Dravet Syndrome Market Summary
- As per DelveInsight, the Dravet syndrome Market is expected to expand at a healthy growth rate during the forecast period (2026–2036), owing to the launch of new therapies in the market and the rise in the number of cases.
- Dravet syndrome, previously referred to as Severe Myoclonic Epilepsy of Infancy (SMEI), is a rare, lifelong form of childhood epilepsy that is typically resistant to treatment. It is classified as a developmental and epileptic encephalopathy, where persistent, difficult-to-control seizures contribute to significant developmental delays and cognitive impairment.
- In Dravet syndrome, seizures typically begin between 2 and 15 months of age. About 3–8% of infants with seizures in the first year may be diagnosed with Dravet syndrome. Early episodes are often fever-triggered, prolonged (15–30 minutes or more), and may present as febrile convulsive status epilepticus requiring urgent care.
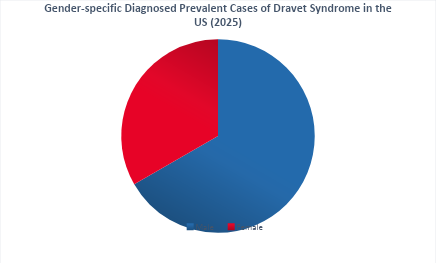
- Dravet syndrome demonstrates a higher prevalence in males compared with females, with a reported male-to-female ratio of approximately 2:1.
- Approximately 85% of patients with Dravet syndrome have a mutation or deletion in the SCN1A gene, which encodes a voltage-gated sodium channel protein.
- Mortality affects up to 20% of children and adolescents with Dravet syndrome before adulthood, primarily due to sudden unexpected death in epilepsy, prolonged seizures, severe infections, or seizure-related accidents.
- Challenges in diagnosing Dravet syndrome include overlap of symptoms with other epilepsy syndromes, progressive evolution of clinical features that delays early recognition, and genetic heterogeneity, as not all patients carry an SCN1A mutation. Timely and accurate diagnosis is further hindered by limited access to specialized genetic testing and low clinical awareness among healthcare providers.
- Dravet syndrome is managed with anti-seizure combination therapy, mainly fenfluramine, cannabidiol, stiripentol, valproate, and clobazam, with avoidance of sodium-channel blockers. Management also includes trigger control, rescue medications for status epilepticus, and long-term supportive care (developmental, behavioral, and safety support).
- Currently, EPIDIOLEX (Jazz Pharmaceuticals), DIACOMIT (Biocodex), FINTEPLA (UCB/Zogenix), and others are approved medications offering treatment options for patients with Dravet syndrome.
- The key therapies for Dravet syndrome include zorevunersen (STK-001) (Stoke Therapeutics/Biogen), ETX101 (Encoded Therapeutics), bexicaserin (LP352) (Longboard Pharmaceuticals), and others.
- Dravet syndrome requires disease-modifying SCN1A-targeted therapies beyond symptomatic seizure control, along with comprehensive management of neurodevelopmental comorbidities and safer, lower-burden treatment options with fewer side effects and drug interactions.
Dravet Syndrome Market Size and Forecast in the 7MM
- 2025 Projected Dravet Syndrome Market Size: USD XXX million
- 2036 Projected Dravet Syndrome Market Size: USD XXX million
- Dravet syndrome Growth Rate (2026–2036): XX% CAGR
DelveInsight's ‘Dravet Syndrome – Market Insights, Epidemiology and Market Forecast – 2036’ report delivers an in-depth understanding of Dravet syndrome, historical and forecasted epidemiology, as well as the Dravet syndrome market trends in the United States, EU4 (Germany, Spain, Italy, and France), the United Kingdom, and Japan.
