Graft Versus Host Disease Market Summary
Graft versus Host Disease Insights and Trends
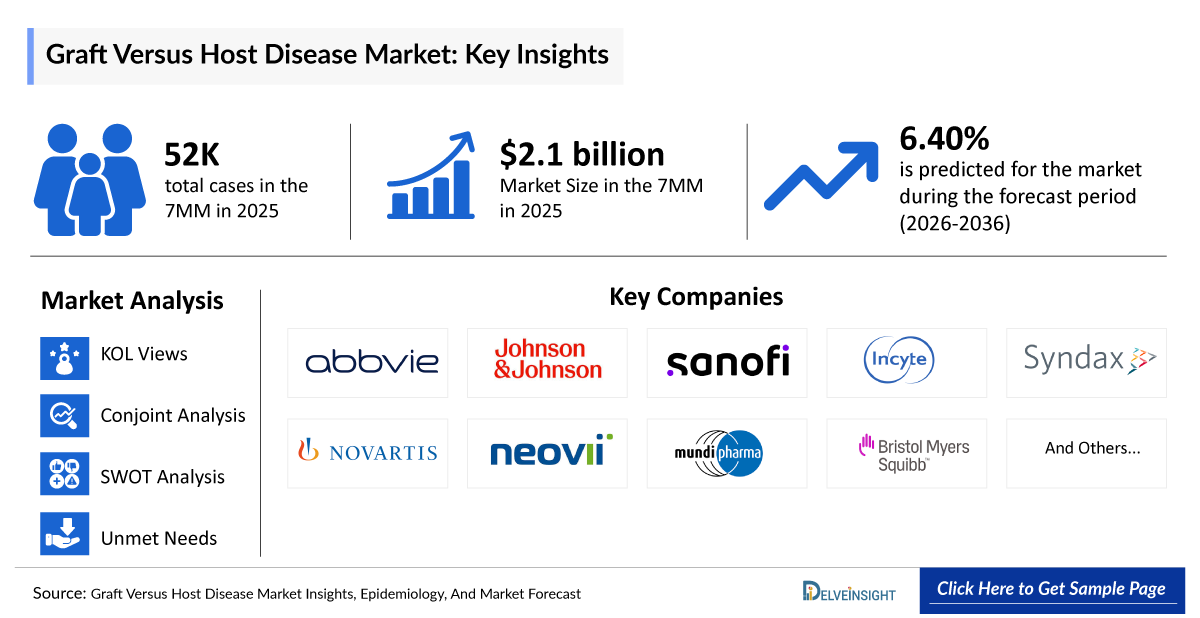
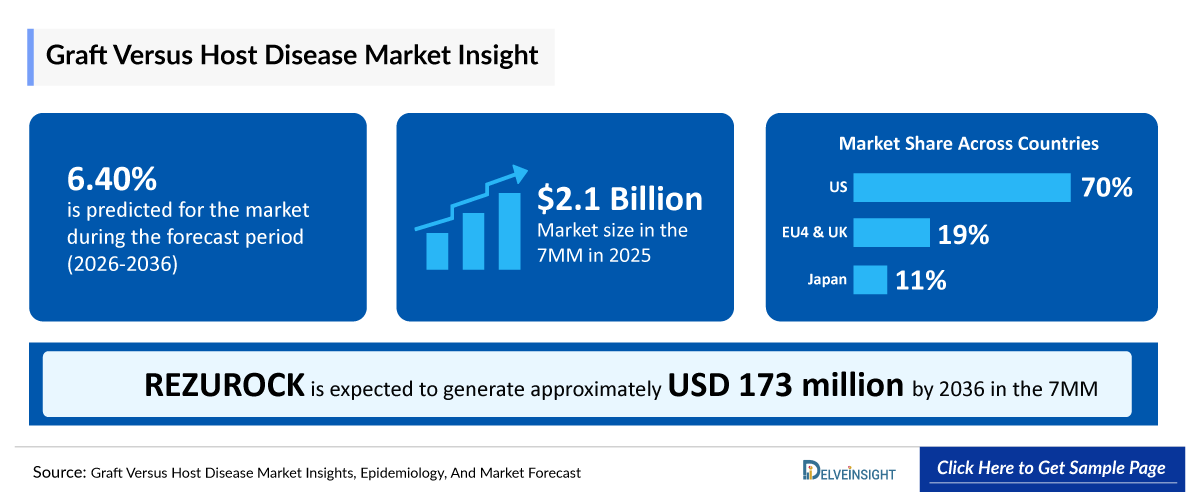
- According to DelveInsight's analysis, the GvHD Market in the 7MM (the US, EU4, the UK, and Japan) was valued at approximately USD 2.1 billion in 2025. The market is expected to expand over the forecast period, driven by a growing Graft Versus Host Disease diagnosed patient population and increasing adoption of emerging therapeutic approaches.
