Idiopathic Membranous Nephropathy Market Summary
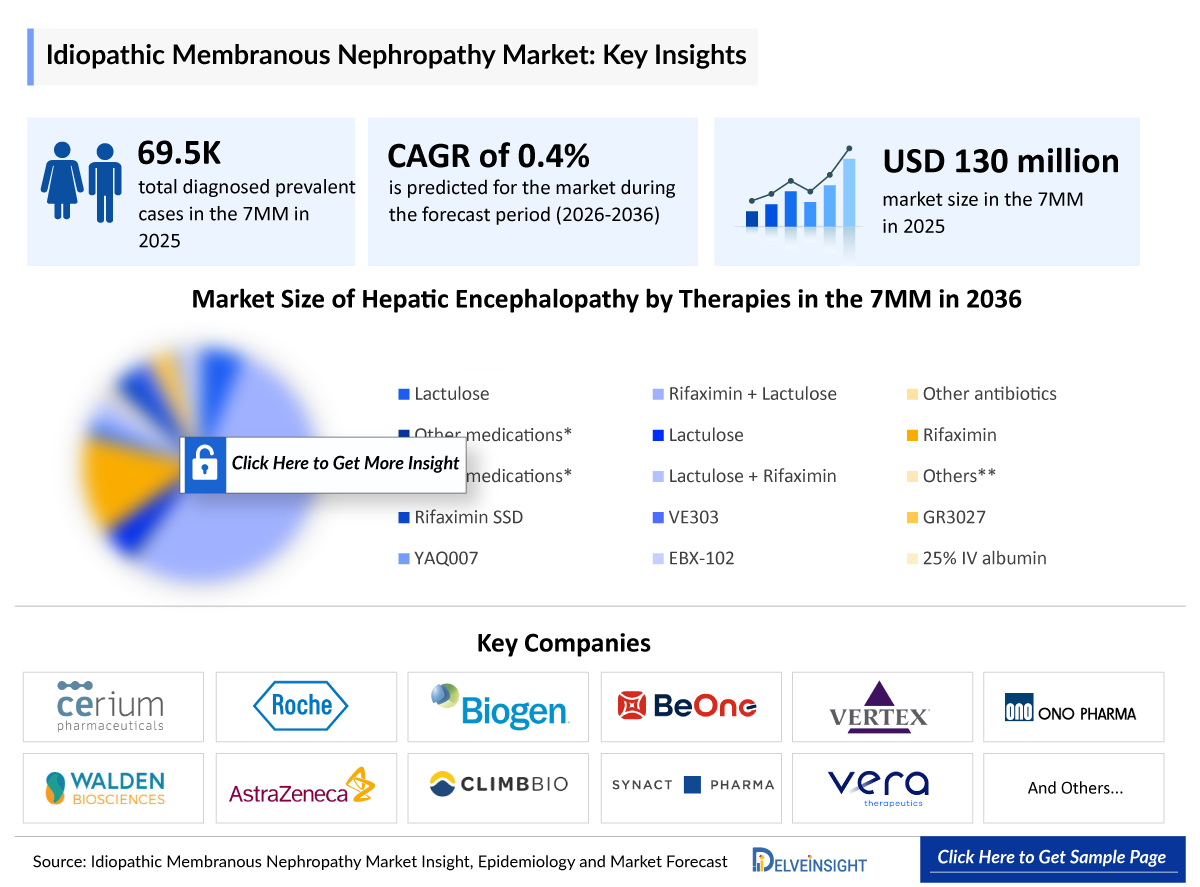
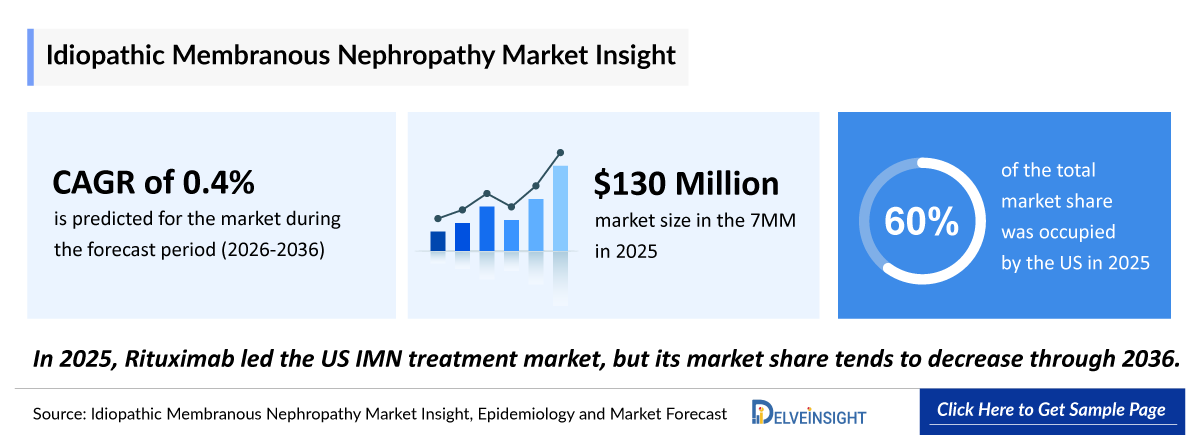
- According to DelveInsight’s analysis, Idiopathic Membranous Nephropathy market size was found to be ~USD 130 million in the leading markets (the United States, the EU4 (Germany, France, Italy, and Spain), the United Kingdom, and Japan) in 2025.
