Anastomosis Device Market Summary
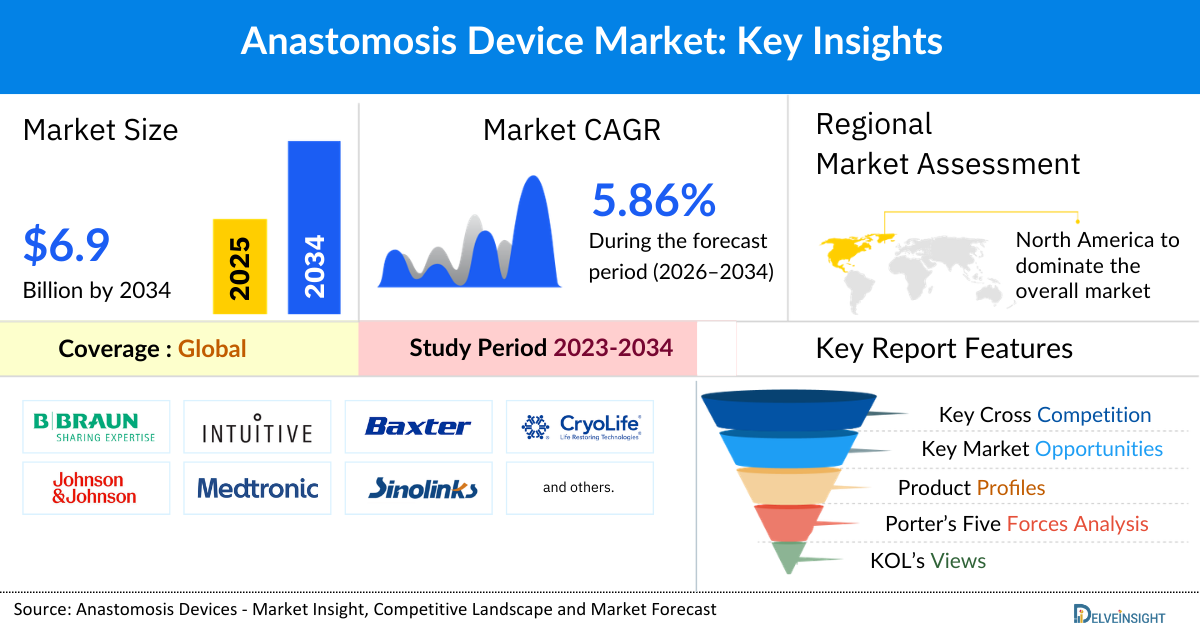
- The global anastomosis devices market size is expected to increase from USD 4,159.35 million in 2025 to USD 6,922.84 million by 2034, reflecting strong and sustained growth.
- The global anastomosis devices market is growing at a CAGR of 5.86% during the forecast period from 2026 to 2034.
- The market of anastomosis devices is being primarily driven by higher demand for surgical procedures due to the expanding burden of chronic illness, increasing adoption of minimally invasive surgical technologies and robotic assisted surgeries, and rising focus on product development initiatives among the key market players.
- The leading companies operating in the anastomosis devices market include B.Braun SE, Intuitive Surgical Operations, Inc., Baxter, Johnson & Johnson Services Inc., Medtronic, Sinolinks Medical Innovation, Inc., Dolphin Sutures, Golden Stapler surgical Co. Ltd., Changzhou Haiers Medical Devices Co., Ltd., Getinge AB, Terumo Corporation, ConMed Corporation, Teleflex Corporation, Dextera Surgical Inc., Peters Surgical, MIZUHO Corporation, CarpoNovum Corporation, Purple Surgical, Seger Surgical Solutions Ltd., Biowin Pharmaceuticals Co., Ltd., and others.
- North America is expected to dominate the overall anastomosis devices market due to its well-established healthcare infrastructure, high surgical procedure volumes, and early adoption of advanced surgical technologies. The region has a strong presence of leading medical device manufacturers and continuous investment in research and product innovation, which accelerates the availability of technologically advanced anastomosis devices. In addition, favorable reimbursement policies, higher healthcare spending, and widespread adoption of minimally invasive and robotic-assisted surgeries support market growth. The rising prevalence of chronic disorders which require surgical resection and reconnection of tissues such as cardiovascular disorders, colorectal cancer, and gastrointestinal conditions further drives the demand for anastomosis devices, while the presence of skilled surgeons and advanced clinical training programs enhances their adoption across hospitals and ambulatory surgical centers. These factors collectively position the region as a leading contributor to the global market.