- The GvHD therapeutic market landscape is increasingly shaped by targeted and cell-based therapies, with key agents such as ruxolitinib (JAKAFI/JAKAVI), Ibrutinib (IMBRUVICA), and remestemcel-L (RYONCIL/TEMCELL HS), among others playing a central role in driving market dynamics across the 7MM. In 2025, ruxolitinib leads in revenue generation, followed by REZUROCK.
- The emerging pipeline for GvHD is focused on novel immunomodulatory and cell-based approaches, with key players advancing assets across clinical trial stages. Notably, therapies such as MaaT013 (XERVYTEG), RGI-2001, CYP-001, and others are expected to shape the future landscape, with some anticipated to enter the US market by 2028.
- The US continues to hold the largest Graft Versus Host Disease Market Share, with a revenue of ~USD 1.6 billion in 2025. DelveInsight estimates this will rise significantly by 2036, with the US expected to account for approximately 77% of the total GvHD Market across the 7MM.
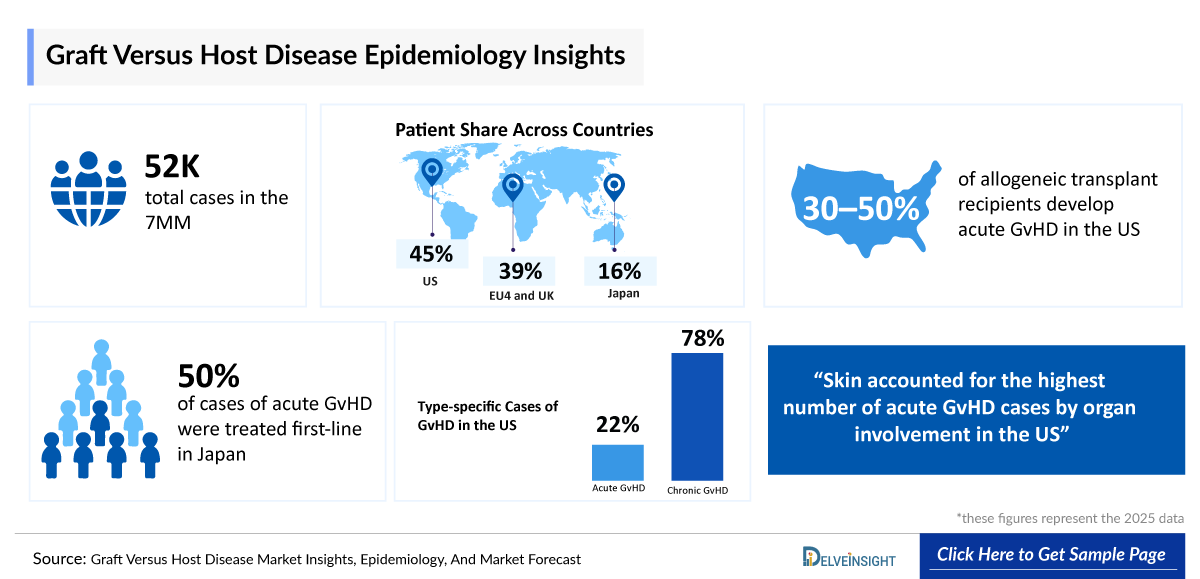
- As per DelveInsight research, the 7MM reported approximately 52,000 total cases of GvHD in 2025. The Graft Versus Host Disease Patient Pool is expected to increase through 2036, driven by the rising number of HSCT procedures and improved survival rates, contributing to a growing burden across these regions.
- Graft versus Host Disease is a serious immune complication following allogeneic HSCT, where donor immune cells attack recipient tissues, commonly affecting skin, liver, and gastrointestinal tract, leading to significant morbidity and impacting overall patient outcomes and quality of life.
- In the US, as per DelveInsight research, type-specific cases of GvHD in 2025 included approximately 4,300 cases of acute GvHD, while the five-year prevalent cases of chronic GvHD were estimated at around 18,500, reflecting a substantial burden across both disease forms.
- Current GvHD prophylaxis is limited by suboptimal efficacy, particularly in high-risk transplant settings, and is associated with significant long-term toxicities; the absence of targeted, durable immune tolerance strategies continues to constrain progress, underscoring the need for safer, more precise prophylactic approaches.
