Paroxysmal Nocturnal Hemoglobinuria Market Summary
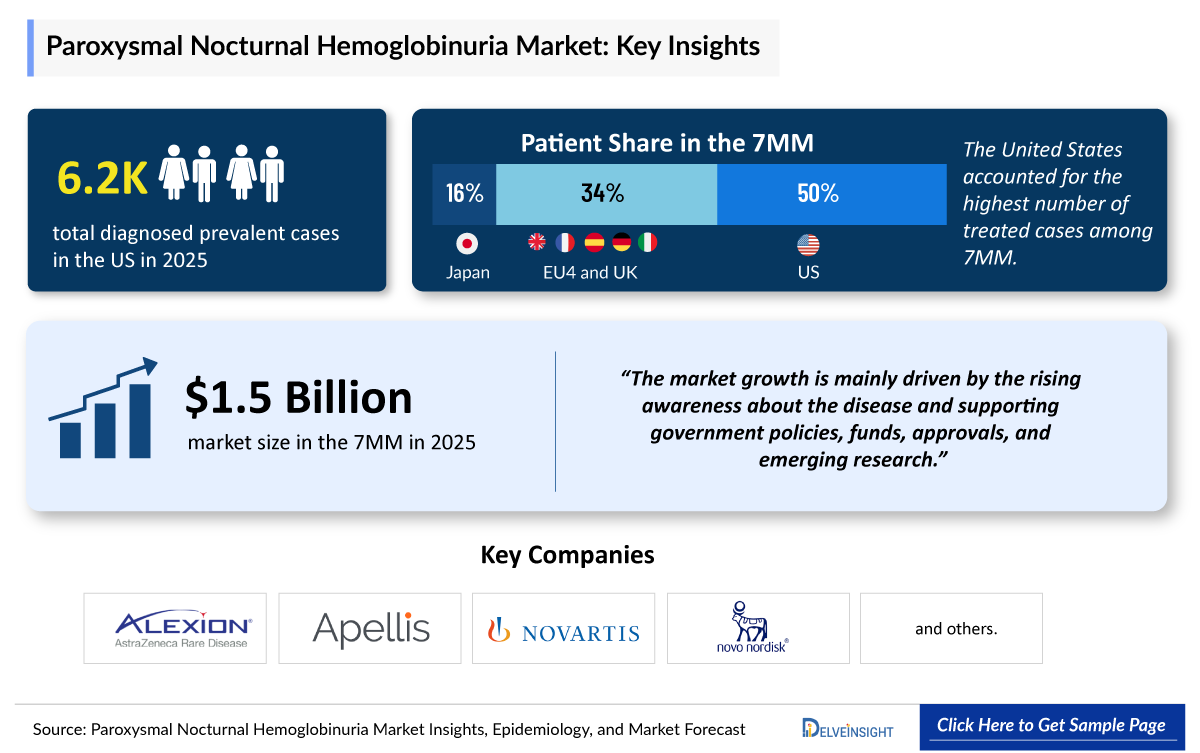
- According to DelveInsight’s analysis, PNH market size was more than USD 1 billion in the leading markets (the United States, the EU4 (Germany, France, Italy, and Spain), the United Kingdom, and Japan) in 2025.
