Psoriasis Treatment Market Summary
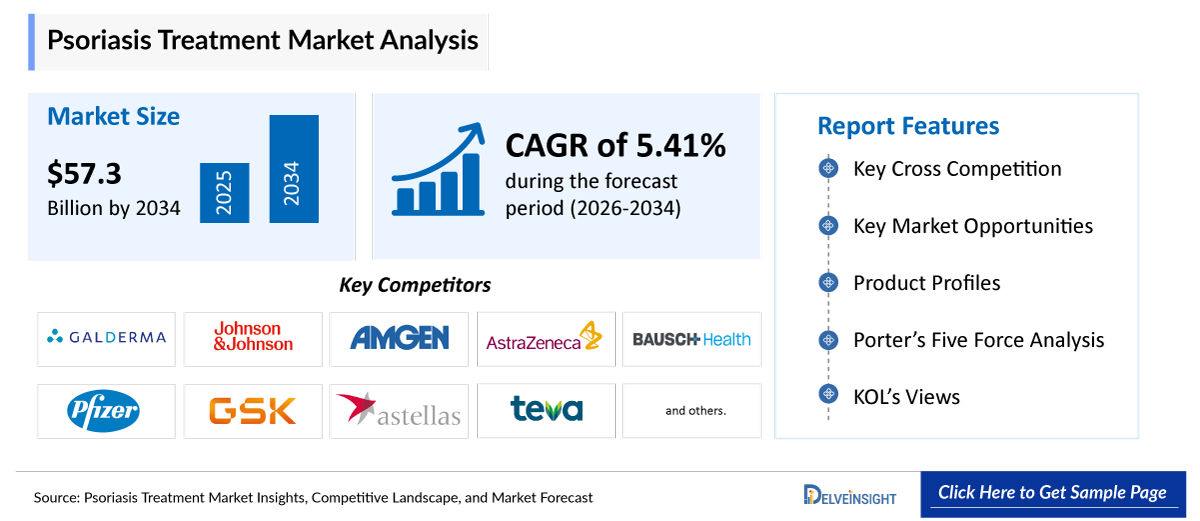
- The 7MM psoriasis market is expected to increase from USD 35,855.16 million in 2025 to USD 57,318.58 million by 2034, reflecting strong and sustained growth.
- The psoriasis treatment market is growing at a CAGR of 5.41% during the forecast period from 2026 to 2034.
- The leading Psoriasis companies operating in the psoriasis treatment market include Galderma, Johnson & Johnson Services, Inc., Amgen Inc., AstraZeneca, Bausch Health Companies Inc., Pfizer Inc., GSK plc., Astellas Pharma Inc., Teva Pharmaceutical Industries Ltd., Eisai Co., Ltd., Medexus Pharmaceuticals, Inc., AbbVie Inc., Novartis AG, and others.
Psoriasis Market and Epidemiology Analysis
- The Psoriasis market for psoriasis treatment is primarily driven by the rising 7MM prevalence of psoriasis, increasing awareness among patients and healthcare professionals, and improved diagnosis rates, which lead to earlier and more consistent treatment adoption. Growing demand for advanced and effective therapies, particularly biologics and targeted oral drugs, is significantly contributing to market expansion due to their improved efficacy and safety profiles compared to traditional treatments. Continuous research and development activities, along with the introduction of innovative therapies and biosimilars, are further enhancing treatment accessibility and affordability. Additionally, expanding healthcare infrastructure, increasing healthcare expenditure, and better access to dermatology services in emerging economies are supporting sustained market growth.
- North America is expected to dominate the psoriasis treatment market due to the high prevalence of psoriasis, strong awareness among patients, and early adoption of advanced therapies such as biologics and targeted oral drugs. The region benefits from well-established healthcare infrastructure, favorable reimbursement policies, and significant healthcare spending, which enable wider access to innovative and high-cost treatments. Additionally, the presence of leading pharmaceutical companies, continuous research and development activities, and frequent product approvals further strengthen market growth in North America.
- In the Psoriasis drug class segment of the psoriasis treatment market, the TNF-alpha inhibitors category is estimated to account for the largest market share in 2025.
Factors Impacting the Psoriasis Market Growth
Epidemiology and Patient Population
The size and growth of the patient population are fundamental to an indication market. An increase in disease prevalence or incidence directly drives demand for therapies. Factors such as aging populations, changing lifestyles, and genetic predispositions can significantly expand the addressable patient pool, thereby supporting market growth.