- In the product segment of the anastomosis devices market, the surgical stapplers category is estimated to account for the largest market share in 2025.
Request for unlocking the report of the @ Anastomosis Devices Market Insights
Anastomosis Devices Market Size and Forecasts
|
Report Metrics |
Details |
|
2025 Market Size |
4,159.35 million |
|
2034 Projected Market Size |
6,922.84 million |
|
Growth Rate (2026-2034) |
5.86% CAGR |
|
Largest Market |
North America |
|
Fastest Growing Market |
Asia-Pacific |
|
Market Structure |
Moderately to Highly Concentrated |
Factors Contributing to the Growth of the Anastomosis Devices Market
- Higher demand for surgical procedures due to the expanding burden of chronic illnesses: The expanding burden of chronic illnesses such as cardiovascular diseases, gastrointestinal disorders, cancer, and diabetes has led to a higher demand for surgical procedures. Anastomosis devices play a critical role in these surgeries by enabling faster, more precise, and consistent tissue joining compared to traditional suturing methods. As the number of complex and repeat surgeries increases, hospitals and surgeons are increasingly adopting anastomosis devices to reduce operating time, minimize surgical complications, and improve patient outcomes. This growing reliance on surgical interventions for chronic disease management is directly driving the demand for advanced anastomosis devices across healthcare settings.
- Increasing adoption of minimally invasive surgical technologies and robotic assisted surgeries: The growing use of minimally invasive surgical techniques is driving the expansion of the anastomosis devices market by changing how complex surgeries are performed. Procedures like laparoscopic and robotic-assisted surgeries require precise, reliable, and efficient methods for connecting tissues, boosting demand for advanced anastomosis devices over traditional manual suturing. These devices allow surgeons to create secure and consistent connections while reducing operating time, blood loss, and complication risks. Because minimally invasive surgeries also offer benefits such as shorter hospital stays, faster recovery, and less post-operative pain, hospitals and surgical centers are increasingly adopting anastomosis devices to enhance procedural efficiency and patient outcomes, fueling market growth.
- Rising focus on product development initiatives: The rising focus on product development initiatives is significantly contributing to the growth of the anastomosis devices market by driving continuous innovation and improving surgical outcomes. Manufacturers are investing in the development of advanced anastomosis devices that offer better precision, reduced operating time, and lower risk of complications such as leakage or infection. New product designs, including minimally invasive, automated, and bio-compatible anastomosis systems, are making procedures safer and easier for surgeons, which encourages wider adoption in hospitals and surgical centers. Additionally, ongoing product improvements help expand the use of anastomosis devices across different surgical specialties, such as gastrointestinal, cardiovascular, and colorectal surgeries, thereby increasing overall market demand.
Anastomosis Devices Market Report Segmentation
This Anastomosis devices market report offers a comprehensive overview of the global Anastomosis devices market, highlighting key trends, growth drivers, challenges, and opportunities. It covers detailed market segmentation by Product (Surgical Staplers {Powered, Manual}, Surgical Sutures, Surgical Sealant and Adhesives, and Others), Usability (Disposable and Reusable), Application (Cardiovascular Surgery, Gastrointestinal Surgery, General Surgery, and Others), End-User (Hospitals & Clinics, Ambulatory Surgical Centers, and Others), and Geography. The report provides valuable insights into the competitive landscape, regulatory environment, and market dynamics across major markets, including North America, Europe, and Asia-Pacific. Featuring in-depth profiles of leading industry players and recent product innovations, this report equips businesses with essential data to identify market potential, develop strategic plans, and capitalize on emerging opportunities in the rapidly growing anastomosis devices market.
Anastomosis devices are medical instruments designed to surgically connect two hollow structures, such as blood vessels or segments of the intestine. They enable surgeons to create secure and precise connections during procedures including gastrointestinal, cardiovascular, and colorectal surgeries.
The overall anastomosis devices market is being collectively boosted by several converging factors, including the rising volume of surgical procedures and the growing burden of chronic diseases such as cardiovascular disorders, cancer, and gastrointestinal conditions which often require surgical resection and reconnection of tissues. Additionally, increased adoption of minimally invasive and robotic-assisted surgeries are driving demand for advanced anastomosis devices that offer precision and faster recovery. Furthermore, continuous product development and technological advancements are improving device safety, ease of use, and clinical outcomes, encouraging wider adoption among surgeons. Favorable healthcare infrastructure, improved reimbursement in developed regions, and increasing awareness about the benefits of automated and stapling-based anastomosis techniques are further supporting market growth. Together, these factors are accelerating adoption across hospitals, clinics, and ambulatory surgical centers, fueling robust growth in the anastomosis devices market.