Paroxysmal Nocturnal Hemoglobinuria Market Insights and Trends
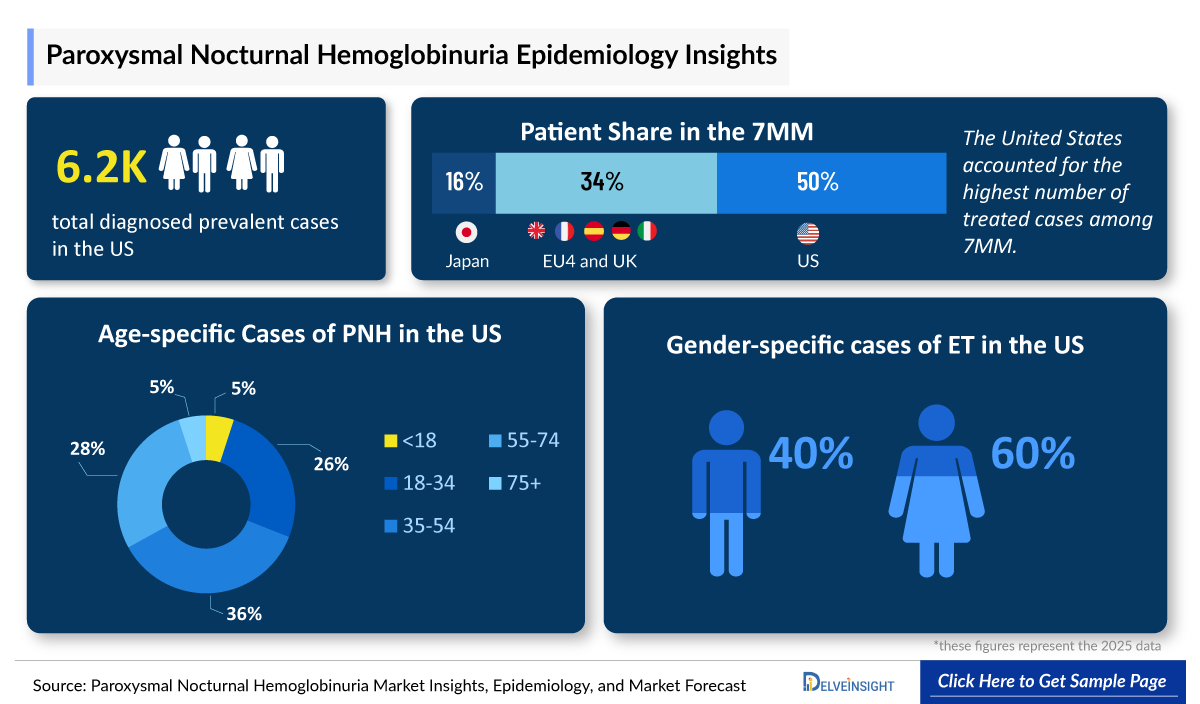
- PNH reported approximately around 6,200 diagnosed prevalent cases in the US in 2025. Gender-specific cases of PNH accounted for approximately 2,900 and 3,300 for male and female, respectively
- Formerly, PNH diagnosis relied on Ham and sucrose tests, now replaced by flow cytometry (FCM) detecting GPI-anchored protein anomalies using monoclonal antibodies or fluorescein-tagged proaerolysin (FLAER). FLAER is suitable for nucleated cells but not red blood cells due to glycophorin interference. Also, the lack of international guidelines for proper screening and diagnosis leads to treatment delays. The main clinical conditions or diseases to consider in the differential diagnosis of PNH include coombs-negative hemolytic anemia, venous thrombosis, anemia, and cytopenias.
- The disease-modifying therapeutic strategy for PNH includes complement inhibition therapy, with drugs like eculizumab (SOLIRIS), ravulizumab (ULTOMIRIS), and pegcetacoplan (EMPAVELI) approved by the FDA, and considered the gold standard. However, safety concerns are associated with eculizumab and ravulizumab, including the risk of meningococcal infections and the persistence of extravascular hemolysis in some individuals treated with eculizumab.
- Investigational drugs, including complement C5 inhibitors and different pathway inhibitors like MASP-3 inhibitors and the drugs which are included in the pipeline, are pozelimab (REGN3918), OMS906 (zaltenibart) and others, which are being evaluated to advance the management of PNH. Many drugs are also in their early stages of clinical development; hence the pipeline ensures better treatment in upcoming years.
- PNH is a rare disorder characterized by a unique triad of clinical features: intravascular hemolysis, thromboembolic events, and cytopenia. However, not all three manifestations are present in all patients, and each patient’s presentation may vary according to the most dominant signs and symptoms. Many investigators have tried classifying PNH according to the most typical clinical presentations. However, distinct categories are hard to define for a disease with unpredictable presentation and evolution. Additionally, complex and multifaceted prothrombotic states arise from the pathological effects of platelet activation, intravascular hemolysis, and neutrophil/monocyte activation. Furthermore, platelet and endothelial microparticles, as well as oxidative stress, may contribute to these states.