Graft versus Host Disease Market Size and Forecast
- 2025 GvHD Market Size in 7MM: ~USD 2.1 billion
- GvHD Growth Rate (2026–2036) in the 7MM: 6.4% CAGR
Request for Sample Page of the "Graft versus Host Disease Treatment Market"
Factors Contributing to the Growth of the Graft Versus Host Disease Market by DelveInsight
-
Increasing Number of Transplant Procedures
The growing volume of hematopoietic stem cell and bone marrow transplants worldwide is a major factor driving the Incidence of graft-versus-host disease, thereby boosting the demand for effective therapies.
-
Rising Prevalence of GVHD
An increase in both acute and chronic GVHD cases is significantly contributing to the expansion of the treatment market.
-
Improved Diagnosis and Awareness
Enhanced awareness among healthcare professionals, along with advancements in diagnostic techniques, is enabling early detection and timely intervention.
-
Advancements in Treatment Options
Continuous developments in immunosuppressive therapies and targeted treatment approaches are improving patient outcomes and supporting market growth.
-
Strong Pipeline of Emerging Therapies
A robust pipeline featuring innovative biologics, cell-based therapies, and novel drug candidates is expected to broaden the treatment landscape.
-
Growing Focus on Personalized Medicine
The shift toward personalized and precision medicine is encouraging the development of tailored treatment strategies for GVHD patients.
-
Increasing Healthcare Expenditure
Rising healthcare spending and improved access to advanced therapies are contributing to the growth of the GVHD market.
-
Expanding Research and Development Activities
Growing investments in R&D, particularly in rare and complex diseases, are playing a crucial role in advancing GVHD treatment options and driving market growth.
DelveInsight’s ‘Graft versus Host Disease Market Insights, Epidemiology and Market Forecast – 2036’ report delivers an in-depth understanding of GvHD, historical and forecasted epidemiology, as well as the market trends in the United States, EU4 (Germany, Spain, Italy, and France), the United Kingdom, and Japan.
The GvHD Treatment Market Report delivers a comprehensive analysis of the current treatment landscape, including standards of care, clinical practices, and evolving therapeutic algorithms. It evaluates, GvHD patient burden trends, revenue & market share dynamics, peak patient share & therapy uptake analysis, and provides an in-depth market size assessment and growth rate projections (Historical & Forecast 2026 –2036) across the 7MM regions. The report highlights key unmet medical needs in GvHD and maps the competitive and clinical landscape to uncover high‑value opportunities, providing a clear outlook on future market growth potential.
Scope of the Graft Versus Host Disease Market Report | |
|
Study Period |
2022–2036 |
|
Historical Year |
2022–2025 |
|
Forecast Period |
2026–2036 |
|
Base Year |
2025 |
|
Geographies Covered |
|
|
GvHD Market CAGR (Forecast period) |
6.4% (2026-2036) |
|
GvHD Epidemiology Segmentation Analysis |
Patient Burden Assessment
|
|
GvHD Companies |
|
|
GvHD Therapies |
|
|
GvHD Market |
Segmented by
|
|
Analysis |
|
Graft versus Host Disease Understanding
GvHD Overview and Diagnosis
GvHD refers to a complex immune-mediated condition that occurs following allogeneic HSCT, in which donor-derived immune cells recognize recipient tissues as foreign and initiate an immune response. This condition can affect multiple organs, most commonly the skin, liver, and gastrointestinal tract, and is broadly classified into acute and chronic forms. The clinical presentation of GvHD varies widely in severity and progression, with manifestations ranging from mild localized involvement to severe, systemic complications that significantly impact patient outcomes and quality of life.
The GvHD Diagnosis requires a comprehensive and systematic approach, integrating clinical evaluation with histopathological and laboratory findings. It begins with the identification of characteristic signs and symptoms across affected organs, followed by confirmation through tissue biopsy in many cases. Additional assessments, including laboratory parameters and imaging studies, support the evaluation of organ involvement and disease severity, ensuring accurate classification and timely clinical decision-making.
Current Graft versus Host Disease Treatment Landscape
The overall GvHD landscape is steadily evolving, driven by advances in targeted immunomodulatory therapies and cell-based approaches aimed at addressing the underlying immune dysregulation. Key components of current standard practice include JAKAFI/JAKAVI, a JAK1/JAK2 inhibitor that modulates cytokine signaling pathways implicated in GvHD pathophysiology. IMBRUVICA, a Bruton's tyrosine kinase inhibitor, is also utilized in specific settings, targeting B-cell signaling and immune activation. Additionally, RYONCIL/TEMCELL HS, a mesenchymal stromal cell-based therapy, represents an advanced therapeutic modality with immunomodulatory properties. These therapies collectively contribute to the evolving treatment paradigm, reflecting ongoing innovation and a shift toward more targeted and mechanism-based approaches in GvHD.