Get More Insights into the Report @ Anastomosis Devices Market Trends
What are the latest anastomosis devices market dynamics and trends?
The global market for anastomosis devices has witnessed significant growth in recent years, largely driven by increasing demand for surgical procedures due to the expanding burden of chronic illnesses such as cardiovascular diseases and gastrointestinal diseases.
Impact of Cardiovascular Diseases:
According to the National Library of Medicine (2025), the global burden of peripheral artery disease (PAD) is rising sharply, with an estimated 113 million people affected in 2021, a number projected to increase to 189 million by 2030, 273 million by 2040, and 363 million by 2050. Peripheral Arterial Disease is characterized by progressive arterial narrowing and blockage, often in the lower limbs, which frequently necessitates bypass surgeries, endarterectomy, and revascularization procedures when medical therapy or angioplasty is insufficient. These procedures rely heavily on vascular anastomosis devices such as coupling systems, proximal anastomosis devices, and suturing or stapling tools to restore blood flow efficiently and safely.
Impact of Gastrointestinal Diseases
According to the International Agency for Research on Cancer (2025), approximately 2.06 million new cases of colorectal cancer were reported worldwide in 2025, and this number is projected to rise to around 3.29 million by 2045. The growth of colorectal cancer cases is mainly because its treatment often requires surgical intervention. Surgery is a primary approach for managing colorectal cancer and typically involves removing the affected section of the bowel and reconnecting the remaining parts using anastomosis devices such as staplers and sutures.
However, the increasing product development activities auch as approvals, new product launches among the key market players are further escalating the market.For Example, in September 2024, GT Metabolic Solutions Inc., a global leader in magnetic compression anastomosis surgery, announced that the U.S. Food and Drug Administration had cleared its MagDI™ System for side-to-side duodeno-ileal (DI) anastomosis, marking a significant milestone in the use of innovative magnetic compression technology for digestive surgery.
However, one of the major challenges in the anastomosis devices market is the risk of anastomotic leakage and other post-operative complications. Anastomotic leakage occurs when the surgical connection between two hollow structures, such as blood vessels or intestinal segments, fails to seal properly, allowing fluids or contents to escape. This can lead to serious complications, including infection, prolonged hospital stays, additional surgeries, and even life-threatening conditions. Because of this risk, surgeons may be cautious in adopting new or advanced anastomosis devices, especially if clinical evidence supporting their safety and reliability is limited. As a result, device manufacturers must focus on improving precision, durability, and safety features to reduce leakage rates, which directly influences the market adoption and growth of anastomosis devices.
Anastomosis Devices Market Segment Analysis
Anastomosis Devices Market by Product (Surgical Staplers {Powered, Manual}, Surgical Sutures, Surgical Sealant, Surgical Adhesives, and Others), Usability (Disposable and Reusable), Application (Cardiovascular Surgery, Gastrointestinal Surgery, General Surgery, and Others), End-Users (Hospitals & Clinics, Ambulatory Surgical Centers, and Others), and Geography (North America, Europe, Asia-Pacific, and Rest of the World)
By Product: Powered Staplers Projected to Register Fastest Growth
In the product segment of anastomosis devices, the powered staplers the subsegment of surgical staplers is projected to grow at the fastest CAGR of 6.54% during the forecast period from 2026 to 2034. Powered staplers are surgical anastomosis devices that use a battery- or electrically driven mechanism to join tissues or organs by placing rows of staples during surgery. They are commonly used in procedures such as colorectal, gastrointestinal, and thoracic surgeries where sections of tissue need to be cut and reconnected.
In anastomosis procedures, powered staplers help create secure and consistent connections between two ends of the intestine or other tissues, improving precision and reducing manual effort for surgeons. Their controlled staple formation and tissue compression support better surgical outcomes and lower the risk of complications like leakage or bleeding. These devices support both open and minimally invasive surgeries by providing consistent staple formation, precise tissue alignment, and reliable anastomotic sealing. Their use helps improve surgical efficiency and outcomes, making them an important tool in procedures that require safe and effective tissue reconnection. These devices shorten operative time and support uniform staple lines, lowering the risk of complications such as leakage or bleeding at the anastomosis site.
