Epidermolysis Bullosa Market Summary
- The Epidermolysis Bullosa market in the 7MM is projected to grow at a significant CAGR through 2036 from ~2 Billion in 2024 in the leading countries (US, EU4, UK and Japan)
Epidermolysis Bullosa Market and Epidemiology Analysis
- Among the type-specific cases, more than 30% of prevalent cases were observed in dystrophic Epidermolysis Bullosa, and only 5% were found in junctional Epidermolysis Bullosa in 2023.
- There are limited effective therapies currently available for Epidermolysis Bullosa patients. Treatment and management options primarily focus on providing support, such as wound care, pain and itch management, infection control, nutritional support, and prevention and treatment of associated complications.
- Stem cell-based therapies are gaining significance, particularly for diseases that were previously considered incurable. ABCB5+ mesenchymal stromal cells (ABCB5+ MSCs) are noteworthy for their special immunomodulatory and anti-inflammatory properties, making them a promising therapeutic option for various chronic inflammatory diseases, including Epidermolysis Bullosa.
- In terms of overall cell therapy development, Japan is a bit ahead of other countries among the 7MM, and in December 2018, Japan became the first country where the first cell therapy for Epidermolysis Bullosa got approved i.e., JACE (human epidermal cell sheet) which is a product of Japan Tissue Engineering.
- Lack of approved therapies for epidermolysis bullosa, highlights the significant unmet need for epidermolysis bullosa patients.
- Krystal has taken the lead in the race for gene therapy treatments for epidermolysis bullosa by securing FDA approval for VYJUVEK, which is effective for both the recessive and dominant forms of the condition.
- Abeona has also generated compelling data from its gene therapy, EB-101, which operates similarly to VYJUVEK by introducing COL7A1 into the body. Although Krystal is 1-year ahead of Abeona in its development process, there is impending competition between the two therapies.
- Despite these approvals, there is still a long way to go in meeting all the needs of Epidermolysis Bullosa patients, and further research and development are essential to improve treatment options and ultimately enhance the quality of life for individuals living with Epidermolysis Bullosa worldwide.
- Epidermolysis Bullosa (EB) is a genetic skin disorder characterized by extreme skin fragility and blistering in response to minimal friction or trauma. The condition is caused by genetic mutations that affect the proteins responsible for maintaining the structural integrity of the skin. The severity of EB can vary widely, ranging from mild to life-threatening, depending on the specific subtype.
- Treatment for epidermolysis bullosa primarily focuses on symptom management and complication prevention. This includes implementing wound care, infection control, pain management, and providing appropriate nutritional support. For individuals with severe epidermolysis bullosa, specialized dressings, bandages, and protective clothing may be necessary to minimize skin trauma. Surgical intervention might also be required to address complications like hand deformities.
- VYJUVEK is an FDA-approved topical gene therapy for dystrophic epidermolysis bullosa (DEB), with recent label expansion enabling broader patient use and at-home administration. FILSUVEZ is approved in both the EU and the US for the treatment of wounds in patients with DEB and junctional EB. More recently, ZEVASKYN (prademagene zamikeracel) received FDA approval (April 2025) as the first autologous, cell-based gene therapy for recessive DEB, marking a significant advancement in the treatment landscape.
- Pharmaceutical companies and researchers have been actively conducting clinical trials and research to investigate potential therapies for EB. This ongoing effort reflects the growing interest and commitment to finding effective treatments for this rare and challenging genetic disorder.
- Regrettably, there are limited effective therapies currently available for epidermolysis bullosa patients. Treatment and management options primarily focus on providing support, such as wound care, pain and itch management, infection control, nutritional support, and prevention and treatment of associated complications.
- New therapies are emerging that target either the primary genetic defect or the secondary inflammatory footprint of the disease; treatment innovations include the early phase clinical testing of gene, cell-based, protein, and miscellaneous small molecule or repositioned drug therapies to help combat this debilitating disorder.