Paroxysmal Nocturnal Hemoglobinuria Market Size and Forecast
- 2025 PNH Market Size in the 7MM: ~USD 1.5 billion
- PNH Growth Rate (2026-2036): 7.1% CAGR
Request for Unlocking the Sample Page of the "Paroxysmal Nocturnal Hemoglobinuria Treatment Market"
Factors Contributing to the Growth of the Paroxysmal Nocturnal Hemoglobinuria Market
-
Increasing Disease Awareness
Growing awareness among healthcare professionals and patients is leading to earlier diagnosis and improved treatment adoption.
-
Advancements in Diagnostic Techniques
Improved diagnostic tools, such as flow cytometry, are enabling accurate and timely detection of Paroxysmal Nocturnal Hemoglobinuria.
-
Rising Prevalence of Rare Hematologic Disorders
Enhanced identification and reporting of rare diseases like PNH are contributing to a larger diagnosed patient pool.
-
Innovative Therapeutic Developments
Continuous R&D efforts are leading to the development of novel targeted therapies, particularly complement inhibitors.
-
Strong Pipeline of Emerging Drugs
A robust pipeline with promising candidates from various pharmaceutical companies is expected to drive future market growth.
-
Increasing Adoption of Targeted Therapies
Shift from traditional treatments to targeted biologics is improving patient outcomes and boosting market demand.
-
Favorable Regulatory Support
Orphan drug designations, fast-track approvals, and other incentives are encouraging companies to invest in PNH therapeutics.
-
Improved Healthcare Infrastructure
Better access to advanced healthcare facilities and specialized treatment centers is facilitating disease management.
-
Higher Healthcare Expenditure
Increasing healthcare spending across major markets is supporting access to high-cost therapies.
-
Growing Focus on Personalized Medicine
Advancements in precision medicine are enabling more effective and individualized treatment approaches.
-
Rising Patient Advocacy and Support Programs
Active patient organizations are promoting awareness, funding research, and improving access to treatments.
DelveInsight’s “Paroxysmal Nocturnal Hemoglobinuria Market Insights, Epidemiology, and Market Forecast – 2036” report delivers an in-depth understanding of PNH, historical and forecasted epidemiology, as well as the PNH market trends in the United States, EU4 (Germany, France, Italy, and Spain), the United Kingdom, and Japan.
The Paroxysmal Nocturnal Hemoglobinuria Treatment Market Report delivers a comprehensive analysis of the current treatment landscape, including standards of care, clinical practices, and evolving therapeutic algorithms. It evaluates, PNH patient burden trends, revenue & market share dynamics, peak patient share & therapy uptake analysis, and provides an in-depth market size assessment, and growth rate projections (Historical & Forecast 2022–2036) across the 7MM regions. The report highlights key unmet medical needs in PNH and maps the competitive and clinical landscape to uncover high‑value opportunities, providing a clear outlook on future market growth potential.
|
Study Period |
2022–2036 |
|
Historical Year |
2022–2025 |
|
Forecast Period |
2026–2036 |
|
Base Year |
2025 |
|
Geographies Covered |
ü North America : The US ü Europe: Germany, France, Italy, Spain and the UK ü Asia-Pacific: Japan |
|
Paroxysmal Nocturnal Hemoglobinuria Market CAGR (Forecast period) |
7.1% (2022-2036) |
|
Paroxysmal Nocturnal Hemoglobinuria Epidemiology Segmentation Analysis |
Patient Burden Assessment
|
|
Paroxysmal Nocturnal Hemoglobinuria Companies |
And more |
|
Paroxysmal Nocturnal Hemoglobinuria Therapies |
And more |
|
Paroxysmal Nocturnal Hemoglobinuria Market |
Segmented by
|
|
Analysis |
|
Paroxysmal Nocturnal Hemoglobinuria Understanding and Treatment Algorithm
Paroxysmal Nocturnal Hemoglobinuria Overview and Diagnosis
PNH is a rare acquired disorder that affects the pluripotent hematopoietic stem cells, which can impact erythrocytes, leukocytes, thrombocytes, and potentially some endothelial cells. This condition arises from a somatic mutation in the X-linked PIG-A gene, which is involved in synthesizing the glycosylphosphatidylinositol (GPI) anchor required for attaching certain proteins to the cell membrane.