A notable milestone in this space came in June 2025, Johnson & Johnson MedTech, a global leader in surgical technologies and solutions, announced the U.S. launch of the ETHICON™ 4000 Stapler, an advanced surgical stapler designed to manage complex tissue conditions and deliver strong staple line integrity. The device was developed to help reduce the risk of surgical leaks and bleeding complications across a wide range of surgical specialties.
By Usability: Disposable Category Dominates the Market
In the global anastomosis devices market, the disposable segment is dominating current usage and revenue share, and this trend is expected to continue in the near-term due to clinical, logistical, and safety advantages. The disposable anastomosis devices accounted for around 80% of the overall market share in 2025.
The primary reason for this dominance is that disposable devices inherently reduce risk of surgical site infections and cross-contamination, a critical factor in cardiovascular and gastrointestinal surgeries where patient outcomes and sterility protocols are paramount. Hospitals and surgical centers favor single-use staples, suturing tools, and connectors because they eliminate the need for complex reprocessing, sterilization validation, and associated workflow burden, allowing faster procedure turnaround and more predictable performance.
From a product innovation standpoint, many recent product launches have focused on disposable technologies, including advanced staplers with bioabsorbable rings, magnetic or leak-detecting features, and integration with minimally invasive laparoscopic and robotic-assisted systems. These innovations not only enhance clinical precision but also align with the broader shift toward single-use, procedure-specific instruments that streamline OR logistics.
By Application: Cardiovascular Surgery Category Dominates the Market
In the global anastomosis devices market, cardiovascular surgery stands out as the dominant application segment and is expected to retain this leadership position over the forecast period. In 2025, cardiovascular surgeries accounted for the largest revenue share, estimated at about 51% of the market, significantly higher than gastrointestinal surgery and other applications such as general or urological procedures. This dominance is largely driven by the high prevalence of cardiovascular diseases worldwide, the aging global population, and the large volume of procedures such as coronary artery bypass grafting (CABG) that fundamentally rely on reliable anastomosis devices to connect blood vessels during open heart and vascular surgeries. These factors combine to make cardiovascular surgeries the most significant application area for these devices.
One of the core reasons cardiovascular surgery leads the market is disease burden and surgical necessity: cardiovascular diseases remain the leading cause of death globally, and surgical interventions are often critical in managing complex cases such as severe coronary or peripheral artery disease. This creates a consistently high procedure volume that sustains strong device demand. Technologies like automated and powered anastomosis systems, including magnetic compression platforms, robot-assisted connectors, and powered staplers designed specifically for cardiac tissue work, are being adopted to optimize outcomes in these high-risk, high-stakes environments. For example, the MagDI magnetic compression anastomosis system received FDA clearance and expanded with a larger 50 mm magnet in 2025, allowing broader use in surgical cases such as duodeno-ileal anastomosis with a cardiovascular focus and emphasizing magnetic techniques in vascular reconnections.
By End-User: Hospitals Category Dominates the Market
The hospitals category dominates the anastomosis devices market with a market share of 68% in 2025 because hospitals handle the largest volume of complex and high-risk surgical procedures that require reliable tissue and vessel connections. Major surgeries such as gastrointestinal resections, colorectal surgeries, cardiovascular bypass procedures, and cancer-related operations are primarily performed in hospital settings, driving consistent demand for anastomosis devices.
Hospitals are also better equipped with advanced operating rooms, skilled surgeons, and multidisciplinary surgical teams, enabling the adoption of technologically advanced anastomosis devices, including surgical staplers, automated systems, and minimally invasive tools. In addition, hospitals benefit from better reimbursement coverage, higher healthcare spending, and access to continuous training programs, which further support the use of advanced surgical technologies.
The availability of post-operative care and intensive monitoring within hospitals also encourages surgeons to perform complex anastomosis procedures in these settings, reinforcing hospitals’ dominant position in the overall anastomosis devices market.
Hence, owing to all the aforementioned factors, the hospitals category is expected to account for a significant revenue share in the Anastomosis devices market during the forecast period.
Anastomosis Devices Market Regional Analysis
North America Anastomosis Devices Market Trends
North America is expected to account for the highest proportion of 42% of the anastomosis devices market in 2025, out of all regions. North America is expected to dominate the overall anastomosis devices market due to its well-established healthcare infrastructure, high surgical procedure volumes, and early adoption of advanced surgical technologies. The region has a strong presence of leading medical device manufacturers and continuous investment in research and product innovation, which accelerates the availability of technologically advanced anastomosis devices.