Innovation and Clinical Pipeline
Continuous advancements in treatment options and a strong pipeline of emerging therapies are key growth drivers. The introduction of novel drugs, biologics, and targeted therapies improves patient outcomes and creates new market opportunities, especially in areas with high unmet medical needs.
Regulatory Environment and Approvals
Regulatory policies and approval timelines play a critical role in determining how quickly new therapies enter the market. Favorable frameworks, such as accelerated approval pathways and orphan drug incentives, can boost growth, while stringent regulations or delays may limit market expansion.
Pricing, Reimbursement, and Market Access
The success of any therapy depends heavily on its affordability and accessibility. Supportive reimbursement policies and insurance coverage increase patient access and adoption, whereas high treatment costs and limited coverage can restrict market penetration.
Competitive Landscape and Healthcare Infrastructure
The presence of competing therapies, including generics and biosimilars, influences pricing and market share dynamics. At the same time, well-developed healthcare infrastructure ensures better access to diagnosis and treatment, enabling faster adoption of advanced therapies and contributing to overall market growth.
Request for unlocking the report of the @Psoriasis Treatment Market
Factors Contributing to the Growth of the Psoriasis Treatment Market
- Increasing prevalence and burden of psoriasis leading to a surge in psoriasis treatment: The increasing prevalence and growing burden of psoriasis worldwide are driving higher demand for effective treatment options. As more patients are diagnosed with this chronic autoimmune skin condition, the need for long-term management therapies, including topical, systemic, and biologic drugs, continues to rise, thereby supporting the overall growth of the psoriasis treatment market.
- Increasing advancement in treatment options is escalating the market of psoriasis treatment: Biologic therapies, targeted small molecules (like PDE4, TYK2 inhibitors), and novel systemic treatments have transformed the treatment landscape with higher efficacy and better safety profiles. This shift drives adoption and expands the market.
- Increase in product development activities among the key market players: The increase in product development activities among key market players is boosting the psoriasis treatment market by introducing innovative and more effective therapies, including advanced biologics and targeted oral drugs. Continuous research and development, along with a large number of clinical trials and new drug approvals, are expanding treatment options, improving patient outcomes, and strengthening market growth 7MMly.
Psoriasis Treatment Market Report Segmentation
This psoriasis treatment market report offers a comprehensive overview of the 7MM psoriasis treatment market, highlighting key trends, growth drivers, challenges, and opportunities. It covers detailed market segmentation by Drug Class (Immunosuppressants, Corticosteroids, TNF-alpha inhibitors, Interleukin Inhibitors, JAK Inhibitors, Vitamin Analogues (A & D), Other), Drug Type (Biologics and Small Molecule), Type (Plaque psoriasis, Multiple psoriasis, and Scalp & Body Psoriasis), Route of Administration (Oral, Topical, and Parenteral), and geography. The report provides valuable insights into the competitive landscape, regulatory environment, and market dynamics across major markets, including North America, Europe, and Asia-Pacific. Featuring in-depth profiles of leading industry players and recent product innovations, this report equips businesses with essential data to identify market potential, develop strategic plans, and capitalize on emerging opportunities in the rapidly growing psoriasis treatment market.
Psoriasis treatment refers to the range of medical therapies and management strategies used to control and reduce the symptoms of psoriasis, a chronic autoimmune skin disorder characterized by red, scaly, and inflamed skin patches. The treatment aims to slow down excessive skin cell growth, reduce inflammation, relieve itching and discomfort, and prevent flare-ups. Psoriasis management may include topical treatments (such as corticosteroids and vitamin D analogs), systemic medications (like methotrexate and cyclosporine), biologic therapies, oral targeted drugs, and phototherapy device, depending on the severity and type of the condition.
The market for psoriasis treatment is primarily driven by the rising 7MM prevalence of psoriasis, increasing awareness among patients and healthcare professionals, and improved diagnosis rates, which lead to earlier and more consistent treatment adoption. Growing demand for advanced and effective therapies, particularly biologics and targeted oral drugs, is significantly contributing to market expansion due to their improved efficacy and safety profiles compared to traditional treatments. Continuous research and development activities, along with the introduction of innovative therapies and biosimilars, are further enhancing treatment accessibility and affordability. Additionally, expanding healthcare infrastructure, increasing healthcare expenditure, and better access to dermatology services in emerging economies are supporting sustained market growth.