To confirm a definite diagnosis of PNH, the following two criteria must be met:
- Presence of PNH-type Red Blood Cells: 1% or more of the red blood cells are PNH-type (cells lacking GPI-anchored membrane proteins).
- High Serum LDH Levels: Serum LDH (lactate dehydrogenase) levels are 1.5 times above the normal limit.
Further details related to country-based variations are provided in the report…
Current PNH Treatment Landscape
- Treatment of subclinical PNH and PNH associated with aplastic anemia: In subclinical PNH, patients do not have any clinical symptoms and do not need any treatment. It is necessary to closely monitor them (every 6–12 months) because an expansion of the clone may occur, and their disease may evolve into classic PNH.
- Treatment of classic PNH: Management of classic PNH was dramatically revolutionized by the development of eculizumab (SOLIRIS) and its approval in 2007 by the US FDA and the European Medicine Agency (EMA) for treating PNH. This humanized monoclonal antibody blocks the activation of terminal complement C5 and prevents the formation of C5a and C5b‐9, preventing hemolysis. Other drugs, such as ravulizumab (ULTOMIRIS) and pegcetacoplan (EMPAVELI), have also been FDA-approved recently. These drugs provide the same benefits as eculizumab with a more convenient and effective dosing schedule.
Further details related to country-based variations are provided in the report…
Paroxysmal Nocturnal Hemoglobinuria Unmet Needs
The section “unmet needs of PNH” outlines the critical gaps between the current state of patient care, diagnosis, and the ideal & effective management of the disease. It highlights the obstacles experienced by patients, clinicians, and researchers and identifies potential solutions for future progress.
1. Limited therapeutic options
2. Need to redefine clinical endpoints
3. Complex disease presentation
4. Pregnancy and PNH
5. Challenges associated with treatment using C5 inhibitors and others…..
Note: Comprehensive unmet needs insights in PNH and their strategic implications are provided in the full report…
Paroxysmal Nocturnal Hemoglobinuria Epidemiology
Key Findings from Paroxysmal Nocturnal Hemoglobinuria Epidemiological Analysis and Forecast
· In 2025, the US accounted for the highest diagnosed prevalent cases of PNH with approximately 6,200 cases, which are expected to increase by 2036.
· In 2025, the UK reported the highest number of diagnosed prevalent cases of PNH among EU4 and the UK, with approximately 2,600 cases. Germany followed with more than 1,300 cases, while Italy recorded the lowest diagnosed prevalence, with nearly 250 cases.
· In 2025, Japan reported approximately 400 diagnosed prevalent cases of PNH in females and nearly 540 cases in males, with numbers projected to rise by 2036.
· In 2025, the treated cases of PNH in Japan were nearly 650. These cases are expected to remain relatively stable in the coming years.
Market Share (%) Distribution of Paroxysmal Nocturnal Hemoglobinuria by Therapies in the 7MM in 2025
Paroxysmal Nocturnal Hemoglobinuria Drug Analysis & Competitive Landscape
The PNH drug chapter provides a detailed, market-focused review of approved therapies and the emerging pipeline across Phase I-III clinical trials. It covers mechanism of action, clinical trial data, regulatory approvals, patents, collaborations, strategic partnerships upcoming key catalyst for each therapy, along with their advantages, limitations, and recent developments. This section offers critical insights into the PNH treatment landscape, supporting market assessment, competitive analysis, and growth forecasting for the PNH therapeutics market.
Approved Therapies for Paroxysmal Nocturnal Hemoglobinuria
Ravulizumab (ULTOMIRIS): AstraZeneca (Alexion Pharmaceuticals)
ULTOMIRIS is a complement inhibitor for treating adult patients with PNH. It is designed to inhibit a specific aspect of the complement component of the immune system and thereby treat inflammation associated with chronic disorders in several therapeutic areas, including hematology, nephrology, and neurology. It is a humanized monoclonal antibody that effectively blocks terminal complement activity at prescribed doses. ULTOMIRIS, approved in US, EU and Japan, is the first and only long-acting C5 inhibitor that provides immediate and complete inhibition for 8 weeks.