According to the Centers for Disease Control and Prevention (2024), approximately 6.5 million people aged 40 years and older in the United States were living with peripheral artery disease (PAD). This high prevalence of PAD significantly boosted the anastomosis devices market, as many patients required vascular interventions such as bypass and revascularization surgeries to restore blood flow. These procedures rely heavily on precise and secure vessel connections, increasing the demand for advanced anastomosis devices that improve surgical efficiency, reduce complications, and support better patient outcomes.
Additionally, according to the Centers for Disease Control and Prevention (2024), the prevalence of inflammatory bowel disease (IBD) in the United States was estimated to be between 2.4 and 3.1 million people, with up to 3.1 million adults affected based on national survey data. This substantial patient population has contributed to the growth of the anastomosis devices market, as a significant number of IBD patients require surgical intervention when medical therapies are no longer effective. Surgical treatment for conditions such as Crohn’s disease and ulcerative colitis often involves removing diseased sections of the intestine and reconnecting healthy bowel segments, a procedure that relies on anastomosis devices. As the incidence of IBD increased, the demand for safe, reliable, and efficient anastomosis devices used in gastrointestinal surgeries also rose, thereby supporting overall market expansion.
However, the increasing product development activities among the key market players are further escalating the market. For instance, in August 2025, Phraxis, Inc., a leader in vascular access innovation, announced the successful completion of the first commercial case using its EndoForce™ Anastomotic Connector, marking a historic milestone in the treatment of dialysis patients worldwide.
Therefore, the above-mentioned factors are expected to bloster the growth of the anastomosis devices market in North America during the forecast period.
Europe Anastomosis Devices Market Trends
In Europe, the groth of anastomosis devices market is driven by a high prevalence of chronic diseases, and rising demand for complex surgeries. Strong healthcare infrastructure, early adoption of advanced, minimally invasive, and robotic-assisted surgical technologies further drive market growth and adoption across hospitals and surgical centers.
According to the International Agency for Research on Cancer (2025), around 552,000 new colorectum cancer cases were reported in 2025, and this number is expected to rise to approximately 682,000 by 2045.The rising number of cancer cases, especially colorectal and gastrointestinal cancers, is driving demand for anastomosis devices, as these surgeries often require reconnecting intestinal or hollow organ sections. Higher cancer prevalence leads to more surgical procedures, increasing the need for safe and efficient anastomosis devices.
Furthermore, the increasing product development activities maong the key market players are further escalating the market. For instance, in August 2025, SafeHeal®, a leading innovator in colorectal cancer surgery, announced that it had been granted European Union marketing approval for its Colovac® device under the new medical device regulation.
Asia-Pacific Anastomosis Devices Market Trends
The anastomosis devices market in the Asia-Pacific region is being driven by the rising incidence of chronic diseases, including cardiovascular disorders, gastrointestinal conditions, and cancer, which increase the demand for surgical procedures.
According to the National Library of Medicine(2025), the incidence of inflammatory bowel disease (IBD) in Japan was 2,456 cases in 2022, and the forecasted incidence per 100,000 population for ulcerative colitis (UC) and Crohn’s disease in 2032 was projected to be 33.28 and 5.68 respectively. Inflammatory bowel disease (IBD), which includes Crohn’s disease and ulcerative colitis, is increasing the demand for anastomosis devices because many patients eventually require surgery when medications are not effective. In severe cases, surgeons need to remove damaged or inflamed parts of the intestine and reconnect the healthy sections, a process that requires anastomosis devices such as staplers and sutures. Therefore, the increasing number of IBD cases is driving higher demand for safe, reliable, and efficient anastomosis devices used in gastrointestinal surgeries. As a result, the demand for Anastomosis devices has surged, driving the growth of the Asia-Pacific market for these devices.
Furthermore, according to the International Agency for Research on Cancer(2025), the estimated number of new colorectal cancer cases in Australia was 18,500 in 2025, and this number was expected to rise to 26,900 by 2045. The rising number of colorectal cancer cases is boosting the anastomosis devices market because many patients need surgery to remove cancerous sections of the colon or rectum. After removing the diseased tissue, surgeons must reconnect the healthy parts of the intestine, a process that relies on anastomosis devices. As more people are diagnosed with colorectal cancer, the demand for these devices increases to ensure safe, precise, and efficient surgical outcomes. Additionally, the demand for safe, precise, and minimally invasive surgical solutions drives wider adoption of advanced anastomosis devices.