The Dravet syndrome market report delivers a comprehensive analysis of the current treatment landscape, including standards of care, clinical practices, and evolving therapeutic algorithms. It evaluates Dravet syndrome patient burden trends, revenue & market share dynamics, peak patient share & therapy uptake analysis, and provides an in-depth market size assessment, and growth rate projections (Historical & Forecast 2022–2036) across global regions. The report highlights key unmet medical needs in Dravet syndrome and maps the competitive and clinical landscape to uncover high‑value opportunities, providing a clear outlook on future market growth potential.
|
Study Period |
2022–2036 |
|
Historical Year |
2022–2025 |
|
Forecast Period |
2026–2036 |
|
Base Year |
2026 |
|
Geographies Covered |
|
|
Dravet Syndrome Market CAGR (Study period/Forecast period) |
XX% (2026 ̶ 2036) |
|
Dravet Syndrome Epidemiology Segmentation Analysis |
Patient Burden Assessment
|
|
Dravet Syndrome Companies |
|
|
Dravet Syndrome Therapies |
|
|
Dravet Syndrome Market |
Segmented by
|
|
Analysis |
|
Key Factors Driving the Dravet Syndrome Market
Rising Dravet Syndrome Prevalence
The increasing prevalence of Dravet syndrome, particularly among children aged ≤16 years, is a key driver of market growth. In the US, there were approximately ~22,600 diagnosed prevalent cases in 2025, which are expected to increase further by 2036, driven by improved diagnosis, greater awareness, and advancements in genetic testing.
Rising Opportunities in Targeted Therapies in Dravet Syndrome
Emerging therapies in Dravet syndrome are increasingly focused on precision and mechanism-based approaches, offering improved seizure control, better safety, and reduced treatment burden. Growing clinical evidence supporting gene therapies, antisense oligonucleotides, and novel neuromodulators highlights a significant opportunity for drug developers to target the underlying SCN1A dysfunction and related neuronal pathways. This shift toward disease-modifying and targeted treatments is expected to drive future innovation and market growth.
Emerging Dravet Syndrome Competitive Landscape
Some of the Dravet syndrome drugs in clinical trials include zorevunersen (STK-001) (Stoke Therapeutics), ETX101 (Encoded Therapeutics), bexicaserin (LP352) (Longboard Pharmaceuticals), and others.
Dravet Syndrome Understanding and Treatment Algorithm
Dravet Syndrome Overview and Diagnosis
Dravet syndrome is a rare, severe form of genetic epilepsy that typically begins in infancy, often within the first year of life. It is primarily caused by mutations in the SCN1A gene, leading to dysfunction in neuronal sodium channels. The condition is characterized by prolonged, treatment-resistant seizures along with developmental delay, cognitive impairment, and behavioral issues. Patients often experience worsening symptoms over time, including motor and speech difficulties. Due to its complexity, Dravet syndrome requires lifelong, multidisciplinary management.
Dravet Syndrome Diagnosis
Diagnosis is primarily based on clinical presentation, including the onset of prolonged febrile seizures in infancy and evolving seizure types. Genetic testing (especially for SCN1A mutations) is key to confirmation, although not all patients test positive. EEG and MRI may support diagnosis, but are often normal in early stages, making early identification challenging.
Further details are provided in the report.
Dravet Syndrome Treatment
Treatment focuses on seizure control using a combination of anti-seizure medications, including disease-specific therapies such as fenfluramine, cannabidiol, and stiripentol, along with standard agents like valproate and clobazam. Certain drugs, particularly sodium channel blockers, are avoided as they can worsen seizures. Rescue therapies (e.g., benzodiazepines) are used for prolonged seizures or status epilepticus. In some cases, non-pharmacological approaches such as the ketogenic diet may be beneficial. Management also includes multidisciplinary supportive care to address developmental, behavioral, and cognitive comorbidities.
Further details related to country-based variations are provided in the report.
Dravet Syndrome Unmet Needs
The section “unmet needs of Dravet syndrome” outlines the critical gaps between the current state of patient care, diagnosis, and the ideal & effective management of the disease. It highlights the obstacles experienced by patients, clinicians, and researchers and identifies potential solutions for future progress.
- No curative or disease-modifying therapies targeting SCN1A
- Refractory seizures with many patients remaining poorly controlled despite available treatments
- High burden of cognitive and behavioral comorbidities
- Safety issues with current treatments, and others…..
Comprehensive unmet needs insights in Dravet syndrome and their strategic implications are provided in the full report.
Dravet Syndrome Epidemiology
Key Findings from Dravet Syndrome Epidemiological Analysis and Forecast
- The total diagnosed prevalence of Dravet syndrome in the 7MM comprised ~25,600 cases in 2025.