- In December 2018, the US FDA granted marketing approval for ravulizumab (ULTOMIRIS) for PNH, a rare, disabling, and life-threatening blood disorder defined by chronic red blood cell destruction, or hemolysis. It continues to be a leading long-acting complement C5 inhibitor for PNH, offering sustained disease control, reduced treatment burden, and strong real-world effectiveness.
|
PNH Marketed/Approved Therapies | ||||||
|
Drug/Therapy |
Company |
Indication |
Molecule Type |
MoA |
RoA |
Marketed Region |
|
Ravulizumab (ULTOMIRIS) |
Alexion Pharmaceuticals/AstraZeneca |
PNH |
Monoclonal antibody |
C5 complement inhibitor |
IV/SC |
US: 2018 EU: 2019 JP: 2019 |
|
Pegcetacoplan (EMPAVELI/ ASPAVELI) |
Apellis Pharmaceuticals |
PNH |
Monoclonal antibody |
C3 complement inhibitor |
SC |
US: 2021 EU: 2021 JP:2023 |
|
Iptacopan (FABHALTA) |
Novartis |
PNH |
Small molecule |
Complement factor B inhibitor |
Oral |
US:2023 EU: 2024 JP: 2024 |
Note: Detailed marketed therapies assessment will be provided in the final report…
Paroxysmal Nocturnal Hemoglobinuria Pipeline Analysis
Zaltenibart (OMS906): Novo Nordisk
Zaltenibart is a humanized monoclonal antibody targeting mannan-binding lectin-associated serine protease-3 (MASP-3), the key activator of the alternative pathway of complement. The alternative pathway is implicated in a wide range of diseases and disorders, including those targeted by marketed alternative pathway inhibitors and/or those in development. However, unlike C3 and C5 inhibitors, MASP-3 inhibition does not interfere with the lytic arm of the classical pathway of complement, which is a critical component of the acquired immune response to infection.
- In December 2025, Omeros Corporation announced the closing of the asset sale and licensing transaction between Omeros and Novo Nordisk for the candidate drug zaltenibart (OMS906).
- In October 2025, Novo Nordisk and Omeros Corporation announced that they had entered into a definitive asset purchase and license agreement for the candidate drug zaltenibart (OMS906).
- In March 2025, Omeros Corporation reported the activation of clinical trial sites for patient enrollment in its Phase III program assessing zaltenibart in PNH.
|
Drug Name |
Company |
Highest Phase |
Indication |
RoA |
MoA |
Anticipated Launch in the US |
|
Pozelimab + Cemdisiran |
Regeneron Pharmaceuticals |
III |
PNH |
IV/SC |
C5 complement inhibitor |
2030 |
|
Zaltenibart (OMS906) |
Novo Nordisk |
III |
PNH |
IV |
MASP-3 inhibitor |
Information is available in the full report |
|
Note: Launch insights are provisional and may change with future report updates or the occurrence of major key catalysts. | ||||||
Note: Detailed emerging therapies assessment will be provided in the final report…
Paroxysmal Nocturnal Hemoglobinuria Key Players, Market Leaders and Emerging Companies
- Alexion Pharmaceuticals/AstraZeneca
- Apellis Pharmaceuticals
- Novartis
- Regeneron Pharmaceuticals
- Novo Nordisk
And more
PNH Drug Updates
- In March 2025, Omeros Corporation reported that clinical trial site activation for enrollment was underway for the company’s Phase III program evaluating zaltenibart in PNH.
- In August 2024, Roche announced today that the European Commission has approved PIASKY for adults and adolescents (12 years of age or older with a weight of 40 kg and above) with PNH who are either new to, or have been previously treated with C5 inhibitors.
- In August 2024, iptacopan (FABHALTA) was approved by Medicines and Healthcare products Regulatory Agency (MHRA) for PNH.