Who are the major players in the anastomosis devices market?
The following are the leading companies in the anastomosis devices market. These companies collectively hold the largest market share and dictate industry trends.
- B.Braun SE
- Intuitive Surgical Operations, Inc.
- Baxter
- Johnson & Johnson Services Inc.
- Medtronic
- Sinolinks Medical Innovation, Inc.
- Biowin Pharmaceuticals Co., Ltd.
- Dolphin Sutures
- Golden Stapler Surgical Co., Ltd.
- Changzhou Haiers Medical Devices Co., Ltd.
- Getinge AB
- Terumo Corporation
- ConMed Corporation
- Teleflex Corporation
- Dextera Surgical Inc.
- Peters Surgical
- MIZUHO Corporation
- CarpoNovum AB
- Purple Surgical .
- Seger Surgical Solutions Ltd., and others
How is the competitive landscape shaping the anastomosis devices market?
The competitive landscape of the anastomosis devices market is shaped by intense innovation, strategic partnerships, regulatory approvals, and product differentiation as companies strive to offer safer, more efficient solutions. Major medical device manufacturers are constantly upgrading their portfolios with advanced staplers, automated systems, and minimally invasive technologies to meet surgeon preferences and reduce complications. In addition, competition is driven by technological advancements such as magnetic compression anastomosis, bio absorbable connectors, and smart devices with integrated feedback, which help improve procedural outcomes. Key players are also engaging in mergers, acquisitions, and collaborations to expand geographic reach and strengthen clinical presence, while regulatory clearances and real world evidence play an important role in gaining market acceptance. Overall, competition encourages continuous product development and broader access to next generation anastomosis devices across hospitals and surgical centers worldwide.
Recent Developmental Activities in the Anastomosis Devices Market
- In April 2025, Intuitive, a global leader in minimally invasive care and a pioneer in robotic-assisted surgery, announced that the U.S. Food & Drug Administration (FDA) had cleared its fully wristed SP SureForm 45 stapler for use with the da Vinci SP surgical system in thoracic, colorectal, and urologic procedures.
- In March 2025, GT Metabolic™ Solutions, Inc., a global leader in magnetic surgery, announced that the U.S. Food and Drug Administration (FDA) had cleared a larger, 50mm MagDI™ System magnet to complement the system’s existing 40mm magnet. This expansion allowed surgeons to perform magnet-assisted duodenal-ileal anastomosis on a broader range of patients.
- In September 2024, GT Metabolic™ Solutions, Inc., a company focused on reinventing bariatric, metabolic, and digestive surgery, announced that its first-generation MagDI™ System for side-to-side duodeno-ileal anastomosis had received De Novo marketing authorization from the U.S. Food and Drug Administration (FDA).In June 2024, Aktiia received regulatory approval for the world’s first optical blood pressure system, CALFREE™, which can be used without the need for calibration with a regular cuff.
- In August 2025, Phraxis, Inc., a leader in vascular access innovation, announced the successful completion of the first commercial case using its EndoForce™ Anastomotic Connector, marking a historic milestone in the treatment of dialysis patients worldwide.
- In May 2024, Ethicon, a Johnson & Johnson MedTech company, announced the U.S. launch of the ECHELON LINEAR™ Cutter. It became the first linear cutter on the market to combine two innovative proprietary technologies 3D-Stapling Technology and Gripping Surface Technology (GST) both designed to enhance staple line security, helping surgeons reduce risks and improve patient outcomes.