- In the 7MM, the US accounted for the highest share (~50%) of diagnosed prevalent cases of Dravet syndrome in 2025.
- As per secondary analysis, the prevalence of Dravet syndrome is estimated to range from 1 in 20,000 to 1 in 40,000 individuals.
- Dravet syndrome is a severe infantile-onset epileptic encephalopathy associated with mutations in the sodium channel alpha 1 subunit gene SCN1A in about 80–90% of cases.
- An analysis reveals notable age-related disparities in Dravet syndrome prevalence in Spain. Pediatric patients exhibit an average rate of 1.1 cases per million inhabitants, while adults show a significantly lower rate of 0.5 cases per million. This underscores the critical need for targeted diagnostic and treatment strategies across different age groups.
Numbers are subject to change with report updation, clinical information updates, etc.
Dravet Syndrome Drug Chapters & Competitive Analysis
The Dravet syndrome drug chapter provides a detailed, market-focused review of approved therapies and the emerging pipeline across Phase I–III clinical trials. It covers the mechanism of action, clinical trial data, regulatory approvals, patents, collaborations, and strategic partnerships for each therapy, along with their advantages, limitations, and recent developments. This section offers critical insights into the Dravet syndrome treatment landscape, supporting market assessment, competitive analysis, and growth forecasting for the Dravet syndrome therapeutics market.
Approved Therapies for Dravet Syndrome
Cannabidiol (EPIDIOLEX/EPIDYOLEX): Jazz Pharmaceuticals
EPIDIOLEX (cannabidiol), known as EPIDYOLEX in the European Union (EU) and the United Kingdom, is a prescription, plant-derived cannabis-based medicine administered as an oral solution. In the United States, EPIDIOLEX is indicated for the treatment of seizures associated with Dravet syndrome in patients 1 year of age and older. In the EU and UK, EPIDIOLEX is indicated for use as adjunctive therapy of seizures associated with Dravet syndrome, in conjunction with clobazam, for patients 2 years of age and older.
Fenfluramin (FINTEPLA): UCB/Zogenix
FINTEPLA is indicated for the treatment of seizures associated with Dravet syndrome in patients 2 years of age and older. Due to risks of valvular heart disease (VHD) and pulmonary arterial hypertension (PAH), it is available only through a restricted distribution program under REMS. FINTEPLA is a low-dose fenfluramine oral liquid solution. Its exact mechanism of action is not fully understood, but it is known to increase extracellular serotonin levels via interaction with serotonin transporter proteins, while also exhibiting agonist activity at serotonin 5-HT2 receptors. It is also approved in the European Union and Japan.
|
Dravet Syndrome Marketed/Approved Therapies | ||||||
|
Drug/Therapy |
Company |
Indication |
Molecule Type |
RoA |
MoA |
Marketed Region |
|
Cannabidiol (EPIDIOLEX/ EPIDYOLEX) |
Jazz Pharmaceuticals |
Dravet syndrome |
Small molecule |
Oral |
Anticonvulsant effects through interaction with cannabinoid receptors |
US: 2018 EU: 2019 |
|
Fenfluramin (FINTEPLA) |
UCB/Zogenix |
Dravet syndrome |
Small molecule |
Oral |
5-HT2B receptor agonist |
US: 2020 EU: 2020 JP: 2022 |
|
Stiripentol (DIACOMIT) |
BIOCODEX |
Seizures associated with Dravet syndrome |
Small molecule |
Oral |
Target gamma-aminobutyric acid (GABA)A receptor |
US: 2018 |
Note: Detailed marketed therapies assessment of therapies will be provided in the final report.
Dravet Syndrome Pipeline Analysis
Zorevunersen (STK-001): Stoke Therapeutics and Biogen
Zorevunersen (STK-001) is an investigational new medicine for the treatment of Dravet syndrome currently being evaluated in ongoing clinical trials. STK-001 is designed to upregulate NaV1.1 protein expression by leveraging the non-mutant (wild-type) copy of the SCN1A gene to restore physiological NaV1.1 levels, thereby reducing both the occurrence of seizures and significant non-seizure comorbidities. The FDA has also granted zorevunersen a rare pediatric disease designation and Breakthrough Therapy Designation for the treatment of Dravet syndrome with a confirmed mutation not associated with gain-of-function in the SCN1A gene.