- The long-awaited approval of VYJUVEK in May 2023 marked the first-ever topical gene therapy in the United States, with subsequent approval in the EU further expanding its geographic availability.
- The pipeline of epidermolysis bullosa treatments includes several robust candidates that are poised to transform the market dynamics and address the unmet needs of patients.
- The competition is intensifying as Abeona has also achieved significant progress with its gene therapy, EB-101, which functions similarly to VYJUVEK by introducing COL7A1 into the body. Despite being one year behind Krystal in the development process, Abeona has shown promising results.
Download the Sample PDF to Get More Insight @ Epidermolysis Bullosa Market
Epidermolysis Bullosa Market Size and Forecast in the 7MM
- 2025 Epidermolysis Bullosa Market Size: ~USD 2,300 million
- Epidermolysis Bullosa Growth Rate (2026–2036): 7% CAGR
DelveInsight’s “Epidermolysis Bullosa Market Insights, Epidemiology and Market Forecast – 2036” report delivers an in-depth understanding of the Epidermolysis Bullosa, historical and forecasted epidemiology as well as the Epidermolysis Bullosa market trends in the United States, EU4 (Germany, Spain, Italy, and France) and the United Kingdom, and Japan.
The Epidermolysis Bullosa Treatment Market report provides current treatment practices, emerging drugs, market share of individual therapies, and current and forecasted 7MM Epidermolysis Bullosa market size from 2020 to 2034. The report also covers current Epidermolysis Bullosa treatment market practices/algorithms and Epidermolysis Bullosa unmet medical needs to curate the best opportunities and assess the market’s underlying potential.
Scope of the Epidermolysis Bullosa Market Report | |
|
Study Period |
2022–2036 |
|
Historical Year |
2022–2025 |
|
Forecast Period |
2026–2036 |
|
Base Year |
2026 |
|
Geographies Covered |
|
|
Epidermolysis Bullosa Market CAGR (Study period/Forecast period) |
7% (2026–2036) |
|
Epidermolysis Bullosa Epidemiology Segmentation Analysis |
Patient Burden Assessment
|
|
Epidermolysis Bullosa Companies |
|
|
Epidermolysis Bullosa Therapies |
|
|
Epidermolysis Bullosa Market |
Segmented by
|
|
Analysis |
|
Key Factors Driving the Epidermolysis Bullosa Market
Rising awareness and improved diagnosis
Growing awareness of epidermolysis bullosa among healthcare professionals and patients, along with advances in genetic testing and diagnostic technologies, is leading to earlier and more accurate identification of cases, thereby expanding the treatable patient pool.
Increasing prevalence and identification of rare genetic disorders
Although epidermolysis bullosa is rare, improved reporting and diagnostic capabilities are identifying more cases globally, contributing to a gradual increase in the patient population and sustained treatment demand.
Advancements in gene, cell, and regenerative therapies
The development and approval of innovative therapies, including gene therapy, cell-based treatments, and protein replacement approaches, are transforming disease management and driving market growth.
Strong pipeline and increasing R&D investments
Rising research and development activities, supported by regulatory incentives for rare diseases, are accelerating the development of novel, targeted therapies and expanding future market opportunities.
Epidermolysis Bullosa Understanding and Treatment Algorithm
Epidermolysis Bullosa Overview
Epidermolysis bullosa is a group of rare inherited skin disorders characterized by extreme skin fragility, causing blistering from minor trauma. It results from genetic mutations affecting proteins that maintain skin integrity. Epidermolysis bullosa is classified into major types: epidermolysis bullosa simplex, junctional epidermolysis bullosa, dystrophic epidermolysis bullosa, and kindler syndrome, with severity ranging from mild to life-threatening and possible systemic complications.
Epidermolysis Bullosa Diagnosis
Diagnosis of epidermolysis bullosa is based on clinical evaluation and confirmatory laboratory testing. Initial assessment includes detailed patient history and physical examination, focusing on blistering patterns, age of onset, and family history.