Graft versus Host Disease Unmet Needs
The section “GvHD Unmet Needs” outlines the critical gaps between the current state of patient care, diagnosis, and the ideal & effective management of the disease. It highlights the obstacles experienced by patients, clinicians, and researchers and identifies potential solutions for future progress.
- Improved early diagnosis and risk stratification
- Limited understanding of cGvHD pathophysiology
- Lack of optimized combinatorial therapies
- Need for novel therapies
Graft versus Host Disease Epidemiology Insights in the 7MM
Key Findings from GvHD Epidemiological Analysis and Forecast
- In 2025, there were an estimated ~7,100 cases of Grade B (II) acute GvHD, followed by ~1,800 cases of Grade C (III) and ~700 cases of Grade D (IV) across the 7MM, with these numbers expected to increase by 2036.
- In US, acute GvHD cases by organ involvement in 2025 were primarily observed in the skin with ~3,700 cases, followed by the gastrointestinal tract with ~2,100 cases and the liver with ~1,300 cases, reflecting the multi-organ impact of the disease.
- In Japan, chronic GvHD cases by organ involvement in 2025 were primarily observed in the oral mucosa with ~3,100 cases and lungs with ~2,400 cases, followed by the eye with ~1,900 cases, skin with ~4000 cases, joints and fascia with ~400 cases, genitals with ~300 cases, gastrointestinal tract with ~1,000 cases, and liver with ~3,000 cases, reflecting diverse multi-organ involvement.
Graft versus Host Disease Epidemiology Segmentation in the 7MM
- Total Graft Versus Host Disease Cases
- Graft Versus Host Disease Type-specific Cases
- Acute Graft Versus Host Disease Cases by Grading.
- Acute Graft Versus Host Disease Cases by Organ Involvement.
- Chronic Graft Versus Host Disease Cases by Grading.
- Chronic Graft Versus Host Disease Cases by Organ Involvement.
- Total Graft Versus Host Disease Treated Cases
- Mortality Adjusted Treated Cases of Graft Versus Host Disease
Market Share (%) Distribution of GvHD by Therapies in the 7MM in 2025
Graft versus Host Disease Drug Analysis & Competitive Landscape
The GvHD drug chapter provides a detailed, market-focused review of approved therapies and the emerging pipeline across Phase I - Phase III Graft Versus Host Disease Clinical Trials. It covers GVHD Mechanism of Action, clinical trial data, regulatory approvals, patents, collaborations, strategic partnerships upcoming Key catalyst for each therapy, along with their advantages, limitations, and recent developments. This section offers critical insights into the GvHD treatment landscape, supporting market assessment, competitive analysis, and growth forecasting for the GvHD therapeutics market.
GvHD Approved Therapies
-
Ruxolitinib (JAKAFI/JAKAVI): Incyte/Novartis
Ruxolitinib is an oral selective JAK1/2 inhibitor developed by Incyte and co-marketed by Novartis. It modulates the JAK-STAT pathway to reduce cytokine-driven inflammation and is indicated for steroid-refractory acute and chronic GvHD after failure of one or two prior systemic therapies in patients aged 12 years and older.
- In August 2023, Novartis reported that ruxolitinib was approved as JAKAVI in Japan for use in GvHD after a HSCT.
- In May 2022, Incyte reported that the EC approved ruxolitinib (JAKAVI) for patients aged 12 years and older with aGvHD or cGvHD who have an inadequate response to corticosteroids or other systemic therapies.
- In September 2021, Incyte reported that the US FDA approved ruxolitinib (JAKAFI) for cGvHD after failure of one or two prior systemic therapies in patients aged 12 years and older.
-
Ibrutinib (IMBRUVICA): AbbVie (Pharmacyclics)/Janssen
Ibrutinib is an oral, once-daily BTK inhibitor that blocks B-cell signaling and reduces abnormal immune activity. It is indicated for cGvHD after failure of one or more prior systemic therapies.
- In August 2022, the US FDA approved IMBRUVICA for pediatric patients ≥1-year of age with cGvHD after the failure of one or more lines of systemic therapy.
- In September 2021, the PMDA approved IMBRUVICA to treat cGvHD after HSCT.
- In August 2017, the US FDA approved IMBRUVICA for treating adult patients with cGvHD after the failure of one or more lines of systemic therapy.