Paroxysmal Nocturnal Hemoglobinuria Market Outlook
Until 2007, PNH was a devastating disease without treatment for hemolysis and thrombosis, the leading cause of death in patients. However, the last decade saw a revolutionary shift with the advent of the anti-C5 agent eculizumab. It significantly reduced hemolysis, decreased transfusion dependency, and, crucially, lowered the thrombosis rate. The disease-modifying therapeutic strategy for paroxysmal nocturnal hemoglobinuria includes complement inhibition therapy, with drugs like SOLIRIS, ULTOMIRIS, and EMPAVELI approved by the FDA and considered the gold standard. SOLIRIS, the pioneering therapy for PNH, was succeeded by ULTOMIRIS, both developed by Alexion Pharmaceuticals, offering C5 inhibitors as essential therapeutic options. Recent approvals of Factor B and D inhibitors like iptacopan (FABHALTA) and danicopan (VOYDEYA) aim to offer improved treatment with fewer side effects.
· In 2025, the PNH market size in the US was around USD 950 million. This figure is expected to grow significantly with the introduction of emerging therapies.
· The total market size of EU4 and the UK was estimated to be approximately USD 500 million in 2025.
· Among EU4 and the UK, the UK accounted for the highest market share with approximately USD 240 million in 2025, followed by Germany with more than USD 130 million in the respective year, and Italy, capturing the least market with nearly USD 20 million in 2025.
· In 2025, the total market size of PNH was approximately USD 90 million in Japan which is anticipated to increase during the forecast period (2026–2036).
Drug Class/Insights into Leading Emerging Therapies in Paroxysmal Nocturnal Hemoglobinuria (2022–2036 Forecast)
- Monoclonal antibody: Monoclonal antibodies have become a cornerstone in the treatment of Paroxysmal Nocturnal Hemoglobinuria. These biologic therapies are designed to bind with high specificity to complement proteins, thereby inhibiting the cascade that leads to hemolysis. A prime example is ravulizumab (ULTOMIRIS), a humanized monoclonal antibody that effectively blocks terminal complement activity at prescribed doses. ULTOMIRIS is notable for being a long-acting C5 inhibitor, providing immediate and complete complement inhibition for up to 8 weeks, which reduces the frequency of infusions compared to earlier treatments.
- Small molecule: Small-molecule proximal complement inhibitors in PNH target early elements of the alternative complement pathway. FABHALTA is an oral small-molecule Factor B inhibitor that blocks the synthesis of the C3 convertase (C3bBb) by targeting the alternative complement pathway proximally.
Paroxysmal Nocturnal Hemoglobinuria Drug Uptake
This section focuses on the uptake rate of potential drugs expected to be launched in the market during the forecast period (2026–2036). The analysis covers the PNH drug’s uptake, performance at peak, factors affecting performance during prime years of growth, patient uptake by therapy, and anticipated sales generated by each drug.
The introduction of the C5 inhibitor SOLIRIS (eculizumab), followed by ravulizumab (ULTOMIRIS), has transformed the disease by greatly reducing intravascular hemolysis, thrombosis, and death, with life expectancies similar to age-matched controls.
Among the emerging therapies, Pozelimab is expected to generate the highest revenue in the 7MM, followed by Zaltenibart. In a Phase II study, Zaltenibart demonstrated strong efficacy in PNH patients with suboptimal responses to ravulizumab.
Detailed insights of emerging therapies' drug uptake is included in the report…
Market Access and Reimbursement of therapies in Paroxysmal Nocturnal Hemoglobinuria
The report further provides detailed insights on the country-wise accessibility and reimbursement scenarios, cost-effectiveness scenario of therapies, programs making accessibility easier and out-of-pocket costs more affordable, insights on patients insured under federal or state government prescription drug programs, etc.
Reimbursement is a crucial factor that affects the drug’s access to the market. Often, the decision to reimburse comes down to the price of the drug relative to the benefit it produces in treated patients. To reduce the healthcare burden of these high-cost therapies, many payment models are being considered by payers and other industry insiders.
NOTE: Further Details are provided in the final report….