Get More Insights into the Report @Psoriasis Treatment Market
What are the latest psoriasis treatment market dynamics and trends?
The 7MM psoriasis treatment market is growing significantly, driven by a combination of psoriasis, increasing awareness among patients and healthcare professionals, and improved diagnosis rates, which lead to earlier and more consistent treatment adoption.
According to the data provided by the 7MM Psoriasis Atlas (2021-2026), an estimated 43 million (from 27 million to 91 million) people have psoriasis based on reporting physician/dermatologist diagnoses. Additionally, an estimated 102 million (from 65 million to 218 million) people have psoriasis based on self-report cases provided by individuals. The rising 7MM prevalence of psoriasis is a major factor driving the growth of the psoriasis treatment market, as the increasing number of diagnosed patients creates sustained demand for long-term and effective therapies. Psoriasis is a chronic autoimmune condition that requires continuous management, leading to repeated prescriptions and ongoing treatment adoption, which supports consistent market revenue.
Additionally, the increasing advancement in treatment options is significantly escalating the psoriasis treatment market by expanding therapeutic choices and improving outcomes for patients. Innovations in both biologic and small-molecule therapies are transforming the treatment landscape. For instance, in November 2024, UCB, a 7MM biopharmaceutical company, announced that the U.S. Food and Drug Administration (FDA) had approved BIMZELX® (bimekizumab-bkzx) for the treatment of adults with moderate-to-severe hidradenitis suppurativa (HS). BIMZELX is the first and only approved medicine designed to selectively inhibit interleukin 17F (IL-17F) in addition to interleukin 17A (IL-17A).
Furthermore, the presence of a strong pipeline and ongoing higher phase clinical trials is further expected to boost the market for psoriasis treatment. For instance, in December 2025, Takeda Pharmaceutical Company Limited announced positive topline results from two pivotal Phase 3 trials of zasocitinib (TAK-279), a next-generation oral TYK2 inhibitor, in adults with moderate-to-severe plaque psoriasis. The studies showed superiority over placebo for the primary endpoints (sPGA 0/1 and PASI 75) at Week 16, with significant improvement observed as early as Week 4 and continuing through Week 24. All ranked secondary endpoints, including PASI 90 and PASI 100, were also met, demonstrating strong efficacy for the once-daily oral therapy.
Thus, the factors mentioned above are expected to boost the overall market of psoriasis treatment during the forecast period.
However, the side effects and safety concerns, such as increased risk of infections or immune-related complications with systemic and biologic treatments, may restrict long-term use. The need for continuous or lifelong therapy can also affect patient adherence and increase treatment burden. Furthermore, stringent regulatory approval processes, variability in reimbursement policies across regions, and limited access to specialized dermatology care in developing countries may slow market expansion.
Psoriasis Treatment Market Segment Analysis
Psoriasis Treatment Market Drug Class (Immunosuppressants, Corticosteroids, TNF-alpha Inhibitors, Interleukin Inhibitors, JAK Inhibitors, Vitamin Analogues (A & D), and Other), Drug Type (Biologics and Small Molecule), Type (Plaque psoriasis, Multiple psoriasis, and Scalp & Body Psoriasis), Route of Administration (Oral, Topical, and Parenteral), and Geography (North America, Europe, Asia-Pacific, and Rest of the World)
By Drug Class: TNF-alpha Inhibitors Category Dominates the Market
In the drug class segment of the psoriasis treatment market, the TNF-alpha inhibitors category is estimated to account for the largest market share of 48% in 2025. The TNF-alpha inhibitor drug class has significantly boosted the overall psoriasis treatment market by providing highly effective systemic therapy for moderate-to-severe plaque psoriasis and related psoriatic conditions. As one of the earliest biologic classes introduced for psoriasis, TNF inhibitors such as Humira (adalimumab), manufactured by AbbVie Inc., Enbrel (etanercept), manufactured by Amgen Inc. (in collaboration with Pfizer Inc.), and Remicade (infliximab) developed by Janssen Pharmaceuticals demonstrated strong clinical efficacy in reducing inflammation, improving skin clearance, and enhancing patient quality of life. Their proven long-term safety data and extensive real-world evidence have supported widespread adoption by dermatologists 7MMly, contributing to substantial market growth. These therapies are especially important for patients who do not respond adequately to topical treatments or conventional systemic drugs, thereby expanding the eligible patient population for advanced biologic therapy. Additionally, the availability of biosimilars to major TNF-alpha inhibitors has improved affordability and access, further increasing treatment uptake across developed and emerging markets. The continued use of these established therapies, along with strong reimbursement support and 7MM commercialization, plays a key role in driving sustained revenue generation and overall expansion of the psoriasis treatment market.