- In 2025, the United States held the largest market share for Idiopathic Membranous Nephropathy among the 7MM, accounting for approximately 60% of the total market.
Idiopathic Membranous Nephropathy Market Insights and Trends
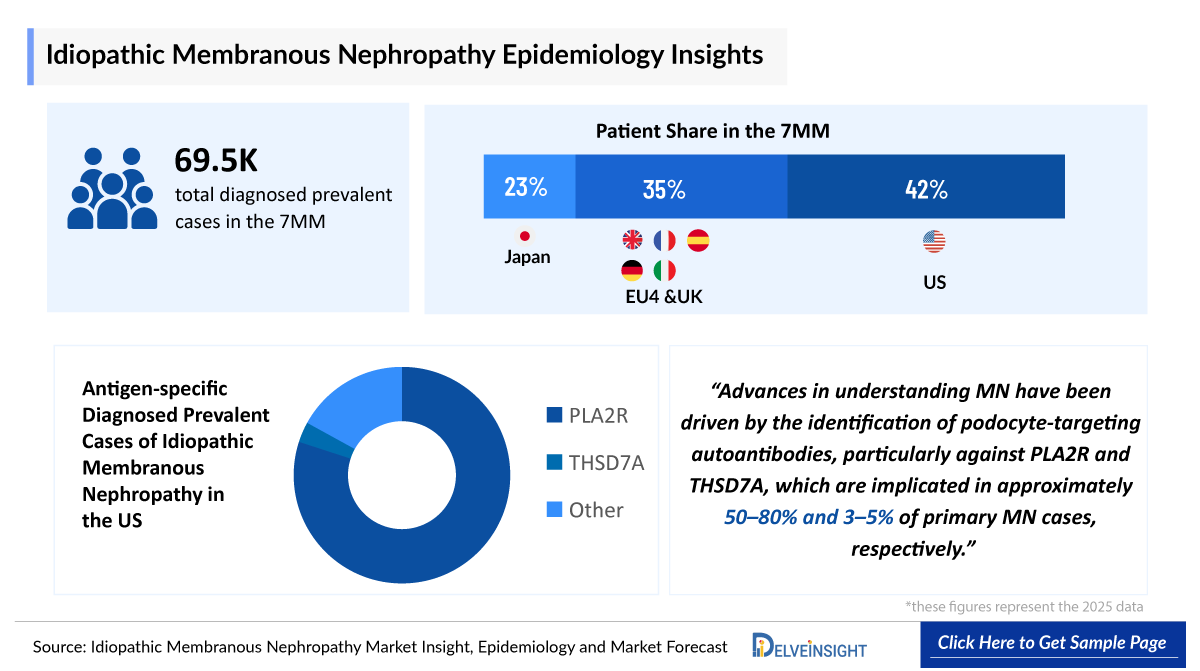
- Idiopathic Membranous Nephropathy is often characterized by the antigens associated with the sub epithelial immune complex deposits, some of them being PLA2R and THSD7A. In 2022, PLA2R, THSD7A, and other antigens were detected in 23,000,800, and 4,500 Idiopathic Membranous Nephropathy patients, respectively, in the US.
- Among EU4 and the UK, Germany had the highest prevalent population of Idiopathic Membranous Nephropathy with 6,000 cases, followed by the UK, which had a prevalence of 5,000 cases in 2022. On the other hand, France had the lowest prevalent population of 4,000 cases in 2022.