|
Report Metrics |
Details |
|
Study Period |
2023 to 2034 |
|
Base Year |
2025 |
|
Forecast Period |
2026 to 2034 |
|
Anastomosis Devices Market CAGR |
5.86% |
|
Key Companies in the Anastomosis Devices Market |
B.Braun SE, Intuitive Surgical Operations, Inc., Baxter, Johnson & Johnson and its affiliates, Medtronic, Sinolinks Medical Innovation, Inc., BIOWIN PHARMACEUTICAL CO., LIMITED, DOLPHIN SUTURES, Golden Stapler Surgical Co., Ltd, Changzhou Haiers Medical Devices Co., Ltd, Getinge AB, Terumo Corporation, ConMed Corporation, Teleflex Incorporated, Dextera Surgical Inc., Peters surgical, MIZUHO Corporation, CarpoNovum AB, Purple Surgical, LP, Seger Surgical Solutions Ltd. and others. |
|
Anastomosis Devices Market Segments |
by Product, by Usability, by Application, by End-user, and by Geography |
|
Anastomosis Devices Regional Scope |
North America, Europe, Asia Pacific, Middle East, Africa, and South America |
|
Anastomosis Devices Country Scope |
U.S., Canada, Mexico, Germany, United Kingdom, France, Italy, Spain, China, Japan, India, Australia, South Korea, and key Countries |
Anastomosis Devices Market Segmentation
- Anastomosis Devices by Product Exposure
- Surgical Staplers
- Powered
- Manual
- Surgical Sutures
- Surgical Sealant
- Surgical Adhesive
- Others
- Surgical Staplers
- Anastomosis Devices by Usability Exposure
- Disposable
- Reusable
- Anastomosis Devices by Application Exposure
- Cardiovascular Surgery
- Gastrointestinal Surgery
- General Surgery
- Others
- Anastomosis Devices End-Users Exposure
- Hospitals & Clinics
- Ambulatory Surgical Centers
- Others
Anastomosis Devices Geography Exposure
- North America Anastomosis Devices Market
- United States Anastomosis Devices Market
- Canada Anastomosis Devices Market
- Mexico Anastomosis Devices Market
- Europe Anastomosis Devices Market
- United Kingdom Anastomosis Devices Market
- Germany Anastomosis Devices Market
- France Anastomosis Devices Market
- Italy Anastomosis Devices Market
- Spain Anastomosis Devices Market
- Rest of Europe Anastomosis Devices Market
- Asia-Pacific Anastomosis Devices Market
- China Anastomosis Devices Market
- Japan Anastomosis Devices Market
- India Anastomosis Devices Market
- Australia Anastomosis Devices Market
- South Korea Anastomosis Devices Market
- Rest of Asia-Pacific Anastomosis Devices Market
- Rest of the World Anastomosis Devices Market
- South America Anastomosis Devices Market
- Middle East Anastomosis Devices Market
- Africa Anastomosis Devices Market
Anastomosis Devices Market Recent Industry Trends and Milestones (2022-2026):
|
Category |
Key Developments |
|
Anastomosis Devices Regulatory Approvals |
Phraxis Inc. – EndoForce™ Connector(FDA), GT Metabolic Solutions – MagDI™ System (FDA De Novo Authorization), SafeHeal® – Colovac® Anastomosis Protection Device (EU MDR), Intuitive Surgical Inc. – SP SureForm 45 Stapler (FDA). |
|
Product Launch in the Anastomosis Devices Market |
Johnson & Johnson launched its ETHICON™ 4000 Stapler and ECHELON LINEAR™ Cutter. |
|
Acquisition in the Anastomosis Devices Market |
Advanced Medical Solutions PLC (AMS acquired Peters Surgical. |
|
Company Strategy |
B.Braun: B. Braun’s strategy centers on innovation and quality, expanding its surgical device portfolio, including anastomosis products. The company emphasizes precision instruments and ergonomic designs for reliable surgical performance, while investing in R&D, digital solutions, and advanced technologies like robotics and automation to enhance workflow and outcomes. |
|
Emerging Technology |
Disposable/powered devices, robotic-assisted anastomosis devices, bioabsorbable and smart materials, ai-powered surgical systems. |
Impact Analysis
AI-Powered Innovations and Applications:
AI-powered innovations are starting to transform the anastomosis devices market by making surgeries safer, faster, and more precise. Artificial intelligence can assist surgeons in planning and performing complex procedures by analyzing patient data, predicting risks, and guiding optimal device placement. Some AI-enabled systems can monitor tissue alignment, detect potential leaks, and provide real-time feedback during anastomosis, reducing complications. These technologies are especially useful in minimally invasive and robotic-assisted surgeries, where precision is critical. By improving surgical outcomes, shortening operation times, and enhancing overall efficiency, AI-powered applications are driving the adoption of advanced anastomosis devices in hospitals and surgical centers worldwide.
U.S. Tariff Impact Analysis on Anastomosis Devices Market:
Tariffs imposed by the United States on imported medical devices can affect the anastomosis devices market in several ways. When tariffs increase the cost of imported components or finished products, manufacturers may face higher production expenses, which can lead to higher prices for hospitals and healthcare providers. This can slow down the adoption of advanced anastomosis devices, especially in cost sensitive healthcare facilities. In some cases, tariffs may encourage manufacturers to shift production to domestic suppliers or invest more in local manufacturing to avoid extra costs. While this can strengthen U.S. based production in the long term, in the short term higher tariffs can make devices more expensive and limit market growth by reducing demand or delaying technology upgrades in surgical centers.