By Drug Type: Biologics Category Dominates the Market
In the drug type segment of the psoriasis treatment market, biologics are projected to hold the largest market share of 55% in 2025, due to their high efficacy, targeted mechanism of action, and ability to manage moderate-to-severe cases that do not respond adequately to topical or traditional systemic therapies. Biologics work by specifically targeting immune system pathways involved in psoriasis, such as TNF-alpha, IL-17, and IL-23, leading to improved skin clearance, reduced inflammation, and better long-term disease control. Their strong clinical outcomes, including high PASI response rates and sustained remission, have made them the preferred treatment option among dermatologists for severe plaque psoriasis and psoriatic arthritis. Additionally, the growing number of approvals, expanding indications, and continuous innovation in next-generation biologics are increasing treatment adoption worldwide. The availability of biosimilars has further enhanced affordability and accessibility, particularly in emerging markets, thereby expanding the patient base. With rising disease prevalence, increasing awareness, improved reimbursement support in developed regions, and ongoing research and development activities, the biologics segment continues to drive substantial revenue growth and remains a key contributor to the overall expansion of the psoriasis treatment market.
Additionally, the increase in approvals and launches of biologic drug type of psoriasis is further boosting the overall market. For instance, in September 2025, Johnson & Johnson announced that the U.S. FDA had approved Tremfya (guselkumab) for the treatment of children aged six years and older weighing at least 40 kg with moderate-to-severe plaque psoriasis or active psoriatic arthritis. This approval made Tremfya the first and only IL-23 inhibitor approved for these pediatric indications and expanded on its earlier FDA approvals in adults in 2017 for plaque psoriasis and in 2020 for psoriatic arthritis.
Thus, the factors mentioned above are expected to boost the market of biologics therby escalating the overall market of psoriasis treatment.
By Type: Plaque Psoriasis Category Dominates the Market
In the type segment of the psoriasis treatment market, plaque psoriasis is projected to hold the largest market share of 58% in 2025. Plaque psoriasis is the most common form of psoriasis, accounting for the majority of total cases, and therefore plays a major role in driving the overall psoriasis treatment market. Its high 7MM prevalence, chronic nature, and recurring flare-ups create sustained demand for long-term therapy. Patients with moderate-to-severe plaque psoriasis often require advanced treatments such as systemic drugs and biologics when topical therapies are insufficient, which significantly increases market value. The growing adoption of biologics and targeted oral therapies for plaque psoriasis has further expanded treatment options and improved clinical outcomes, leading to higher therapy uptake. Additionally, increasing awareness, improved diagnosis rates, and rising healthcare access are enabling more patients to seek medical care. Since plaque psoriasis typically requires continuous management to control symptoms and prevent complications, it generates recurring prescriptions and long-term revenue, thereby strongly contributing to the overall growth of the psoriasis treatment market.
By Route of Administration: Oral Category Driving the Market
The oral route of administration is significantly boosting the overall psoriasis treatment market by offering greater convenience, improved patient compliance, and an alternative to injectable biologics. Oral therapies are generally preferred by patients due to their ease of use, non-invasive nature, and suitability for long-term treatment, especially in moderate-to-severe cases. The introduction of advanced oral small-molecule drugs, such as PDE4 inhibitors and TYK2 inhibitors, has expanded treatment options beyond traditional systemic therapies, improving efficacy while maintaining manageable safety profiles. Oral medications also reduce the need for clinic visits and self-injections, making them more accessible and acceptable to a broader patient population. Additionally, ongoing research and development of next-generation oral targeted therapies are further strengthening market growth by increasing innovation and expanding therapeutic choices in psoriasis management.