- The United States contributed to the largest diagnosed prevalent cases of Idiopathic Membranous Nephropathy, accounting for ~40% in 2025.
- In 2025, the total market size of Idiopathic Membranous Nephropathy in US was approximately USD ~85 million and is projected to increase during the forecast period (2026–2036).
Idiopathic Membranous Nephropathy Market Size and Forecast
- 2025 Idiopathic Membranous Nephropathy Market Size: ~USD 130 million
- 2036 Projected Idiopathic Membranous Nephropathy Market Size: ~USD 1,000 million
- Idiopathic Membranous Nephropathy Growth Rate (2026–2036): 0.4% CAGR
DelveInsight's ‘Idiopathic Membranous Nephropathy – Market Insights, Epidemiology and Market Forecast – 2036’ report delivers an in-depth understanding of the Idiopathic Membranous Nephropathy, historical and forecasted epidemiology, as well as the Idiopathic Membranous Nephropathy market trends in the United States, EU4 (Germany, Spain, Italy, and France) and the United Kingdom, and Japan.
The Idiopathic Membranous Nephropathy market report delivers a comprehensive analysis of the current treatment landscape, including standards of care, clinical practices, and evolving therapeutic algorithms. It evaluates, Idiopathic Membranous Nephropathy patient burden trends, revenue & market share dynamics, peak patient share & therapy uptake analysis, and provides an in-depth market size assessment, and growth rate projections (Historical & Forecast 2022–2036) across global regions. The report highlights key unmet medical needs in Idiopathic Membranous Nephropathy and maps the competitive and clinical landscape to uncover high-value opportunities, providing a clear outlook on future market growth potential.
Scope of the Idiopathic Membranous Nephropathy Market Report | |
|
Study Period |
2022–2036 |
|
Historical Year |
2022–2025 |
|
Forecast Period |
2026–2036 |
|
Base Year |
2026 |
|
Geographies Covered |
|
|
Idiopathic Membranous Nephropathy Market CAGR (Study period/Forecast period) |
0.4% (2026-2036) |
|
Idiopathic Membranous Nephropathy Epidemiology Segmentation Analysis |
Patient Burden Assessment
|
|
Idiopathic Membranous Nephropathy Companies |
|
|
Idiopathic Membranous Nephropathy Therapies |
|
|
Idiopathic Membranous Nephropathy Market |
Segmented by
|
|
Analysis |
|
Idiopathic Membranous Nephropathy Understanding and Treatment Algorithm
Idiopathic Membranous Nephropathy Overview and Diagnosis
Membranous nephropathy (MN) is a rare immune-mediated glomerular disorder marked by podocyte injury that increases protein permeability, leading to proteinuria, hypoalbuminemia, and edema characteristic of nephrotic syndrome; it is the leading cause of nephrotic syndrome in Caucasian adults (~30%), predominantly affecting men aged 30–50 years and occurring infrequently in children. The disease is driven by in situ formation of subepithelial immune complexes along the glomerular basement membrane, triggering complement activation, mainly via lectin and alternative pathways, and resulting in capillary wall thickening, structural damage, and impaired filtration without significant inflammation.
Key antigens in primary MN include PLA2R, THSD7A, NELL-1, and Sema3B, with disease progression influenced by epitope spreading and elevated autoantibody levels, while triggers for loss of immune tolerance remain unclear but may involve environmental or infectious factors. MN is clinically heterogeneous, with one-third of patients achieving spontaneous remission, one-third remaining stable, and one-third progressing, with up to half developing end-stage renal disease over time. It can be classified as primary, secondary (associated with infections, autoimmune disorders, malignancies, or drugs), alloimmune, or idiopathic. Clinically, it presents insidiously with edema, foamy urine, hyperlipidemia, fatigue, and hypertension, and is defined histologically by subepithelial deposits and characteristic basement membrane “spikes.” Advances in biomarker identification, particularly circulating autoantibodies, have enhanced diagnosis, monitoring, and prognostication, with declining antibody levels preceding remission and persistent titers predicting relapse.
Membranous nephropathy (MN) is primarily diagnosed through detection of proteinuria on urine testing, often supported by blood tests assessing renal function, hypoalbuminemia, lipid levels, and specific autoantibodies such as anti-PLA2R and anti-THSD7A, while evaluation for secondary causes is guided by clinical context. Kidney biopsy remains the diagnostic gold standard, demonstrating glomerular basement membrane thickening, subepithelial immune deposits, and characteristic IgG (predominantly IgG4) and C3 staining, with electron microscopy confirming dense deposits and enabling disease staging. The advent of highly specific anti-PLA2R assays has refined diagnosis and monitoring, allowing classification into active or inactive disease and guiding treatment decisions, with elevated antibody levels and significant proteinuria indicating active disease requiring immunosuppression, whereas declining or absent antibodies suggest remission and support conservative management.