How This Analysis Helps Clients
- Cost Management: By understanding the tariff landscape, clients can anticipate cost increases and adjust pricing strategies accordingly, ensuring profitability.
- Supply Chain Optimization: Clients can identify alternative sourcing options and diversify their supply chains to reduce dependency on high-tariff regions, enhancing resilience.
- Regulatory Navigation: Expert guidance on navigating the evolving regulatory environment helps clients maintain compliance and avoid potential legal challenges.
- Strategic Planning: Insights into tariff impacts enable clients to make informed decisions about manufacturing locations, partnerships, and market entry strategies
Startup Funding & Investment Trends
|
Company Name |
Total Funding |
Main Products |
Stage of Development |
Core Technology |
|
GI Windows Surgical |
$37M |
Self-Forming Magnet, FlexagonTM |
Series B |
Uses magnetic forces and a flexible, foldable structure to create precise, controlled connections between tissues without the need for traditional sutures or staples. |
Key takeaways from the Anastomosis devices market report study
- Market size analysis for the current anastomosis devices market size (2025), and market forecast for 8 years (2026 to 2034)
- Top key product/technology developments, mergers, acquisitions, partnerships, and joint ventures happened over the last 3 years.
- Key companies dominating the anastomosis devices market.
- Various opportunities available for the other competitors in the anastomosis devices market space.
- What are the top-performing segments in 2025? How these segments will perform in 2034?
- Which are the top-performing regions and countries in the current anastomosis devices market scenario?
- Which are the regions and countries where companies should have concentrated on opportunities for the anastomosis devices market growth in the future?
Frequently Asked Questions for the Anastomosis Devices Market
1. What is the growth rate of the anastomosis devices market?
The anastomosis devices market is estimated to grow at a CAGR of 5.86% during the forecast period from 2026 to 2034.
2. What is the market for anastomosis devices?
The global anastomosis devices market is expected to increase from 4,159.35 million in 2025 to 6,922.84 million by 2034, reflecting strong and sustained growth.
3. Which region has the highest share in the anastomosis devices market?
North America is expected to account for the highest proportion of the anastomosis devices market in 2025, out of all regions. The region dominates the anastomosis devices market due to its strong healthcare infrastructure, high surgical volumes, and early adoption of advanced technologies. A presence of leading medical device manufacturers, continuous innovation, favorable reimbursement, and rising healthcare spending support growth. Additionally, the increasing prevalence of chronic diseases, along with skilled surgeons and advanced training programs, drives demand and adoption of anastomosis devices across hospitals and surgical centers.
4. What are the drivers for the anastomosis devices market?
The overall anastomosis devices market is being collectively boosted by several converging factors, including the rising volume of surgical procedures and the growing burden of chronic diseases such as cardiovascular disorders, cancer, and gastrointestinal conditions. Increased adoption of minimally invasive and robotic-assisted surgeries is driving demand for advanced anastomosis devices that offer precision and faster recovery. In addition, continuous product development and technological advancements are improving device safety, ease of use, and clinical outcomes, encouraging wider adoption among surgeons. Favorable healthcare infrastructure, improved reimbursement in developed regions, and increasing awareness about the benefits of automated and stapling-based anastomosis techniques are further supporting market growth. Together, these factors are accelerating adoption across hospitals, clinics, and ambulatory surgical centers, fueling robust growth in the Anastomosis devices market.
5. Who are the key players operating in the anastomosis devices market?
Some of the key market players operating in the anastomosis devices market include B.Braun SE, Intuitive Surgical Operations, Inc., Baxter, Johnson & Johnson Services Inc., Medtronic, Sinolinks Medical Innovation, Inc., Dolphin Sutures, Golden Stapler surgical Co. Ltd., Changzhou Haiers Medical Devices Co., Ltd., Getinge AB, Terumo Corporation, ConMed Corporation, Teleflex Corporation, Dextera Surgical Inc., Peters Surgical, MIZUHO Corporation, CarpoNovum Corporation, Purple Surgical, Seger Surgical Solutions Ltd., Biowin Pharmaceuticals Co., Ltd., others.