Psoriasis Treatment Market Regional Analysis
North America Psoriasis Treatment Market Trends
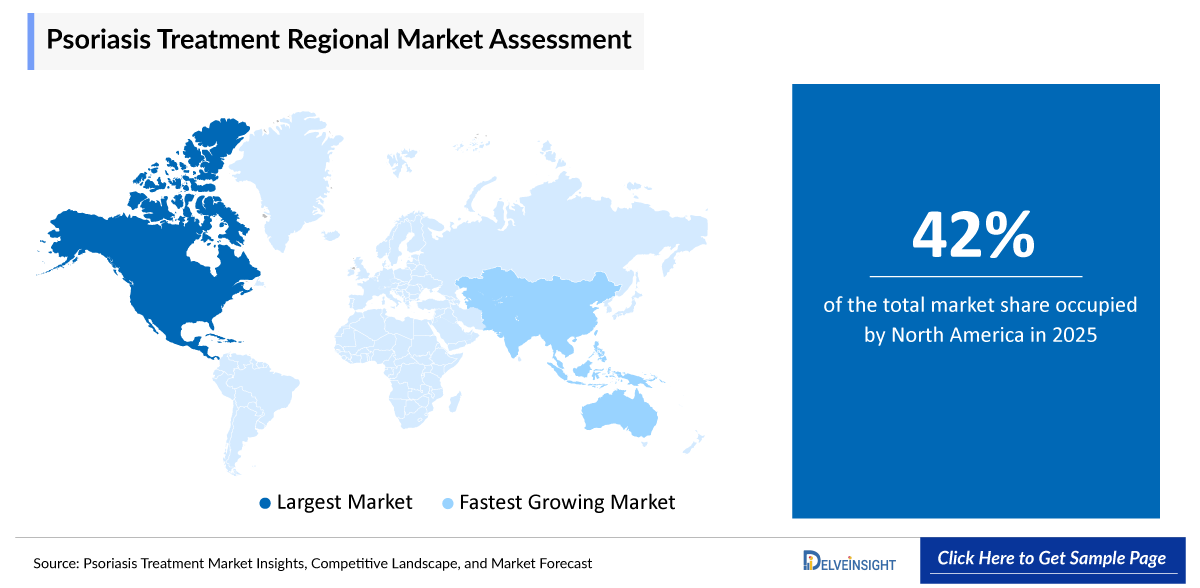
North America is poised to secure the largest share of 42% in the 7MM psoriasis treatment market in 2025 due to the high prevalence of psoriasis, strong awareness among patients, and early adoption of advanced therapies such as biologics and targeted oral drugs. The region benefits from well-established healthcare infrastructure, favorable reimbursement policies, and significant healthcare spending, which enable wider access to innovative and high-cost treatments. Additionally, the presence of leading pharmaceutical companies, continuous research and development activities, and frequent product approvals further strengthen market growth in North America.
According to the data provided by the National Psoriasis Foundation/USA (199-2026), more than 8 million Americans have psoriasis. Psoriasis is a chronic autoimmune condition that requires continuous management, leading to repeated prescriptions and ongoing treatment adoption, which supports consistent market revenue.
Additionally, the Increasing awareness of psoriasis in North America has played a key role in boosting the psoriasis treatment market by encouraging more patients to seek diagnosis and appropriate care, while also driving earlier treatment initiation and broader acceptance of advanced therapies. Campaigns such as the National Psoriasis Awareness Month, which highlights the experiences of millions living with the condition and educates the public on its impact beyond just skin symptoms, have helped reduce stigma and increase understanding of available treatments. Similarly, initiatives like the ‘So, Have You Found It?’ nationwide campaign have worked to empower people with plaque psoriasis to recognize their condition and engage in meaningful conversations with dermatologists about treatment options. Patient advocacy and education by organizations such as the National Psoriasis Foundation also support outreach, research, and professional education, which enhances awareness among both patients and healthcare providers. As a result, more individuals are entering clinical pathways for effective therapies, including topical agents, biologics, and newer oral options, ultimately expanding market uptake and contributing to robust growth in the North American psoriasis treatment market.
Furthermore, the increasing product approvals across the regions are further escalating the overall psoriasis treatment market. For instance, in May, 2025, the FDA approved Arcutis Biotherapeutics' ZORYVE (roflumilast) topical foam 0.3% for treating plaque psoriasis, including scalp and body, in adults and adolescents aged 12+. This steroid-free, once-daily PDE4 inhibitor provides a new, non-steroidal, long-term treatment option for psoriasis in hair-bearing and sensitive areas.