EU4 and the UK
|
Drug/Therapy |
IQWiG Assessment |
|
Ravulizumab |
No suitable data was available for assessing the added benefit of ravulizumab in comparison with the ACT. This resulted in no hint of an added benefit of ravulizumab in comparison with the ACT. |
Reimbursement is a crucial factor that affects the drug’s access to the market. Often, the decision to reimburse comes down to the price of the drug relative to the benefit it produces in treated patients. To reduce the healthcare burden of these high-cost therapies, many payment models are being considered by payers and other industry insiders.
NOTE: Further Details are provided in the final report.
Paroxysmal Nocturnal Hemoglobinuria Therapies Price Scenario & Trends
Pricing and analogue assessment of PNH therapies highlights evolving price dynamics structures. This section summarizes the cost of approved treatments, closest and most appropriate analogue selection for emerging therapies, and understanding of how pricing influences market access, adherence, and long-term uptake.
- Pricing of PNH Approved Drugs
Ravulizumab (ULTOMIRIS), developed by AstraZeneca, is a long-acting C5 inhibitor for PNH. It has a treatment course cost of around USD 500,000 in the US.
Industry Experts and Physician Views for Paroxysmal Nocturnal Hemoglobinuria
To keep up with PNH market trends, we take Key Opinion Leaders (KOLs) and Subject Matter Experts (SMEs) opinions working in the domain through primary research to fill the data gaps and validate our secondary research. Industry Experts were contacted for insights on the PNH emerging therapies, evolving treatment landscape, patient adherence to conventional therapies, therapy switching trends, drug adoption and uptake, accessibility challenges, and epidemiology and real-world prescription patterns in PNH, including MD, PhD, Instructor, Postdoctoral Researcher, Professor, Researcher, and others.
DelveInsight’s analysts connected with 10+ KOLs to gather insights at country level. Centers such as the Faculty of Medicine, University of Tsukuba, University of Glasgow, Duke University, Washington University School of Medicine, The Yale School of Medicine etc. were contacted.
|
Region |
Key Opinion Leaders (KOLs) and Subject Matter Experts (SMEs) |
|
United States |
“Alternative complement pathway inhibitors may offer further benefit as long as terminal complement is completely inhibited to reduce IVH and disease activity. This may lead to improvements in adherence and health-related quality of life for patients with PNH.” |
|
Germany |
“Pegcetacoplan (EMPAVELI) is the first PNH treatment that targets C3 and can inhibit both IVH and EVH and dramatically improve the lives of PNH patients by raising hemoglobin and decreasing the need for blood transfusions.” |
Their opinion helps understand and validate current and emerging PNH therapies, highlight unmet medical needs, provide epidemiological context, and support strategic decisions for market access, therapy adoption, and pipeline prioritization in PNH.
Qualitative Analysis: SWOT and Attribute Analysis
We perform qualitative and market Intelligence analysis using various approaches, such as SWOT analysis and attribute analysis.
In the SWOT analysis of PNH, strengths, weaknesses, opportunities, and threats in terms of disease diagnosis, patient awareness, patient burden, competitive landscape, cost-effectiveness, and geographical accessibility of therapies are provide attribute analysis analyzes emerging therapies based on relevant attributes such as safety, efficacy, frequency of administration, route of administration, and order of entry. Scoring is given based on these parameters to analyze the effectiveness of therapy.
The team of analysts analyzes promising emerging therapies based on relevant attributes such as safety, efficacy, frequency of administration, route of administration, and order of entry. In efficacy, the trial’s primary and secondary outcome measures are evaluated, whereas the therapies’ safety is evaluated, wherein the acceptability, tolerability, and adverse events are majorly observed. In addition, the scoring is also based on the route of administration, order of entry, probability of success, and the addressable patient pool for each therapy. According to these parameters, the final weightage score and the ranking of the emerging therapies are decided.
Scope of the Report
· The report covers a segment of key events, an executive summary, a descriptive overview of PNH, explaining their causes, signs and symptoms, pathogenesis, and currently available treatments.
· Comprehensive insight has been provided into the epidemiology segments and forecasts, the future growth potential of the diagnosis rate, and disease progression along treatment guidelines.