Further details are provided in the report.
Current Idiopathic Membranous Nephropathy Treatment Landscape
Idiopathic Membranous Nephropathy management includes supportive measures such as renin–angiotensin system blockade and blood pressure control, followed by immunosuppressive therapies (e.g., corticosteroids, cyclophosphamide, calcineurin inhibitors, mycophenolate mofetil, or rituximab) in resistant cases, while antigen-specific therapies remain an area of ongoing development.
Further details related to country-based variations are provided in the report.
Idiopathic Membranous Nephropathy Unmet Needs
The section “unmet needs of Idiopathic Membranous Nephropathy (Idiopathic Membranous Nephropathy)” outlines the critical gaps between the current state of patient care, diagnosis, and the ideal & effective management of the disease. It highlights the obstacles experienced by patients, clinicians, and researchers and identifies potential solutions for future progress.
- Lack of Approved therapy
- Economic burden of disease
- Shortcomings associated with current treatment
- Patient progression to end-stage renal disease, and others…..
Note: Comprehensive unmet needs insights in Idiopathic Membranous Nephropathy and their strategic implications are provided in the full report.
Idiopathic Membranous Nephropathy Epidemiology
Key Findings from Idiopathic Membranous Nephropathy Epidemiological Analysis and Forecast
- According to DelveInsight’s estimates, the total number of prevalent cases of Idiopathic Membranous Nephropathy in the 7MM was nearly 89,000 cases in 2025 and is projected to increase during the forecasted period.
- In the 7MM, the US accounted for the highest diagnosed prevalent cases of Idiopathic membranous nephropathy in 2025, with around ~35,000 cases; these numbers are expected to increase during the forecast period.
- The total treated cases in the US for Idiopathic Membranous Nephropathy in 2025 were 35,000 which is expected to increase 37,000 by 2036.
- In the EU4 and the UK, individuals were the most affected by PLAR2-specific Idiopathic membranous nephropathy, with approximately 15,000 cases reported in 2025.
Idiopathic Membranous Nephropathy Drug Analysis & Competitive Landscape
The Idiopathic Membranous Nephropathy drug chapter provides a detailed, market-focused review of approved therapies and the emerging pipeline across Phase I/II–II clinical trials. It covers mechanism of action, clinical trial data, regulatory approvals, patents, collaborations, strategic partnerships upcoming Key catalyst for each therapy, along with their advantages, limitations, and recent developments. This section offers critical insights into the Idiopathic Membranous Nephropathy treatment landscape, supporting market assessment, competitive analysis, and growth forecasting for the Idiopathic Membranous Nephropathy therapeutics market.
Approved Therapies for Idiopathic Membranous Nephropathy
The therapeutic landscape for idiopathic membranous nephropathy underscores a notable unmet need, as no therapies are currently marketed or specifically approved for this condition; accordingly, the marketed drugs segment remains absent, with the focus primarily on evaluating emerging candidates in Phase III and Phase II stages of development.
Note: Detailed marketed therapies assessment will be provided in the final report.
Idiopathic Membranous Nephropathy Pipeline Analysis
SNP-ACTH (1-39) Gel: Cerium Pharmaceuticals
Cerium has developed SNP-ACTH (1-39) Gel, a subcutaneous formulation of a synthetic porcine ACTH peptide, offering a quantifiable and consistent dose that may enhance optimization of treatment response in primary membranous nephropathy (PMN). Preclinical studies and a Phase I trial in healthy subjects have demonstrated its safety, tolerability, and potency. Following regulatory discussions, the company has initiated a Phase III randomized superiority trial to evaluate whether SNP-ACTH (1-39) Gel is more effective than rituximab in achieving sustained proteinuria remission as a first-line immunosuppressive therapy in moderate- to high-risk PMN patients, with patient enrollment currently underway.