Definitive diagnosis typically involves specialized tests such as:
- Skin biopsy with immunofluorescence mapping (IFM): Determines the level of skin separation and helps classify EB subtype.
- Transmission electron microscopy (TEM): Provides detailed structural analysis of skin layers.
- Genetic testing: Identifies specific gene mutations, confirms diagnosis, and supports subtype classification as well as genetic counseling.
Early and accurate diagnosis is critical for appropriate disease management, prognosis assessment, and family planning.
Further details are provided in the report.
Epidermolysis Bullosa Treatment
Treatment of EB is primarily supportive and focuses on symptom management, as no definitive cure exists for most forms. Wound care is central, involving protective dressings, infection prevention, and pain control. Medications such as antibiotics, analgesics, and anti-inflammatory agents are used to manage infections and discomfort. Nutritional support is important, especially in severe cases, to promote healing and growth. Advanced approaches, including gene therapy, cell-based therapies, and protein replacement treatments, are under development and offer potential for disease modification. Multidisciplinary care is essential to manage complications and improve the quality of life.
Further details related to country-based variations are provided in the report.
Epidermolysis Bullosa Unmet Needs
The section “unmet needs of Epidermolysis Bullosa” outlines the critical gaps between the current state of patient care, diagnosis, and the ideal & effective management of the disease. It highlights the obstacles experienced by patients, clinicians, and researchers and identifies potential solutions for future progress.
- Lack of approved therapies
- Patients and family awareness
- Early diagnosis
- Deterioration of the quality of life of the patient, and others…..
Comprehensive unmet needs insights in epidermolysis bullosa and their strategic implications are provided in the full report.
Epidermolysis Bullosa Epidemiology
Key Findings from Epidermolysis Bullosa Epidemiological Analysis and Forecast
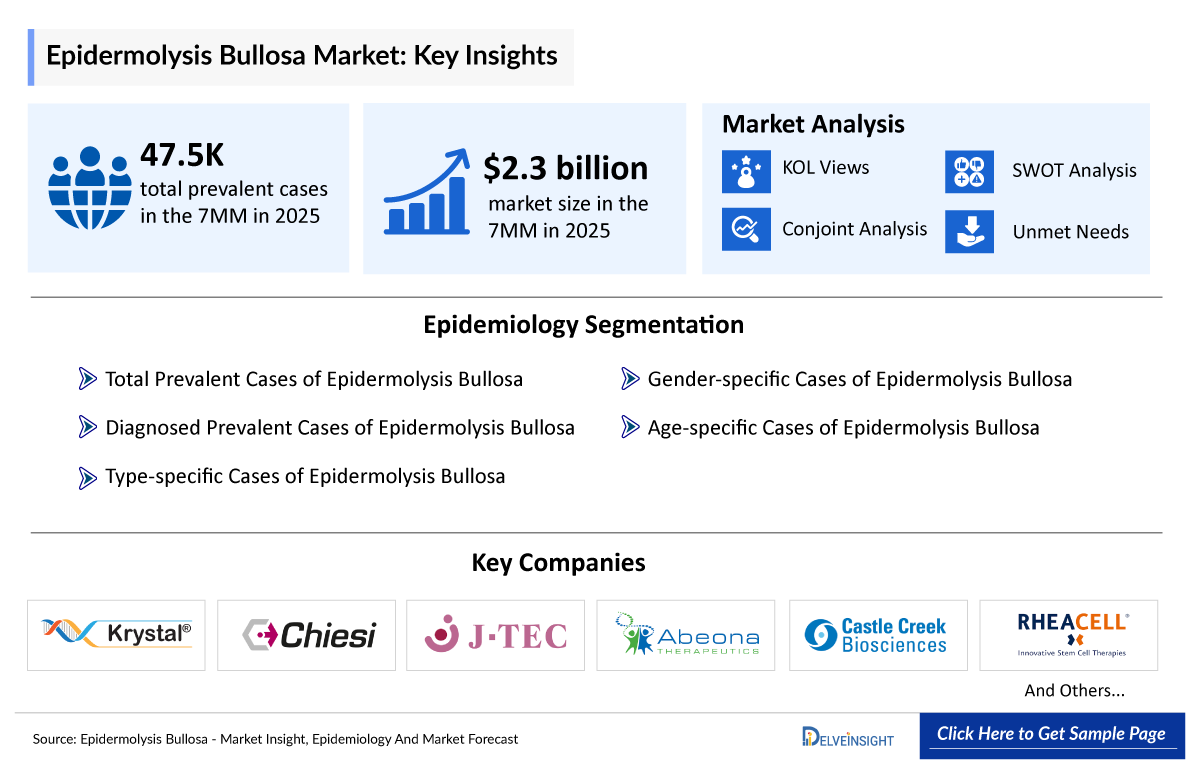
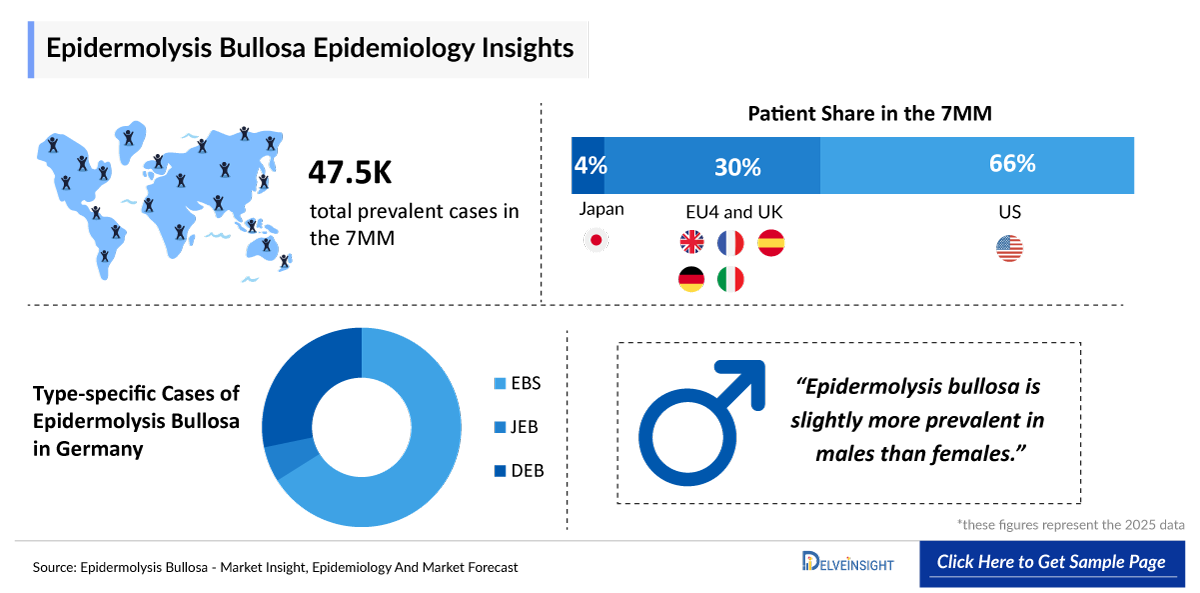
- The total prevalent cases of epidermolysis bullosa in the 7MM were ~47,500 cases in 2025, which is expected to increase during the forecast period (2026–2036).
- The highest number of prevalent cases was observed in the United States. On the other hand, within the EU4 and the UK, the UK accounted for the highest number of epidermolysis bullosa cases, with approximately 5,100 cases reported in 2025.
- In Japan, the 0-9 year age group has the highest contribution, followed by the 10-19 year age group, and then those over 19 years of age.
- Epidermolysis bullosa affects males and females equally. The condition is more prevalent in children and young adults, with approximately 78% of cases in the 7MM occurring in individuals under 19 years old in 2025.
