recurrence after discontinuation.
Progestin-only Pills (POPs): Common alternatives for patients who cannot tolerate estrogen. POPs reduce endometrial proliferation and inflammation but are frequently limited by irregular bleeding, mood effects, and variable pain control.
GnRH agonists: Potent suppressors of gonadotropin release that induce a hypoestrogenic state and provide strong pain reduction. Their use is constrained by delayed onset, flare effects, vasomotor symptoms, bone density loss, and duration limits.
GnRH antagonists: Oral agents that rapidly suppress gonadotropins without an initial flare. They offer dose-dependent estrogen suppression and improved convenience compared with injectables, but long-term use still raises concerns around hypoestrogenic adverse effects and bone health.
Danazol: An older androgenic agent that effectively suppresses endometriosis activity but has largely fallen out of favor due to masculinizing side effects, metabolic effects, and poor tolerability.
Opioids: Sometimes used for severe or refractory pain, particularly during flares. While they may provide short-term analgesia, they do not address disease drivers and carry substantial risks, including dependency, tolerance, and impaired functioning.
Endometriosis Drug Uptake
This section focuses on the uptake rate of potential drugs expected to be launched in the market during the forecast period (2026–2036). The analysis covers the endometriosis drug’s uptake, performance at peak, factors affecting performance during prime years of growth, patient uptake by therapy, and anticipated sales generated by each drug.
Among medical therapies, progestin-only pills were most commonly used (16.1–17.4%), followed by combined oral contraceptives (14.2–14.4%). GnRH agonists were prescribed in 9.3–11.3%, while GnRH antagonists showed limited uptake at 1.8–2.2%; danazol use was rare (0.3%). Opioids were used in 27.8–28.8% of patients overall and as first-line therapy in 12.5–12.7%, with over half of opioid-first patients requiring subsequent treatment.
The emergence of next-generation pipeline therapies including HMI-115, and TU2670 (MERIGOLIX) are broadening the therapeutic landscape by targeting distinct hormonal and disease-modifying mechanisms. These innovations are expected to diversify treatment options, address limitations of current standards of care, and support sustained market growth and therapeutic innovation over the long term. HMI-115, developed by Hope Medicine, is a subcutaneously administered monoclonal antibody that blocks the prolactin receptor signaling pathway, a novel, non-hormonal mechanism implicated in endometriosis-associated pain.
By avoiding direct ovarian hormone suppression, HMI-115 may offer a differentiated option for patients intolerant to GnRH-based therapies. If approved, HMI-115 is anticipated to launch in major markets in 2030 and is projected to increase revenue by 2036.
Detailed insights of emerging therapies' drug uptake is included in the report
Market Access and Reimbursement of Approved therapies in Endometriosis
The report further provides detailed insights on the country-wise accessibility and reimbursement scenarios, cost-effectiveness scenario of approved therapies, programs making accessibility easier and out-of-pocket costs more affordable, insights on patients insured under federal or state government prescription drug programs, etc.
EU4 and the UK
|
EU4 and the UK Reimbursement of Therapies Approved for Endometriosis | |
|
Drug/Therapy |
Indication |
|
Linzagolix choline (YSELTY) |
Symptomatic treatment of endometriosis in adult women of reproductive age with a history of previous medical or surgical treatment for their endometriosis, with concomitant hormonal addback therapy. |
Reimbursement is a crucial factor that affects the drug’s access to the market. Often, the decision to reimburse comes down to the price of the drug relative to the benefit it produces in treated patients. To reduce the healthcare burden of these high-cost therapies, many payment models are being considered by payers and other industry insiders.
NOTE: Further Details are provided in the final report….
Endometriosis therapies Price Scenario & Trends
Pricing and analogue assessment of endometriosis therapies highlights evolving price dynamics structures. This section summarizes the cost of approved treatments, closest and most approproiate analogue selection for emerging therapies, and understanding of how pricing influences market access, adherence, and long-term uptake.
Pricing of Endometriosis Approved Drugs
Linzagolix choline (YSELTY) is expected to be launched in Japan in 2028. Using internal benchmarks and regional price-adjustment ratios, an annual cost of therapy of approximately USD 1,050 was assumed for Japan, based on a 200 mg daily dose and a 6-month active treatment window annualized to 12 months.
Industry Experts and Physician Views for Endometriosis
To keep up with endometriosis market trends, we take Key Opinion Leaders (KOLs) and Subject Matter Experts (SMEs) opinions working in the domain through primary research to fill the data gaps and validate our secondary research. Industry Experts were contacted for insights on the endmetriosis emerging therapies, evolving treatment landscape, patient adherence to conventional therapies, therapy switching trends, drug adoption and uptake, accessibility challenges, and epidemiology and real-world prescription patterns in endometriosis, including MD, PhD, Instructor, Postdoctoral Researcher, Professor, Researcher, and others.
DelveInsight’s analysts connected with 8+ KOLs to gather insights at country level. Centers such as theUniversity of California, Institut Franco Européen Multidisciplinaire d'endométriose, the and Royal College of Obstetricians and Gynaecologists, etc. were contacted.