Thus, the factors mentioned above are expected to boost the overall market of psoriasis treatment across the region.
Europe Psoriasis Treatment Market Trends
Europe is increasingly poised to play a leading role in the overall psoriasis treatment market due to several converging factors that strengthen both demand and innovation across the region. The European psoriasis drugs market is expected to grow substantially over the coming decade, driven by a robust healthcare infrastructure, high disease prevalence, extensive clinical research capabilities, and strong reimbursement policies in key markets such as Germany, France, the UK, and other Western European countries.
Moreover, the adoption of biosimilars and cost-effective therapies supported by nationalized healthcare systems and proactive regulatory frameworks improves accessibility for patients, further accelerating regional growth. Healthcare investment, increasing awareness of psoriasis management, and a pipeline rich in innovative treatments also contribute to Europe’s growing influence on the 7MM stage. European markets are witnessing both steady uptake of biologics and increasing research into novel therapies, positioning the region to capture a larger share of the 7MM psoriasis treatment landscape.
Additionally, the increasing product development activities are further boosting the overall market of the psoriasis treatment market. For instance, in December 2025, the European Commission expanded approval of Tremfya® (guselkumab) to include additional indications, such as pediatric plaque psoriasis from age 6, enhancing its therapeutic reach across age groups.
Thus, the factors mentioned are expected to boost the market of psoriasis treatment market in Europe.
Asia-Pacific Psoriasis Treatment Market Trends
The Asia-Pacific region is rapidly emerging as one of the fastest-growing and most influential markets in the 7MM psoriasis treatment landscape, driven by a combination of demographic, healthcare, and economic factors. A significant increase in the prevalence of psoriasis across densely populated countries such as China, India, Japan, and South Korea is creating a substantial patient pool that needs effective therapies. Growing awareness of the disease, improved diagnostic capabilities, and rising healthcare expenditures are enabling earlier diagnosis and better access to both traditional and advanced treatment options. Governments in the region are also investing in healthcare infrastructure and research, and supportive policies are helping expand the availability of biologic and targeted therapies that were previously limited to Western markets. Additionally, expanding reimbursement for innovative treatments is broadening patient access, while a shift toward biologics and biosimilars, often at competitive price points, further fuels adoption. Collectively, these trends have helped position Asia-Pacific as a major growth driver in the psoriasis treatment market, with projections showing it will outpace other regions in terms of compound annual growth in the coming years.
Who are the major players in the psoriasis treatment market?
The following are the leading companies in the psoriasis treatment market. These companies collectively hold the largest market share and dictate industry trends.
- Galderma
- Johnson & Johnson Services, Inc.
- Amgen Inc.
- AstraZeneca
- Bausch Health Companies Inc.
- Pfizer Inc.
- GSK plc.
- Astellas Pharma Inc.
- Teva Pharmaceutical Industries Ltd.
- Eisai Co., Ltd.
- Medexus Pharmaceuticals, Inc.
- AbbVie Inc.
- Novartis AG
- Others
How is the competitive landscape shaping the psoriasis treatment market?
The competitive landscape of the 7MM psoriasis treatment market is shaped by a mix of large multinational pharmaceutical companies, specialized dermatology firms, and emerging biotech innovators, creating both concentrated leadership and dynamic competition. Major established players such as Pfizer, AbbVie, Novartis, Johnson & Johnson, AstraZeneca, Eli Lilly, Merck & Co., and LEO Pharma dominate the market with well-established biologics and systemic therapies that capture substantial shares of the overall revenue due to strong brand recognition, extensive clinical data, and broad geographic reach. These large firms tend to maintain market leadership through continued investment in advanced biologic agents—including IL-17 and IL-23 inhibitors—which represent a significant portion of market sales and patient adoption.
Despite this concentration at the top, competitive intensity is increasing as emerging biotech companies and smaller firms introduce novel mechanisms of action and alternative delivery formats, particularly in the oral small-molecule and TYK2 inhibitor classes. For example, companies like Dermavant Sciences, Celltrion, and Sun Pharmaceutical are expanding their portfolios alongside traditional leaders, while new clinical advancements (such as experimental therapies with longer dosing intervals or differentiated efficacy) are challenging incumbents and attracting investor interest.