GvHD Marketed/Approved Therapies | ||||||
|
Drug/Therapy |
Company |
Indication |
Molecule Type |
MoA |
RoA |
Marketed Region |
|
Ruxolitinib (JAKAFI/JAKAVI) |
Incyte/ Novartis |
SR-aGVHD and cGvHD |
Small molecule |
JAK1/2 inhibitor |
Oral |
US: 2019, 2021; EU: 2022; JP: 2023. |
|
Ibrutinib (IMBRUVICA) |
AbbVie (Pharmacyclics)/Janssen |
cGvHD |
Small molecule |
BTK inhibitor |
Oral |
US: 2017; JP: 2021 |
|
Remestemcel-L (RYONCIL/TEMCELL HS) |
Mesoblast/JCR Pharmaceuticals |
SR-aGvHD |
MSC therapy |
T-cell activation inhibitor |
IV infusion |
US: 2024; JP: 2015 |
|
XX |
XX |
XX |
XX |
XX |
XX |
XX |
Note: Detailed marketed therapies assessment will be provided in the final report.
Graft versus Host Disease Pipeline Analysis
-
MaaT013 (XERVYTEG): MaaT Pharma
MaaT013 is an off-the-shelf, pooled-donor microbiome therapy designed to restore gut microbiota balance and modulate immune tolerance in GI-predominant acute GvHD. It has received ODD from the US FDA and EMA, with a Phase III trial ongoing showing favorable efficacy and safety signals.
-
- In July 2025, MaaT Pharma and Clinigen signed an exclusive long-term licensing, distribution agreement, and commercial supply agreement for MaaT013 for the treatment of patients with aGvHD.
- In July 2025, MaaT Pharma reported that it has secured a Euro 37.5 million, 4-tranche financing from the European Investment Bank (EIB). The financing will support the advancement of its late-stage hemato-oncology clinical programs including the lead-asset MaaT013.
-
- In June 2025, MaaT Pharma reported the submission of an MAA to the EMA for MaaT013 (XERVYTEG), with potential approval anticipated by mid-2026, positioning it as a first-in-class microbiota-based therapy for third-line steroid-refractory acute GvHD.
-
RGI-2001: REGiMMUNE
RGI-2001 is a liposomal alpha-GalCer formulation that induces regulatory T cells to promote immune tolerance while preserving normal immune function. It has shown potential to reduce GvHD and improve survival without compromising GvL effects and has received ODD from the US FDA.
-
- In April 2024, REGiMMUNE reported a poster presentation at the AACR Annual Meeting highlighting Phase IIb results of RGI-2001, including analyses of Treg and NKT-cell dynamics to evaluate its role in preventing acute GvHD.
- In February 2024, REGiMMUNE reported positive Phase IIb results for RGI-2001 in preventing acute GvHD after alloHCT, with plans to initiate a Phase III randomized study to further evaluate its efficacy and safety.
- In March 2023, REGiMMUNE and San Fu Biotech entered a licensing agreement to develop and commercialize RGI-2001 for aGvHD prophylaxis in major Asian countries, collaborating to accelerate development and conduct clinical studies evaluating its efficacy.
Competitive Landscape of Pipeline Drugs | ||||||
|
Drug Name |
Company |
Highest Phase |
Indication |
RoA |
MoA |
Anticipated Launch in the US |
|
XERVYTEG (MaaT013) |
MaaT Pharma |
III |
aGvHD |
Rectal |
Allogeneic fecal microbiota therapy |
Information is available in the full |
|
RGI-2001 |
REGiMMUNE |
II |
Prevention of aGvHD |
IV infusion |
Activation of Tregs |
Information is available in the full |
|
CYP-001 |
Cynata Therapeutics |
II |
aGvHD |
IV infusion |
Immunomodulatory stem cell therapy |
Information is available in the full |
|
XX |
XX |
XX |
XX |
XX |
XX |
XX |
|
Note: Launch insights are provisional and may change with future report updates or the occurrence of major key catalysts. | ||||||
Graft versus Host Disease Companies, Market Leaders and Emerging Companies
- AbbVie
- Johnson and Johnson
- Sanofi
- Incyte
- Syndax Pharmaceuticals
- Novartis
- Neovii
- Mundipharma
- Platinum Life Excellence Biotech
- Shionogi
- JCR Pharmaceuticals
- Bristol Myers Squibb
- And more
Graft versus Host Disease Drug Updates
- A pivotal Phase III trial of MaaT013 in corticosteroid- and ruxolitinib-refractory GI acute GvHD is ongoing, with DSMB review of the ARES study indicating a favorable benefit–risk profile with high efficacy and low toxicity.