Their opinion helps understand and validate current and emerging endometriosis therapies, highlight unmet medical needs, provide epidemiological context, and support strategic decisions for Market access, therapy adoption, and pipeline prioritization in endometriosis.
|
Region |
Key Opinion Leaders (KOLs) and Subject Matter Experts (SMEs) |
|
United States |
“Endometriosis is a chronic inflammatory condition with ectopic endometrial-like tissue that triggers pain and infertility through hormonally driven immunologic and inflammatory mechanisms, not merely a surgical disease.” |
|
Germany |
“Endometriosis pathology is not uniform, lesions exhibit variable aromatase activity and progesterone resistance, which contributes to heterogeneous treatment responses. Research should focuses on understanding pain neurobiology and lesion-associated inflammation, with the goal of diversifying therapeutic strategies beyond conventional hormonal suppression into modulators of neuroinflammation.” |
Qualitative Analysis: SWOT and Conjoint Analysis
We perform qualitative and market Intelligence analysis using various approaches, such as SWOT analysis and conjoint analysis.
In the SWOT analysis of endometriosis, strengths, weaknesses, opportunities, and threats in terms of disease diagnosis, patient awareness, patient burden, competitive landscape, cost-effectiveness, and geographical accessibility of therapies are provided.Attribute analysis analyzes emerging therapies based on relevant attributes such as safety, efficacy, frequency of administration, route of administration, and order of entry. Scoring is given based on these parameters to analyze the effectiveness of therapy.
The team of analysts analyzes promising emerging therapies based on relevant attributes such as safety, efficacy, frequency of administration, route of administration, and order of entry. In efficacy, the trial’s primary and secondary outcome measures are evaluated, whereas the therapies’ safety is evaluated, wherein the acceptability, tolerability, and adverse events are majorly observed. In addition, the scoring is also based on the route of administration, order of entry, probability of success, and the addressable patient pool for each therapy. According to these parameters, the final weightage score and the ranking of the emerging therapies are decided.
Scope of the Report
- The report covers a segment of key events, an executive summary, a descriptive overview of endometriosis, explaining their causes, signs and symptoms, pathogenesis, and currently available treatments.
- Comprehensive insight has been provided into the epidemiology segments and forecasts, the future growth potential of the diagnosis rate, and disease progression along treatment guidelines.
- Additionally, an all-inclusive account of both the current and emerging treatments, along with the elaborative profiles of late-stage and prominent therapies, will have an impact on the current treatment landscape.
- A detailed review of the endometriosis market, historical and forecasted market size, market share by therapies, detailed assumptions, and rationale behind our approach is included in the report, covering the 7MM drug outreach.
- The report provides an edge while developing business strategies by understanding trends through SWOT analysis and expert insights/KOL views, patient journey, and treatment preferences that help in shaping and driving the 7MM endometriosis market.
Report Insights
- Endometriosis Patient Population Forecast
- Endometriosis Therapeutics Market Size
- Endometriosis Pipeline Analysis
- Endometriosis Market Size and Trends
- Endometriosis Market Opportunity (Current and forecasted)
Report Key Strengths
- Epidemiology‑Based (Epi‑based) Bottom‑up Forecasting
- Artificial Intelligence (AI)-enabled market research report
- 11-year forecast
- Endometriosis Market Outlook (North America, Europe, Asia-Pacific)
- Patient Burden Trends (by geography)
- Endometriosis Treatment Addressable Market (TAM)
- Endometriosis Competitve Landscape
- Endometriosis Major Companies Insights
- Endometriosis Price trends and Analogue Assessment
- Endometriosis Therapies Drug Adoption/Uptake
- Endometriosis Therapies Peak Patient Share analysis
Report Assessment
- Endometriosis Current Treatment Practices
- Endometriosis Unmet Needs
- Endometriosis Clinical Development Analysis
- Endometriosis Emerging Drugs Product Profiles
- Endometriosis Market Attractiveness
- Endometriosis Qualitative Analysis (SWOT and conjoint analysis)
FAQs
Market Insights
- What was the endometriosis market size, the market size by therapies, market share (%) distribution in 2025, and what would it look like by 2036? What are the contributing factors for this growth?
- What are the anticipated pricing variations among different geographies for the emerging therapies in the future?
- What can be the future treatment paradigm of endometriosis?
- What are the disease risks, burdens, and unmet needs of endometriosis? What will be the growth opportunities across the 7MM concerning the patient population with endometriosis?
- Who is the major future competitor in the market, and how will the competitors affect their market share?
- What are the current options for the treatment of endometriosis? What are the current guidelines for treating endometriosis in the US, Europe, and Japan?
Reasons to Buy
- The report will help in developing business strategies by understanding the latest trends and changing treatment dynamics driving the endometriosis market.
- Bottom up forecasting builds from the affected population to product forecasts, delivering a robust, data driven approach ideal for new therapies and novel classes.
- Insights on patient burden/disease, evolution in diagnosis, and factors contributing to the change in the epidemiology of the disease during the forecast years.
- Understand the existing market opportunities in varying geographies and the growth potential over the coming years.
- Identifying strong upcoming players in the market will help devise strategies to help get ahead of competitors.
- Detailed analysis and ranking of class-wise potential current and emerging therapies under the attribute analysis section to provide visibility around leading classes.
- To understand KOLs’ perspectives on the accessibility, acceptability, and compliance-related challenges of existing treatment to overcome barriers in the future.
- Detailed insights on the unmet needs of the existing market so that the upcoming players can strengthen their development and launch strategy.
- This Artificial Intelligence (AI) enabled report summarize and simplify complex datasets withing the report into clear, actionable insights for stakeholders, investors, and healthcare providers, enabling faster, data driven decisions.