Competitive Landscape of Idiopathic Membranous Nephropathy Pipeline Drugs | ||||||
|
Drug Name |
Company |
Highest Phase |
Indication |
RoA |
MoA |
Anticipated Launch in the US |
|
SNP-ACTH (1-39) |
Cerium Pharmaceuticals |
III |
Patients with primary membranous nephropathy (PMN) at month 24 |
Subcutaneous |
Melanocortin receptor agonists |
2027 |
|
GAZYVA ( Obinutuzumab ) |
Hoffmann-La-Roche |
III |
Participants with primary membranous nephropathy |
Intravenous |
Antibody-dependent cell cytotoxicity |
Information is available in the full report |
|
MOR202 (felzartamab) |
Biogen |
III |
Adult participants with PMN |
Intravenous |
Antibody-dependent cell cytotoxicity |
Information is available in the full report |
|
Note: Launch insights are provisional and may change with future report updates or the occurrence of major key catalysts. | ||||||
Idiopathic Membranous Nephropathy Key Players, Market Leaders and Emerging Companies
- Cerium Pharmaceuticals
- Hoffmann-La-Roche
- Biogen
- BeOne Medicines Ltd., among others
Idiopathic Membranous Nephropathy Drug Updates
- In January 2026, EMA has granted Priority Medicines (PRIME) designation to povetacicept for primary membranous nephropathy.
- In November 2025, FDA has granted Fast Track Designation to povetacicept for primary membranous nephropathy.
- In November 2025, Vertex Pharmaceuticals Incorporated announced updated data for povetacicept (pove) in IgA nephropathy (IgAN) and primary membranous nephropathy (pMN) from the ongoing RUBY-3 trial at the American Society of Nephrology (ASN) Kidney Week 2025 in Houston, Texas.
Idiopathic Membranous Nephropathy Market Outlook
The membranous nephropathy (MN) market is driven by a significant unmet need, as there is no curative or specifically approved therapy, with treatment largely centered on supportive care and broad immunosuppression. Current management relies on ACE inhibitors/ARBs, diuretics, and statins, while immunosuppressive agents such as calcineurin inhibitors, cyclophosphamide, and corticosteroids remain key revenue drivers despite safety concerns and relapse risks. Rituximab has emerged as a preferred first-line therapy for primary MN due to improved efficacy and sustained remission compared to cyclosporine. However, real-world utilization of advanced therapies remains relatively low, highlighting gaps in treatment adoption. Ongoing research and clinical trials are focused on targeted and combination therapies, indicating a shift toward more effective and personalized treatment approaches.
Obinutuzumab is an anti-CD19 mab being evaluated in a Phase III study for treating pMN. It has demonstrated a good safety and efficacy profile in clinical studies and is already approved for indications like lymphocytic leukemia and follicular lymphoma. The real-world retrospective study demonstrated that the drug showed strong efficacy in pMN, achieving high immunological (92.0%) and clinical remission rates (83.6%) with improvements in proteinuria, serum albumin, and kidney function over a median 13-month follow-up. Clinical responses were similar whether used as initial or alternative therapy and were maintained in rituximab-refractory and anti-PLA2R–negative patients, supporting its potential as a promising treatment option.
SNP-ACTH [1-39] Gel is a subcutaneous formulation of synthetic porcine ACTH drug substance. It is similar to other synthetic long-acting ACTH products containing highly purified synthetic ACTH peptides. . The company has completed preclinical studies demonstrating the safety and potency of SNP-ACTH (1-39) Gel, as well as a Phase I clinical trial in healthy subjects confirming its safety and tolerability. Following interactions with the US FDA on trial design and outcome measures, the company initiated a prospective, randomized Phase III superiority study evaluating SNP-ACTH (1-39) Gel versus rituximab as a first-line immunosuppressive therapy in adults with moderate- to high-risk PMN, with the primary objective of inducing durable remission of proteinuria; patient enrollment is currently ongoing. The program has received regulatory support, including Orphan Drug Designation from the FDA in June 2023, and is backed by solid financing, with Cerium raising USD 12 million in a Series B round in November 2021, bringing total funding to USD 19 million to advance the asset through pivotal development.
Idiopathic Membranous Nephropathy Companies, including Cerium Pharmaceuticals, Hoffmann-La-Roche, Biogen, BeOne Medicines Ltd., and others, are investigating potential drug candidates that can significantly change the market landscape during the forecast period. Approaches include Melanocortin receptor agonists (SNP-ACTH) and Antibody-dependent cell cytotoxicity (GAZYVA), and Agammaglobulinaemia tyrosine kinase inhibitors (BRUKINSA), and others.
Overall, the launch of first-in-class therapies, improved diagnosis, and rising disease awareness are expected to drive steady growth in the 7MM Idiopathic Membranous Nephropathy market from 2022–2036, with strong commercial implications for both marketed products and emerging pipelines.