ETX101: Encoded Therapeutics
Encoded is developing ETX101, a potential one-time, disease-modifying gene regulation therapy for SCN1A+ Dravet syndrome. ETX101, Encoded’s lead program, is specifically designed to address the underlying cause of Dravet syndrome, the most common developmental and epileptic encephalopathy. ETX101 is a cell-selective gene therapy in development to potentially address the full range of seizure, cognitive, behavioral, developmental, and motor manifestations of Dravet syndrome. As per the company’s pipeline, the drug is in early clinical trials.
|
Comparison of Emerging Drugs Under Development | |||||||
|
Drug Name |
Company |
Highest Phase |
Indication |
RoA |
MoA |
Molecule Type |
Anticipated Launch in the US |
|
Zorevunersen (STK-001) |
Stoke Therapeutics and Biogen |
III |
Dravet syndrome with SCN1A |
Intrathecal |
Targeting SCN1A gene |
Antisense oligonucleotide |
Information is available in the full report |
|
Bexicaserin (LP352) |
Longboard Pharmaceuticals |
III |
Dravet syndrome |
Oral |
5-hydroxytryptamine 2c receptor superagonist |
Small Molecule |
Information is available in the full report |
|
Clemizole Hydrochloride (EPX-100) |
Harmony Biosciences |
III |
Dravet syndrome |
Oral |
Targeting central 5HT-2 serotonin receptors |
Small Molecule |
Information is available in the full report |
|
ETX101 |
Encoded Therapeutics |
I/II |
SCN1A-Positive Dravet syndrome |
Intracerebroventricular |
Targeting SCN1A gene |
Gene therapy |
Information is available in the full report |
|
Note: Launch insights are provisional and may change with future report updates or the occurrence of major key catalysts. | |||||||
Note: A detailed emerging therapies assessment will be provided in the final report
Dravet Syndrome Key Players, Market Leaders, and Emerging Companies
- Stoke Therapeutics
- Biogen
- Longboard Pharmaceuticals
- Harmony Biosciences
- Encoded Therapeutics
- Jazz Pharmaceuticals
- UCB
- Zogenix
- BIOCODEX, and others
Dravet Syndrome Drug Updates
- In April 2026, Jazz Pharmaceuticals presented research on EPIDIOLEX (cannabidiol) Oral Solution on Dravet syndrome at the 2026 American Academy of Neurology Annual Meeting.
- In March 2026, Stoke Therapeutics and Biogen reported in The New England Journal of Medicine that their investigational RNA therapy zorevunersen showed early evidence of potential disease modification in Dravet syndrome, based on Phase I/IIa and ongoing extension study results.
- In March 2026, Encoded Therapeutics announced the successful completion of its Initial Comprehensive Multidisciplinary Regenerative Medicine Advanced Therapy (RMAT) meeting with the US Food and Drug Administration (FDA), and alignment on the pivotal study design supporting the company’s planned BLA submission for ETX101, an AAV9-based gene regulation therapy designed as a one-time, disease-modifying treatment for SCN1A+ Dravet syndrome.
- As per the January 2026 JP Morgan presentation, the Phase III EMPEROR study for Zorevunersen is on track, with enrollment of 150 patients expected by Q2 2026, ongoing recruitment in younger patients, and data readout planned for mid-2027 to support a rolling NDA submission in H1 2027.
- In January 2026, Encoded Therapeutics announced that the US FDA had granted Breakthrough Therapy Designation to ETX101 for the treatment of SCN1A+ Dravet syndrome.
- In December 2025, Encoded Therapeutics announced positive interim results from its ongoing POLARIS clinical development program evaluating ETX101 in children with SCN1A+ Dravet syndrome.
Drug Class Insights
Dravet Syndrome Market Outlook
The market outlook for Dravet syndrome is progressively expanding, driven by high unmet medical needs and continuous therapeutic advancements.