- RGI-2001 completed a Phase II clinical trial (NCT04014790) in 2022 for the prevention of aGvHD in subjects following alloHSCT, and earlier completed a Phase I/II trial (NCT01379209) in 2015 in patients undergoing alloHSCT.
- CYP-001 was evaluated in a global Phase II clinical trial (NCT05643638) in combination with corticosteroids versus corticosteroids alone for high-risk acute GvHD under a cleared US FDA IND.
Graft versus Host Disease Market Outlook
GvHD is a serious and potentially life-threatening immune-mediated condition that occurs following allogeneic HSCT, leading to multi-organ involvement, most commonly affecting the skin, liver, and gastrointestinal tract. It arises when donor immune cells attack recipient tissues, resulting in significant morbidity and long-term complications. GvHD can present as acute or chronic forms, each with variable severity and clinical manifestations. The condition remains a major challenge due to its complex immunopathology, impact on survival and quality of life, and the increasing number of transplant procedures contributing to a growing patient burden across the 7MM.
Key Marketed Therapies shaping current management
- Belumosudil (REZUROCK; Sanofi (Kadmon)/Meiji Seika Pharmaceuticals): An oral, first-in-class selective ROCK2 inhibitor approved by the US FDA in July 2021 for chronic GvHD after failure of at least two prior systemic therapies, with subsequent approvals across other regions. By downregulating STAT3 and upregulating STAT5, it reduces Type 17 and follicular T-helper cells while enhancing regulatory T-cell function.
- Axatilimab-csfr (NIKTIMVO; Incyte/Syndax): A first-in-class monoclonal antibody targeting CSF-1R, granted US FDA approval in August 2024 for chronic GvHD following inadequate response to at least two previous systemic treatments. Inhibiting CSF-1R signaling on monocytes and macrophages helps reduce pathogenic inflammation and fibrosis, central mechanisms driving chronic GvHD.
Overall, the GvHD Market stands at a pivotal transformation point. Historically defined by CS-centered, empirically layered immunosuppression, management has long relied on reactive and symptom-driven strategies with limited durability. Now, a new generation of mechanism-specific therapies spanning precision biologics, cell-based immunomodulators, and microbiome-restorative agents is reshaping the landscape toward prevention, immune tolerance, and tissue repair in 7MM GvHD market from 2022–2036, with strong commercial implications for both marketed products and emerging pipelines.
- In 2025, the US dominated the GvHD market across the 7MM, accounting for 77% of the total Graft Versus Host Disease Market Share. The Graft Versus Host Disease Market Size in the US was valued at USD 2.1 billion, driven largely by the anticipated impact of emerging therapies on market growth.
Graft versus Host Disease Drug Class (2022–2036 Forecast)
The GvHD Market landscape includes both established therapies and a growing pipeline targeting acute GvHD, chronic GvHD, and preventive strategies in the context of HSCT. These approaches primarily focus on modulating immune dysregulation and reducing harmful donor-derived immune activity responsible for tissue damage. Small molecules and monoclonal antibodies form the backbone of current and emerging strategies, offering targeted pathway inhibition and immune cell modulation, while additional modalities such as MSC-based therapies and T-cell activation inhibitors are expanding the scope of innovation. Collectively, these advancements reflect a continuous evolution in addressing unmet needs and the complex clinical spectrum of GvHD across the 7MM.
- Small molecules: Small molecules play a central role in the GvHD landscape, offering targeted modulation of key immune signaling pathways. Marketed agents such as JAK inhibitors, ROCK2 inhibitors, and alkylating agents continue to shape current clinical practice, while emerging therapies including RGI-2001 and ivarmacitinib (SHR0302) are advancing next-generation strategies focused on immune tolerance and cytokine regulation.
- Monoclonal antibodies: Monoclonal antibodies are driving innovation in GvHD by enabling precise targeting of immune cell subsets and inflammatory pathways. Emerging agents such as MEDI-5071 (siplizumab) and itolizumab (EQ001) are being developed to selectively modulate T-cell activity, while approved therapies like axatilimab-csfr (NIKTIMVO) highlight progress in this class. These biologics aim to improve clinical outcomes with targeted immune suppression, reduced off-target effects, and potential for durable disease control.
Graft versus Host Disease Drug Uptake
This section focuses on the uptake rate of potential Graft Versus Host Disease drugs expected to be launched in the market during the forecast period (2026–2036). The analysis covers the GvHD drug’s uptake, performance at peak, factors affecting performance during prime years of growth, patient uptake by therapy, and anticipated sales generated by each drug.