Key findings from Idiopathic Membranous Nephropathy Market Forecast Report
- In 2025, the United States held the largest market share for Idiopathic Membranous Nephropathy among the 7MM, accounting for approximately 60% of the total market.
- In 2025, Rituximab led the US Idiopathic Membranous Nephropathy treatment market but it’s market share tends to decrease through 2036.
- In 2025, total treated cases for Idiopathic Membranous Nephropathy were ~45,000 across the 7MM countries.
Further details will be provided in the report….
Drug Class/Insights into Leading Emerging and Marketed Therapies in Idiopathic Membranous Nephropathy (2022–2036 Forecast)
The Idiopathic Membranous Nephropathy market comprises of different mechanism class, including Melanocortin receptor agonists and Antibody-dependent cell cytotoxicity, and Agammaglobulinaemia tyrosine kinase inhibitors, each designed to target distinct inflammatory pathways underlying disease pathophysiology.
Melanocortin receptor agonists: Melanocortin receptor agonists represent a promising approach in idiopathic membranous nephropathy by exerting direct podocyte-protective, anti-inflammatory, and immunomodulatory effects. Through activation of receptors such as MC1R and MC5R, they help reduce proteinuria, stabilize podocyte function, and mitigate complement-mediated injury. Early clinical evidence, particularly with ACTH-based therapies, indicates potential in inducing remission, especially in patients unresponsive to standard immunosuppression; however, further large-scale studies are needed to confirm their efficacy and optimize their use.
Brunton’s (Agammaglobulinaemia) tyrosine kinase: Bruton’s tyrosine kinase (BTK) is a key regulator of B-cell activation and autoantibody production in idiopathic membranous nephropathy (Idiopathic Membranous Nephropathy). BTK inhibition reduces pathogenic antibodies (e.g., anti-PLA2R), limits immune complex formation, and dampens inflammatory signaling, thereby mitigating glomerular injury. Consequently, BTK inhibitors represent a promising targeted alternative to conventional immunosuppression, though further clinical validation is required.
Idiopathic Membranous Nephropathy Drug Uptake
This section focuses on the uptake rate of potential drugs expected to be launched in the market during the forecast period (2026–2036). The analysis covers the Idiopathic Membranous Nephropathy drug’s uptake, performance at peak, factors affecting performance during prime years of growth, patient uptake by therapy, and anticipated sales generated by each drug
Drug uptake in idiopathic membranous nephropathy is driven by a combination of supportive care and immunosuppressive therapies, with treatment selection guided by disease severity and risk of progression. While angiotensin-converting enzyme inhibitors/angiotensin receptor blockers and other supportive measures are widely used across patients, immunosuppressive uptake remains concentrated in high-risk populations. Among these, calcineurin inhibitors and corticosteroids have historically seen substantial use, contributing significantly to treatment volumes, whereas cyclophosphamide is reserved for severe cases. More recently, Rituximab has emerged as the preferred first-line therapy due to its favorable efficacy and safety profile, although real-world utilization remains relatively low compared to traditional therapies. Despite moderate remission rates with existing options, issues such as relapse, adverse effects, and treatment resistance persist, creating a shift toward targeted, B-cell–directed and novel immunomodulatory therapies currently in development. Overall, drug uptake is expected to evolve significantly with the introduction of these emerging agents, potentially reshaping the treatment paradigm over the coming decade.
Detailed insights of emerging therapies' drug uptake is included in the report
Market Access and Reimbursement of Approved therapies in Idiopathic Membranous Nephropathy
The Idiopathic Membranous Nephropathy market report further provides detailed insights on the country-wise accessibility and reimbursement scenarios, cost-effectiveness scenario of approved therapies, programs making accessibility easier and out-of-pocket costs more affordable, insights on patients insured under federal or state government prescription drug programs, etc.
Reimbursement is a crucial factor that affects the drug’s access to the market. Often, the decision to reimburse comes down to the price of the drug relative to the benefit it produces in treated patients. To reduce the healthcare burden of these high-cost therapies, many payment models are being considered by payers and other industry insiders.
NOTE: Further Details are provided in the final report….
Idiopathic Membranous Nephropathy Therapies Price Scenario & Trends
Pricing and analogue assessment of Idiopathic Membranous Nephropathy therapies highlights evolving price dynamics structures. This section summarizes the cost of emerging treatments and understanding of how pricing influences market access, adherence, and long-term uptake.