Current treatment is largely symptomatic, with first-line therapies such as clobazam and valproic acid widely used due to their broad efficacy. Second-line options like stiripentol and adjunctive approaches (e.g., ketogenic diet) add to treatment flexibility, while later-line therapies, including levetiracetam and device-based interventions such as Vagus Nerve Stimulation, support refractory patients. Despite these options, most patients remain inadequately controlled, reinforcing demand for better therapies.
The market is witnessing steady growth due to increasing diagnosis rates, improved awareness, and the introduction of newer targeted drugs (e.g., cannabidiol- and fenfluramine-based therapies). Pharmaceutical companies are actively investing in pipeline innovations, including gene-targeted and disease-modifying approaches, which are expected to significantly reshape the future landscape. However, challenges persist, including heterogeneous treatment response, safety concerns, high treatment burden, and the lack of curative or disease-modifying therapies. Additionally, contraindicated sodium channel–blocking drugs limit therapeutic choices.
Overall, the launch of first-in-class therapies, improved diagnostic approaches, and increasing disease awareness are expected to drive steady growth in the 7MM Dravet syndrome market from 2022–2036, with strong commercial implications for both marketed products and emerging pipelines.
- The Dravet syndrome market is evolving as treatment focus shifts from broad-spectrum biologics to more targeted therapies, with JAK inhibitors and IL-targeted agents driving adoption due to their favorable efficacy, safety, and expanded pediatric indications.
- Regional market dynamics show the US leading in revenue, while Japan and EU markets demonstrate steady growth, reflecting increasing diagnosis rates, broader pediatric access, and rising adoption of novel mechanisms.
- The entry of mid- to late-stage candidates such as Zorevunersen (STK-001), Bexicaserin (LP352), Clemizole Hydrochloride (EPX-100), and others is expected to intensify competition in the Dravet syndrome treatment landscape during the forecast period.
Further details will be provided in the report….
Drug Class/Insights into Leading Emerging and Marketed Therapies in Dravet Syndrome (2022–2036 Forecast)
The Dravet syndrome market is increasingly defined by precision-driven and mechanism-based therapies, moving beyond conventional anti-seizure medications toward gene-targeted treatments, serotonergic modulators, and novel neuromodulatory approaches that address the underlying pathophysiology, particularly SCN1A-related dysfunction.
- Targeted genetic therapies: Emerging therapies such as Zorevunersen (STK-001) are antisense oligonucleotides designed to directly target the SCN1A gene, aiming to restore sodium channel function. Similarly, gene therapies like ETX101 focus on disease modification through one-time interventions, representing a transformative approach with long-term therapeutic potential.
- Serotonin pathway modulators (targeted small molecules): Agents such as Bexicaserin (LP352) and Clemizole Hydrochloride (EPX-100) act on central serotonin (5-HT) receptors, improving seizure control through neuromodulation. These oral small molecules highlight the growing role of non-GABAergic, targeted mechanisms in improving outcomes.
- Established and marketed anti-seizure therapies: Approved treatments like Cannabidiol (EPIDIOLEX/EPIDYOLEX), Fenfluramine (FINTEPLA), and Stiripentol (DIACOMIT) remain the backbone of therapy. These drugs act via cannabinoid pathways, serotonergic signaling, and GABA modulation, respectively, and are widely adopted across major markets.
Overall, gene therapies and antisense oligonucleotides are expected to drive the next wave of innovation with potential disease-modifying benefits, while serotonergic agents and established anti-seizure medications will continue to support near-term market growth.
Further details will be provided in the report….
Dravet Syndrome Drug Uptake
This section focuses on the uptake rate of potential drugs expected to be launched in the market during the forecast period (2026–2036). The analysis covers the Dravet syndrome market's uptake by drugs, patient uptake by therapy, and sales of each drug.
The uptake of therapies in Dravet syndrome is expected to vary across novel targeted therapies, small molecules, and conventional anti-seizure treatments. Recently approved and emerging therapies such as Fenfluramine and Cannabidiol are anticipated to witness moderate to strong uptake, supported by their improved efficacy in seizure reduction, defined patient populations, and increasing physician familiarity in managing refractory epilepsy.