Overall, while a handful of top pharmaceutical giants currently exert considerable influence, resulting in moderate to high market concentration among leading biologics, ongoing innovation and pipeline developments are gradually diffusing competitive power, driving both broader therapeutic choice and potential market share shifts in the coming decade
Recent Developmental Activities in the Psoriasis Treatment Market
- In December 2025, the European Commission approved TREMFYA® (guselkumab) for the treatment of children with plaque psoriasis, marking the first paediatric indication for an IL-23 inhibitor.
- In December 2025, Takeda Pharmaceutical Company Limited announced positive topline results from two pivotal Phase 3 trials of zasocitinib (TAK-279), a next-generation oral TYK2 inhibitor, in adults with moderate-to-severe plaque psoriasis.
- In September 2025, Johnson & Johnson announced that the U.S. FDA had approved Tremfya (guselkumab) for the treatment of children aged six years and older weighing at least 40 kg with moderate-to-severe plaque psoriasis or active psoriatic arthritis. This approval made Tremfya the first and only IL-23 inhibitor approved for these pediatric indications and expanded on its earlier FDA approvals in adults in 2017 for plaque psoriasis and in 2020 for psoriatic arthritis.
- In July 2025, Johnson & Johnson announced the submission of a New Drug Application (NDA) to the U.S. Food and Drug Administration (FDA) seeking the first approval of icotrokinra, a first-in-class investigational targeted oral peptide that selectively blocks the IL-23 receptor for the treatment of adults and pediatric patients 12 years of age and older with moderate to severe plaque psoriasis (PsO).
- In May 2025, the FDA approved Arcutis Biotherapeutics' ZORYVE (roflumilast) topical foam 0.3% for treating plaque psoriasis, including scalp and body, in adults and adolescents aged 12+. This steroid-free, once-daily PDE4 inhibitor provides a new, non-steroidal, long-term treatment option for psoriasis in hair-bearing and sensitive areas.
- In November 2024, UCB, a 7MM biopharmaceutical company, announced that the U.S. Food and Drug Administration (FDA) had approved BIMZELX® (bimekizumab-bkzx) for the treatment of adults with moderate-to-severe hidradenitis suppurativa (HS). BIMZELX is the first and only approved medicine designed to selectively inhibit interleukin 17F (IL-17F) in addition to interleukin 17A (IL-17A).
|
Report Metrics |
Details |
|
Study Period |
2023 to 2034 |
|
Base Year |
2025 |
|
Forecast Period |
2026 to 2034 |
|
Psoriasis Treatment Market CAGR (2026-2034) |
5.41% |
|
Key Companies in the Psoriasis Treatment Market |
Galderma, Johnson & Johnson Services, Inc., Amgen Inc., AstraZeneca, Bausch Health Companies Inc., Pfizer Inc., GSK plc., Astellas Pharma Inc., Teva Pharmaceutical Industries Ltd., Eisai Co., Ltd., Medexus Pharmaceuticals, Inc., AbbVie Inc., Novartis AG, and others. |
|
Psoriasis Treatment Market Segments |
by Drug Class, by Drug Type, by Type, by Route of Administration, and by Geography |
|
Psoriasis Treatment Regional Scope |
North America, Europe, Asia Pacific, Middle East, Africa, and South America |
|
Psoriasis Treatment Country Scope |
U.S., Canada, Mexico, Germany, United Kingdom, France, Italy, Spain, China, Japan, India, Australia, South Korea, and key Countries |
Psoriasis Treatment Market Segmentation
- Psoriasis Treatment by Product Type Exposure
- Immunosuppressants
- Corticosteroids
- TNF-alpha Inhibitors
- Interleukin Inhibitors
- JAK Inhibitors
- Vitamin Analogues (A & D)
- Other
- Psoriasis Treatment Drug Type Exposure
- Biologics
- Small Molecule
- Psoriasis Treatment Type Exposure
- Plaque psoriasis
- Multiple psoriasis
- Scalp & Body Psoriasis
- Psoriasis Treatment Route of Administration Exposure
- Oral
- Topical
- Parenteral
- Psoriasis Treatment Geography Exposure
- North America Psoriasis Treatment Market
- United States Psoriasis Treatment Market
- Canada Psoriasis Treatment Market
- Mexico Psoriasis Treatment Market
- Europe Psoriasis Treatment Market
- United Kingdom Psoriasis Treatment Market
- Germany Psoriasis Treatment Market
- France Psoriasis Treatment Market
- Italy Psoriasis Treatment Market
- Spain Psoriasis Treatment Market
- Rest of Europe Psoriasis Treatment Market
- Asia-Pacific Psoriasis Treatment Market