Current therapies for GvHD primarily focus on immunosuppression and symptom control, highlighting a significant unmet need for approaches that restore immune tolerance. The future landscape appears promising, with ongoing development of innovative therapies such as MaaT013 (XERVYTEG), which is anticipated to enter the US market in 2027. The therapy is expected to generate notable revenue at launch, with growth projected through 2036, driven by a medium uptake.
Graft versus Host Disease Market Access and Reimbursement of Approved Therapies
The report further provides detailed insights on the country-wise accessibility and reimbursement scenarios, cost-effectiveness scenario of approved therapies, programs making accessibility easier and out-of-pocket costs more affordable, insights on patients insured under federal or state government prescription drug programs, etc.
Graft versus Host Disease Price Scenario & Trends
Pricing and analogue assessment of GvHD therapies highlights evolving price dynamics structures. This section summarizes the cost of approved treatments, closest and most appropriate analogue selection for emerging therapies, and understanding of how pricing influences market access, adherence, and long-term uptake.
Pricing of GVHD Approved Drugs
Ruxolitinib (JAKAVI), a targeted JAK1/2 inhibitor used in GvHD, carries a relatively high treatment cost compared to conventional therapies, reflecting its targeted mechanism and clinical value. In Japan, where it is approved for GvHD, the price is approximately JPY 8,185, with no directly comparable standardized pricing established across the US and EU markets due to regional variations in reimbursement and access.
Industry Experts and Physician Views for Graft versus Host Disease
To keep up with GvHD market trends, we take Key Opinion Leaders (KOLs) and Subject Matter Experts (SMEs) opinions working in the domain through primary research to fill the data gaps and validate our secondary research. Industry Experts were contacted for insights on the GvHD emerging therapies, evolving treatment landscape, patient adherence to conventional therapies, therapy switching trends, drug adoption and uptake, accessibility challenges, and epidemiology and real-world prescription patterns in GvHD, including MD, PhD, Instructor, Postdoctoral Researcher, Professor, Researcher, and others.
DelveInsight’s analysts connected with 8+ KOLs to gather insights at country level. Centers such as the University of Michigan, University Hospital Regensburg, University Hospital Kyoto Prefectural University of Medicine, etc. were contacted. Their opinion helps understand and validate current and emerging GvHD therapies, highlight unmet medical needs, provide epidemiological context, and support strategic decisions for market access, therapy adoption, and pipeline prioritization in GvHD.
Region |
Key Opinion Leaders (KOLs) and Subject Matter Experts (SMEs) |
|
United States |
“Genetic variations in cytokine-related genes, including TNF-α, IL-10, and IFN-γ, contribute to the risk of GvHD by influencing the intensity of the immune response, particularly the cytokine storm. While the associations between these polymorphisms and GvHD remain variable and inconclusive, future advancements in donor selection are expected to incorporate both HLA and non-HLA genetic markers to refine transplant compatibility and improve patient outcomes.” |
|
Germany |
“Acute GvHD, traditionally defined as occurring before Day 100, has been expanded in recent classifications to include late-onset acute GvHD and an overlap syndrome, particularly following RIC. Clinical manifestations typically affect the skin, gastrointestinal tract, and liver, with emerging evidence suggesting that the lungs could also be involved in experimental GvHD.” |
|
Japan |
“For patients with acute GvHD, early referral to a transplant center is crucial to access effective treatments and clinical trials, especially as the condition can become steroid-refractory. cGvHD typically begins with steroids, but referral to a specialized center ensures multidisciplinary care, addressing complications like steroid side effects, infections, and muscle weakness.” |
Graft Versus Host Disease Qualitative Analysis: SWOT and Attribute Analysis
We perform qualitative and Graft Versus Host Disease Market Intelligence analysis using various approaches, such as SWOT analysis and attribute analysis.
In the SWOT analysis of GvH, strengths, weaknesses, opportunities, and threats in terms of disease diagnosis, patient awareness, patient burden, competitive landscape, cost-effectiveness, and geographical accessibility of therapies are provided. Attribute Analysis analyzes emerging therapies based on relevant attributes such as safety, efficacy, frequency of administration, route of administration, and order of entry. Scoring is given based on these parameters to analyze the effectiveness of therapy.
The team of analysts analyzes promising emerging therapies based on relevant attributes such as safety, efficacy, frequency of administration, route of administration, and order of entry. In efficacy, the trial’s primary and secondary outcome measures are evaluated, whereas the therapies’ safety is evaluated, wherein the acceptability, tolerability, and adverse events are majorly observed. In addition, the scoring is also based on the route of administration, order of entry, probability of success, and the addressable patient pool for each therapy. According to these parameters, the final weightage score and the ranking of the emerging therapies are decided.