The adoption of these newer therapies is largely driven by the limitations of traditional anti-seizure medications, which often fail to achieve sustained seizure control and do not address the underlying genetic cause. Additionally, the need for better long-term outcomes and reduced treatment burden in pediatric patients is accelerating the shift toward more targeted approaches. In comparison, next-generation therapies such as gene-targeted treatments and antisense oligonucleotides are expected to show gradual but progressive uptake as clinical evidence matures and these therapies are integrated into treatment algorithms. Overall, the market will see a steady transition toward precision and disease-modifying therapies, although variability in patient response and high costs may influence the pace of adoption.
Further detailed analysis of emerging therapies' drug uptake in the report…
Market Access and Reimbursement of Dravet Syndrome
- The United States
|
The US Reimbursement for Dravet Syndrome Therapies | |
|
Drug |
Access Program |
|
Cannabidiol (EPIDIOLEX/EPIDYOLEX) |
JazzCares |
|
Fenfluramin (FINTEPLA) |
ONWARD |
Reimbursement is a crucial factor that affects the drug’s access to the market. Often, the decision to reimburse comes down to the price of the drug relative to the benefit it produces in treated patients. To reduce the healthcare burden of these high-cost therapies, many payment models are being considered by payers and other industry insiders.
Further details are provided in the final report….
Dravet Syndrome Therapies Price Scenario & Trends
Pricing and analogue assessment of Dravet syndrome therapies highlights evolving price dynamics structures. This section summarizes the cost of approved treatments, the closest and most appropriate analogue selection for emerging therapies, and understanding of how pricing influences market access, adherence, and long-term uptake.
Further details are provided in the final report….
Industry Experts and Physician Views for Dravet Syndrome
To keep up with Dravet syndrome market trends, we take Key Opinion Leaders (KOLs) and Subject Matter Experts (SMEs) opinions working in the domain through primary research to fill the data gaps and validate our secondary research. Industry experts were contacted for insights on the Dravet syndrome emerging therapies, evolving treatment landscape, patient adherence to conventional therapies, therapy switching trends, drug adoption and uptake, accessibility challenges, and epidemiology and real-world prescription patterns in Dravet syndrome, including MD, PhD, Instructor, Postdoctoral Researcher, Professor, Researcher, and others.
DelveInsight’s analysts connected with 10+ KOLs to gather insights; however, interviews were conducted with 6+ KOLs in the 7MM. Centers such as the University of North Carolina at Chapel Hill, the University of Tokyo Hospital, and King’s College London, etc. were contacted. Their opinion helps understand and validate current and emerging Dravet syndrome therapies, highlight unmet medical needs, provide epidemiological context, and support strategic decisions for market access, therapy adoption, and pipeline prioritization in Dravet syndrome.
|
Region |
Key Opinion Leaders (KOLs) and Subject Matter Experts (SMEs) |
|
United Kingdom |
“Despite the challenges of drug development in rare diseases, the current landscape for Dravet syndrome treatment is promising. New, effective, and well-tolerated therapies are emerging, potentially revolutionizing DS care and paving the way for advances in treating other developmental and epileptic encephalopathies (DEEs).” |
|
Japan |
“Dravet syndrome emphasizes the significant disease burden and rising prevalence, advocating for urgent advancements in therapeutic options. They stress the necessity for comprehensive care strategies to improve patient outcomes and alleviate the profound impact on affected families and healthcare systems.” |
Qualitative Analysis: SWOT and Conjoint Analysis
We perform qualitative and market Intelligence analysis using various approaches, such as SWOT analysis and conjoint analysis.
In the SWOT analysis of Dravet syndrome, strengths, weaknesses, opportunities, and threats in terms of disease diagnosis, patient awareness, patient burden, competitive landscape, cost-effectiveness, and geographical accessibility of therapies are provided.
Conjoint analysis analyzes emerging therapies based on relevant attributes such as safety, efficacy, frequency of administration, route of administration, and order of entry. Scoring is given based on these parameters to analyze the effectiveness of therapy.
The team of analysts analyzes promising emerging therapies based on relevant attributes such as safety, efficacy, frequency of administration, route of administration, and order of entry. In efficacy, the trial’s primary and secondary outcome measures are evaluated, whereas the therapies’ safety is evaluated, wherein the acceptability, tolerability, and adverse events are mainly observed. In addition, the scoring is also based on the route of administration, order of entry, probability of success, and the addressable patient pool for each therapy. According to these parameters, the final weightage score and the ranking of the emerging therapies are decided.