· Additionally, an all-inclusive account of both the current and emerging treatments, along with the elaborative profiles of late-stage and prominent therapies, will have an impact on the current treatment landscape.
· A detailed review of the PNH market, historical and forecasted market size, market share by therapies, detailed assumptions, and rationale behind our approach is included in the report, covering the 7MM drug outreach.
· The report provides an edge while developing business strategies by understanding trends through SWOT analysis and expert insights/KOL views, patient journey, and treatment preferences that help in shaping and driving the 7MM PNH market.
Report Insights
· Paroxysmal Nocturnal Hemoglobinuria Patient Population Forecast
· Paroxysmal Nocturnal Hemoglobinuria Therapeutics Market Size
· Paroxysmal Nocturnal Hemoglobinuria Pipeline Analysis
· Paroxysmal Nocturnal Hemoglobinuria Market Size and Trends
· Paroxysmal Nocturnal Hemoglobinuria Market Opportunity (Current and Forecasted)
Report Key Strengths
· Epidemiology‑based (Epi‑based) Bottom‑up Forecasting
· Artificial Intelligence (AI) - enabled Market Research Report
· 11-year forecast
· Paroxysmal Nocturnal Hemoglobinuria Market Outlook (North America, Europe, Asia-Pacific)
· Patient Burden Trends (by geography)
· Paroxysmal Nocturnal Hemoglobinuria Treatment Addressable Market (TAM)
· Paroxysmal Nocturnal Hemoglobinuria Competitive Landscape
· Paroxysmal Nocturnal Hemoglobinuria Major Companies Insights
· Paroxysmal Nocturnal Hemoglobinuria Price Trends and Analogue Assessment
· Paroxysmal Nocturnal Hemoglobinuria Therapies Drug Adoption/Uptake
· Paroxysmal Nocturnal Hemoglobinuria Therapies Peak Patient Share analysis
Report Assessment
· Paroxysmal Nocturnal Hemoglobinuria Current Treatment Practices
· Paroxysmal Nocturnal Hemoglobinuria Unmet Needs
· Paroxysmal Nocturnal Hemoglobinuria Clinical Development Analysis
· Paroxysmal Nocturnal Hemoglobinuria Emerging Drugs Product Profiles
· Paroxysmal Nocturnal Hemoglobinuria Market Attractiveness
· Paroxysmal Nocturnal Hemoglobinuria Qualitative Analysis (SWOT and Attribute analysis)
FAQs
Market Insights
- What was the PNH market size, the market size by therapies, market share (%) distribution in 2025, and what would it look like by 2036? What are the contributing factors for this growth?
- What are the anticipated pricing variations among different geographies for the emerging therapies in the future?
- What can be the future treatment paradigm of PNH?
- What are the disease risks, burdens, and unmet needs of PNH? What will be the growth opportunities across the 7MM concerning the patient population with PNH?
- Who is the major future competitor in the market, and how will the competitors affect their market share?
- What are the current options for the treatment of PNH? What are the current guidelines for treating PNH in the US, Europe, and Japan?
Reasons to Buy
The report will help in developing business strategies by understanding the latest trends and changing treatment dynamics driving the PNH market.
Bottom‑up forecasting builds from the affected population to product forecasts, delivering a robust, data‑driven approach ideal for new therapies and novel classes.
Insights on patient burden/disease prevalence, evolution in diagnosis, and factors contributing to the change in the epidemiology of the disease during the forecast years.
Understand the existing market opportunities in varying geographies and the growth potential over the coming years.
Identifying strong upcoming players in the market will help devise strategies to help get ahead of competitors.
Detailed analysis and ranking of class-wise potential current and emerging therapies under the attribute analysis section to provide visibility around leading classes.
To understand KOLs’ perspectives on the accessibility, acceptability, and compliance-related challenges of existing treatment to overcome barriers in the future.
Detailed insights on the unmet needs of the existing market so that the upcoming players can strengthen their development and launch strategy.
This Artificial Intelligence (AI) ‑ enabled report summarize and simplify complex datasets with in the report into clear, actionable insights for stakeholders, investors, and healthcare providers, enabling faster, data‑driven decisions.