Pricing of Idiopathic Membranous Nephropathy Emerging Drugs
Pricing for SNP-ACTH (1–39) Gel was benchmarked against repository corticotrophin injection therapies, considering its chronic injectable use and niche immunologic positioning, resulting in an estimated annual US cost of USD 105,936. Similarly, ALPN-303 (povetacicept) was priced using rituximab-based anti-CD20 biologics as analogues, reflecting its B-cell–targeted biologic mechanism and anticipated use in immune-mediated conditions, with an estimated annual US cost of USD 85,000.
Industry Experts and Physician Views for Idiopathic Membranous Nephropathy
To keep up with Idiopathic Membranous Nephropathy market trends, we take Key Opinion Leaders (KOLs) and Subject Matter Experts (SMEs) opinions working in the domain through primary research to fill the data gaps and validate our secondary research. Industry Experts were contacted for insights on the Idiopathic Membranous Nephropathy emerging therapies, evolving treatment landscape, patient adherence to conventional therapies, therapy switching trends, drug adoption and uptake, accessibility challenges, and epidemiology and real-world prescription patterns in Idiopathic Membranous Nephropathy, including MD, Ph.D, Instructor, Postdoctoral Researcher, Professor, Researcher, and others.
DelveInsight’s analysts connected with 15+ KOLs to gather insights at country level. Centers such as the UCL Centre for Nephrology, Rowland Hill Street, London, and Boston University School of Medicine, Department of Medicine, Renal Section, etc. were contacted. Their opinion helps understand and validate current and emerging Idiopathic Membranous Nephropathy therapies, highlight unmet medical needs, provide epidemiological context, and support strategic decisions for market access, therapy adoption, and pipeline prioritization in Idiopathic Membranous Nephropathy.
|
Region |
Key Opinion Leaders (KOLs) and Subject Matter Experts (SMEs) |
|
United Kingdom |
“The standard treatment for MN has been, for many years, the Ponticelli regimen, which is based on alternating cyclophosphamide and corticosteroid administration for 6 months. The anti-CD20 monoclonal antibody Rituximab has emerged as a viable first-line therapy.” |
|
United States |
“The prevalence rates are not very well studied since the kidney biopsy performed for diagnosis occurs at various points along the spectrum of disease, and the duration of clinical disease and the number of relapses can impact the total number of people suffering from this disease at any point in time. We use the following statement “the annual incidence rates of MN are estimated at 10–12 per million in North America and 2–17 per million in Europe,” but there are no data beyond this that I am aware of.” |
Idiopathic Membranous Nephropathy Qualitative Analysis: SWOT and Conjoint Analysis
We perform qualitative and market Intelligence analysis using various approaches, such as SWOT analysis and conjoint analysis.
In the SWOT analysis of Idiopathic Membranous Nephropathy, strengths, weaknesses, opportunities, and threats in terms of disease diagnosis, patient awareness, patient burden, competitive landscape, cost-effectiveness, and geographical accessibility of therapies are provided.
Conjoint analysis analyzes emerging Idiopathic Membranous Nephropathy therapies based on relevant attributes such as safety, efficacy, frequency of administration, route of administration, and order of entry. Scoring is given based on these parameters to analyze the effectiveness of therapy.
The team of analysts analyzes promising emerging therapies based on relevant attributes such as safety, efficacy, frequency of administration, route of administration, and order of entry. In efficacy, the trial’s primary and secondary outcome measures are evaluated, whereas the therapies’ safety is evaluated, wherein the acceptability, tolerability, and adverse events are majorly observed. In addition, the scoring is also based on the route of administration, order of entry, probability of success, and the addressable patient pool for each therapy. According to these parameters, the final weightage score and the ranking of the emerging therapies are decided.
Scope of the Idiopathic Membranous Nephropathy Market Report
- The Idiopathic Membranous Nephropathy market report covers a segment of key events, an executive summary, a descriptive overview of Idiopathic Membranous Nephropathy (Idiopathic Membranous Nephropathy), explaining their causes, signs and symptoms, pathogenesis, and currently available treatments.
- Comprehensive insight has been provided into the epidemiology segments and forecasts, the future growth potential of the diagnosis rate, and disease progression along treatment guidelines.
- Additionally, an all-inclusive account of both the current and emerging treatments, along with the elaborative profiles of late-stage and prominent therapies, will have an impact on the current treatment landscape.
- A detailed review of the Idiopathic Membranous Nephropathy market, historical and forecasted market size, market share by therapies, detailed assumptions, and rationale behind our approach is included in the report, covering the 7MM drug outreach.