Scope of the Report
- The report covers a segment of key events, an executive summary, a descriptive overview of Dravet syndrome, explaining its causes, signs and symptoms, pathogenesis, and currently available treatments.
- Comprehensive insight has been provided into the epidemiology segments and forecasts, the future growth potential of the diagnosis rate, and disease progression along treatment guidelines.
- Additionally, an all-inclusive account of both the current and emerging treatments, along with the elaborate profiles of late-stage and prominent therapies, will have an impact on the current treatment landscape.
- A detailed review of the Dravet syndrome market, historical and forecasted market size, market share by therapies, detailed assumptions, and rationale behind our approach is included in the report, covering the 7MM drug outreach.
- The report provides an edge while developing business strategies by understanding trends through SWOT analysis and expert insights/KOL views, patient journey, and treatment preferences that help in shaping and driving the 7MM Dravet syndrome market.
Report Insights
- Dravet Syndrome Patient Population Forecast
- Dravet Syndrome Therapeutics Market Size
- Dravet Syndrome Pipeline Analysis
- Dravet Syndrome Market Size and Trends
- Dravet Syndrome Market Opportunity (Current and forecasted)
Report Key Strengths
- Epidemiology‑based (Epi‑based) Bottom‑up Forecasting
- Artificial Intelligence (AI)-enabled Market Research Report
- 11-year forecast
- Dravet Syndrome Market Outlook (North America, Europe, Asia-Pacific)
- Patient Burden Trends (by geography)
- Dravet Syndrome Treatment Addressable Market (TAM)
- Dravet Syndrome Competitive Landscape
- Dravet Syndrome Major Companies Insights
- Dravet Syndrome Price Trends and Analogue Assessment
- Dravet Syndrome Therapies Drug Adoption/Uptake
- Dravet syndrome Therapies Peak Patient Share analysis
Report Assessment
- Dravet Syndrome Current Treatment Practices
- Dravet Syndrome Unmet Needs
- Dravet Syndrome Clinical Development Analysis
- Dravet Syndrome Emerging Drugs Product Profiles
- Dravet Syndrome Market Attractiveness
- Dravet Syndrome Qualitative Analysis (SWOT and Conjoint Analysis)
FAQs
Market Insights
- What was the Dravet syndrome market size, the market size by therapies, market share (%), distribution in 2025, and what would it look like by 2036? What are the contributing factors for this growth?
- What are the anticipated pricing variations among different geographies for the emerging therapies in the future?
- What can be the future treatment paradigm of Dravet syndrome?
- What are the disease risks, burdens, and unmet needs of Dravet syndrome? What will be the growth opportunities across the 7MM concerning the patient population with Dravet syndrome?
- Who is the major future competitor in the market, and how will the competitors affect their market share?
- What are the current options for the treatment of Dravet syndrome? What are the current guidelines for treating Dravet syndrome in the US, Europe, and Japan?
Reasons to Buy
- The report will help in developing business strategies by understanding the latest trends and changing treatment dynamics driving the Dravet syndrome market.
- Bottom-up forecasting builds from the affected population to product forecasts, delivering a robust, data-driven approach ideal for new therapies and novel classes.
- Insights on patient burden/disease incidence, evolution in diagnosis, and factors contributing to the change in the epidemiology of the disease during the forecast years.
- Understand the existing market opportunities in varying geographies and the growth potential over the coming years.
- Identifying strong upcoming players in the market will help devise strategies to help get ahead of competitors.
- Detailed analysis and ranking of class-wise potential current and emerging therapies under the conjoint analysis section to provide visibility around leading classes.
- To understand KOLs’ perspectives on the accessibility, acceptability, and compliance-related challenges of existing treatment to overcome barriers in the future.
- Detailed insights into the unmet needs of the existing market so that the upcoming players can strengthen their development and launch strategy.
- This Artificial Intelligence (AI)-enabled report summarizes and simplifies complex datasets within the report into clear, actionable insights for stakeholders, investors, and healthcare providers, enabling faster, data-driven decisions.