- China Psoriasis Treatment Market
- Japan Psoriasis Treatment Market
- India Psoriasis Treatment Market
- Australia Psoriasis Treatment Market
- South Korea Psoriasis Treatment Market
- Rest of Asia-Pacific Psoriasis Treatment Market
- Rest of the World Psoriasis Treatment Market
- South America Psoriasis Treatment Market
- Middle East Psoriasis Treatment Market
- Africa Psoriasis Treatment Market
- North America Psoriasis Treatment Market
Psoriasis Treatment Market Recent Industry Trends and Milestones (2022-2026)
|
Category |
Key Developments |
|
Psoriasis Treatment Regulatory Approvals |
TREMFYA® was approved by the European Commission. Tremfya was approved by the FDA for children, ORYVE (roflumilast) was approved by the FDA, and BIMZELX® (bimekizumab-bkzx) was approved by the FDA. |
|
Company Strategy |
AbbVie continues to fortify its dermatology portfolio by focusing on next-generation biologics and strategic acquisitions. Lilly is actively expanding its psoriasis and autoimmune therapy footprint through innovative partnerships and internal R&D. Takeda is pursuing AI-driven innovation with its experimental oral psoriasis drug zasocitinib, developed using artificial intelligence. |
|
Emerging Technology |
Advanced Nanotechnology-Based Delivery Systems, Smart Microneedle Platforms with AI Integration, Personalized and Precision Medicine, Gene and Molecular-Level Therapies, and others |
Impact Analysis
U.S. Tariff Impact Analysis on Psoriasis Treatment Market:
The U.S. tariff impact analysis on the psoriasis treatment market indicates that tariffs on imported pharmaceuticals and active pharmaceutical ingredients (APIs) could increase manufacturing and procurement costs, particularly for biologics and specialty drugs that rely on 7MM supply chains. Since many psoriasis therapies are produced or sourced from Europe and Asia, additional import duties may raise overall drug prices if companies pass costs to payers and patients, potentially affecting treatment accessibility and reimbursement dynamics. Tariffs may also disrupt supply chains, delay product availability, and encourage manufacturers to shift toward U.S.-based production to mitigate financial risk. While companies with strong domestic manufacturing capabilities may gain a competitive advantage, higher operational costs and pricing pressures could slow adoption of innovative therapies and reshape competitive positioning within the U.S. psoriasis treatment market.
How This Analysis Helps Clients
- Cost Management: By understanding the tariff landscape, clients can anticipate cost increases and adjust pricing strategies accordingly, ensuring profitability.
- Supply Chain Optimization: Clients can identify alternative sourcing options and diversify their supply chains to reduce dependency on high-tariff regions, enhancing resilience.
Regulatory Navigation: Expert guidance on navigating the evolving regulatory environment helps clients maintain compliance and avoid potential legal challenges. - Strategic Planning: Insights into tariff impacts enable clients to make informed decisions about manufacturing locations, partnerships, and market entry strategies.
Startup Funding & Investment Trends
|
Company Name |
Total Funding |
Main Products |
Stage of Development |
Core Technology |
|
Alumis Inc. |
$259 million |
ESK-001 — an oral TYK2 inhibitor targeting plaque psoriasis. |
Series C |
Small-molecule TYK2 inhibition to regulate immune signaling in psoriasis; advancing into Phase 3 clinical trials. |
Key takeaways from the Psoriasis Treatment market report study
- Market size analysis for the current psoriasis treatment market size (2025), and market forecast for 8 years (2026 to 2034)
- Top key product/technology developments, mergers, acquisitions, partnerships, and joint ventures happened over the last 3 years.
- Key companies dominating the psoriasis treatment market.
- Various opportunities available for the other competitors in the psoriasis treatment market space.
- What are the top-performing segments in 2025? How these segments will perform in 2034?
- Which are the top-performing regions and countries in the current psoriasis treatment market scenario?
- Which are the regions and countries where companies should have concentrated on opportunities for the psoriasis treatment market growth in the future?