- The Idiopathic Membranous Nephropathy market report provides an edge while developing business strategies by understanding trends through SWOT analysis and expert insights/KOL views, patient journey, and treatment preferences that help in shaping and driving the 7MM Idiopathic Membranous Nephropathy market.
Idiopathic Membranous Nephropathy Market Report Insights
- Idiopathic Membranous Nephropathy Patient Population Forecast
- Idiopathic Membranous Nephropathy Therapeutics Market Size
- Idiopathic Membranous Nephropathy Pipeline Analysis
- Idiopathic Membranous Nephropathy Market Size and Trends
- Idiopathic Membranous Nephropathy Market Opportunity (Current and forecasted)
Idiopathic Membranous Nephropathy Market Report Key Strengths
- Epidemiology-based (Epi-based) Bottom-up Forecasting
- Artificial Intelligence (AI)-enabled Market Research Report
- 11-year forecast
- Idiopathic Membranous Nephropathy Market Outlook (North America, Europe, Asia-Pacific)
- Patient Burden Trends (by geography)
- Idiopathic Membranous Nephropathy Treatment Addressable Market (TAM)
- Idiopathic Membranous Nephropathy Competitive Landscape
- Idiopathic Membranous Nephropathy Major Companies Insights
- Idiopathic Membranous Nephropathy Price Trends and Analogue Assessment
- Idiopathic Membranous Nephropathy Therapies Drug Adoption/Uptake
- Idiopathic Membranous Nephropathy Therapies Peak Patient Share Analysis
Idiopathic Membranous Nephropathy Market Report Assessment
- Idiopathic Membranous Nephropathy Current Treatment Practices
- Idiopathic Membranous Nephropathy Unmet Needs
- Idiopathic Membranous Nephropathy Clinical Development Analysis
- Idiopathic Membranous Nephropathy Emerging Drugs Product Profiles
- Idiopathic Membranous Nephropathy Market Attractiveness
- Idiopathic Membranous Nephropathy Qualitative Analysis (SWOT and conjoint analysis)
Frequently Asked Questions from Idiopathic Membranous Nephropathy Market Report
Idiopathic Membranous Nephropathy Market Insights
- What was the Idiopathic Membranous Nephropathy market size, the market size by therapies, market share (%) distribution in 2025, and what would it look like by 2036? What are the contributing factors for this growth?
- What are the anticipated pricing variations among different geographies for the emerging therapies in the future?
- What can be the future treatment paradigm of Idiopathic Membranous Nephropathy?
- What impact will patent expiry have on the Idiopathic Membranous Nephropathy therapy market?
- What are the disease risks, burdens, and unmet needs of Idiopathic Membranous Nephropathy? What will be the growth opportunities across the 7MM concerning the patient population with Idiopathic Membranous Nephropathy?
- Who is the major future competitor in the market, and how will the competitors affect their market share?
- What are the current options for the treatment of Idiopathic Membranous Nephropathy? What are the current guidelines for treating Idiopathic Membranous Nephropathy in the US, Europe, and Japan?
Reasons to Buy the Idiopathic Membranous Nephropathy Market Report
- The Idiopathic Membranous Nephropathy market report will help in developing business strategies by understanding the latest trends and changing treatment dynamics driving the Idiopathic Membranous Nephropathy market.
- Bottom up forecasting builds from the affected population to product forecasts, delivering a robust, data driven approach ideal for new therapies and novel classes.
- Insights on patient burden/disease incidence, evolution in diagnosis, and factors contributing to the change in the epidemiology of the disease during the forecast years.
- Understand the existing market opportunities in varying geographies and the growth potential over the coming years.
- Identifying strong upcoming Idiopathic Membranous Nephropathy companies in the Idiopathic Membranous Nephropathy market will help devise strategies to help get ahead of competitors.
- Detailed analysis and ranking of class-wise potential current and emerging therapies under the conjoint analysis section to provide visibility around leading classes.
- To understand KOLs’ perspectives on the accessibility, acceptability, and compliance-related challenges of existing treatment to overcome barriers in the future.
- Detailed insights on the unmet needs of the existing Idiopathic Membranous Nephropathy market so that the upcoming players can strengthen their development and launch strategy.
- This Artificial Intelligence (AI) enabled report summarize and simplify complex datasets within the report into clear, actionable insights for stakeholders, investors, and healthcare providers, enabling faster, data driven decisions.