Scope of the Graft Versus Host Disease Market Report
- The Graft Versus Host Disease Therapeutics Market Report covers a segment of key events, an executive summary, a descriptive overview, explaining their causes, signs and symptoms, pathogenesis, and currently available treatments.
- Comprehensive insight has been provided into the Graft Versus Host Disease epidemiology segments and forecasts, the future growth potential of the diagnosis rate, and disease progression along treatment guidelines.
- Additionally, an all-inclusive account of both the current and emerging treatments, along with the elaborative profiles of mid and late stage therapies, will have an impact on the current Graft Versus Host Disease Treatment landscape.
- A detailed review of the Graft Versus Host Disease Treatment Market, historical and forecasted Graft Versus Host Disease Market Size, Graft Versus Host Disease Market Share by therapies, detailed assumptions, and rationale behind our approach is included in the report, covering the 7MM drug outreach.
- The Graft Versus Host Disease Therapeutics Market Report provides an edge while developing business strategies by understanding trends through SWOT analysis and expert insights/KOL views, patient journey, and treatment preferences that help in shaping and driving the 7MM GvHD Market.
Graft Versus Host Disease Market Report Insights
- GvHD Patient Population Forecast
- GvHD Therapeutics Market Size
- GvHD Pipeline Analysis
- GvHD Market Size and Trends
- GvHD Market Opportunity (Current and forecasted)
Graft Versus Host Disease Market Report Key Strengths
- Epidemiology‑based (EPI ‑ based) bottom‑up forecasting
- Artificial Intelligence (AI) - enabled market research report
- 11-year forecast
- GvHD Market Outlook (North America, Europe, Asia-Pacific)
- GvHD Burden trends (by geography)
- GvHD Treatment Addressable Market (TAM)
- GvHD Competitive Landscape
- Major GvHD Companies Insights
- GvHD Price trends and analogue assessment
- GvHD Therapies Drug Adoption/Uptake
- GvHD Therapies Peak Patient Share analysis
Graft Versus Host Disease Market Report Assessment
- Current GvHD Treatment Practices
- GvHD Unmet Needs
- GvHD Clinical Development Analysis
- GvHD emerging drugs product profiles
- GvHD Market attractiveness
- GvHD Qualitative analysis (SWOT and conjoint analysis)
Key Questions Answered in the Graft Versus Host Disease Market Report
Graft Versus Host Disease Market Insights
- What was the GvHD Market Size, the Graft Versus Host Disease Market Size by therapies, market share (%) distribution in 2025, and what would it look like by 2036? What are the contributing factors for this growth?
- What are the anticipated pricing variations among different geographies for the emerging therapies in the future?
- What can be the future treatment paradigm of GvHD?
- What are the disease risks, burdens, and Graft Versus Host Disease Unmet Needs? What will be the growth opportunities across the 7MM concerning the patient population with GvHD?
- Who is the major future competitor in the market, and how will the competitors affect their market share?
- What are the current options for the treatment of GvHD? What are the current guidelines for treating GvHD in the US, Europe, and Japan?
Reasons to Buy the Graft Versus Host Disease Market Report
- The Graft Versus Host Disease Therapeutics Market Report will help in developing business strategies by understanding the latest trends and changing treatment dynamics driving the GvHD Drugs Market.
- Bottom-up forecasting builds from the affected population to product forecasts, delivering a robust, data-driven approach ideal for new therapies and novel classes.
- Insights on patient burden/disease Graft Versus Host Disease Prevalence, evolution in diagnosis, and factors contributing to the change in the epidemiology of the disease during the forecast years.
- Understand the existing market opportunities in varying geographies and the growth potential over the coming years.
- Identifying strong upcoming players in the Graft Versus Host Disease Drugs Market will help devise strategies to help get ahead of competitors.
- Detailed analysis and ranking of class-wise potential current and emerging therapies under the attribute analysis section to provide visibility around leading classes.
- To understand KOLs’ perspectives on the accessibility, acceptability, and compliance-related challenges of existing treatment to overcome barriers in the future.
- Detailed insights on the unmet needs of the existing Graft Versus Host Disease Drugs Market so that the upcoming players can strengthen their development and launch strategy.
- This Artificial Intelligence (AI) - enabled report summarize and simplify complex datasets within the report into clear, actionable insights for stakeholders, investors, and healthcare providers, enabling faster, data-driven decisions.
Stay updated with us for Recent Articles:-