- Epidermolysis bullosa simplex is the most common form of epidermolysis bullosa, accounting for approximately 61%, 5%, and 34% cases of EBS, JEB, and DEB in the 7MM in 2025.
Epidermolysis Bullosa Drug Chapters & Competitive Analysis
The epidermolysis bullosa drug chapter provides a detailed, market-focused review of approved therapies and the emerging pipeline across Phase I–III clinical trials. It covers the mechanism of action, clinical trial data, regulatory approvals, patents, collaborations, and strategic partnerships for each therapy, along with their advantages, limitations, and recent developments. This section offers critical insights into the epidermolysis bullosa treatment landscape, supporting market assessment, competitive analysis, and growth forecasting for the epidermolysis bullosa therapeutics market.
Approved Therapies for Epidermolysis Bullosa
VYJUVEK (beremagene geperpavec): Krystal Biotech
Beremagene geperpavec (B-VEC) is a non-invasive, topical, redosable gene therapy designed to deliver two copies of the COL7A1 gene when applied directly to DEB wounds. B-VEC was designed to treat DEB at the molecular level by providing the patient’s skin cells with the template to make normal COL7 protein, thereby addressing the fundamental disease-causing mechanism.
In September 2025, Krystal Biotech announced that the US FDA approved a label update for VYJUVEK that expands the VYJUVEK eligible patient population to include DEB patients from birth and provides patients full flexibility with respect to VYJUVEK application and managing wound dressings.
In July 2025, Krystal Biotech announced that the Japanese Ministry of Health, Labour and Welfare (MHLW) granted marketing authorization to VYJUVEK for the treatment of wounds in patients with DEB, starting from birth. VYJUVEK is the first genetic medicine approved in Japan for the treatment of DEB. The Japanese approval allows for dosing at home or in a healthcare setting, with the option for administration by patients or their family members.
FILSUVEZ (oleogel-S10): Chiesi Farmaceutici
FILSUVEZ is a herbal medicinal product that contains birch triterpenes from birch bark. This sterile gel is being developed to speed up the healing of skin wounds in adults and children with severe subtypes of epidermolysis bullosa; these subtypes are DEB and JEB. FILSUVEZ joined the Chiesi portfolio as part of the agreement reached during the acquisition of Amryt Pharma in January of this year.
In December 2023, Chiesi Global Rare Diseases announced that the US FDA approved FILSUVEZ (birch triterpenes) topical gel for the treatment of partial thickness wounds in patients 6 months and older with JEB and DEB. FILSUVEZ is the first approved treatment for wounds associated with JEB, a rare, moderate-to-severe form of epidermolysis bullosa with blisters beginning in infancy.
Epidermolysis Bullosa Marketed/Approved Therapies | ||||||
|
Drug/Therapy |
Company |
Indication |
Molecule Type |
RoA |
MoA |
Marketed Region |
|
VYJUVEK (beremagene geperpavec) |
Krystal Biotech |
Wounds in patients 6 months of age and older with DEB with mutations in the collagen type VII alpha 1 chain (COL7A1) gene |
Herpes simplex virus type 1 (HSV-1) vector-based gene therapy |
Topical gel |
Deliver two copies of the COL7A1 gene when applied directly to DEB wounds |
US: May 2023 EU: November 2023 JP: July 2025 |
|
FILSUVEZ (oleogel-S10) |
Chiesi Farmaceutici |
Partial thickness wounds associated with DEB and JEB in patients 6 months and older |
Birch bark extract (small molecule) |
Topical gel |
Accelerates wound closure through the effects of birch triterpenes involving keratinocyte differentiation and migration |
US: December 2022 EU: June 2022
|
Note: Detailed marketed therapies assessment will be provided in the final report.
Epidermolysis Bullosa Pipeline Analysis
D-Fi (dabocemagene autoficel): Castle Creek Biosciences
Using its proprietary ex vivo platform, Lenti-VEX, the company is developing and evaluating D-Fi, also known as FCX-007 (dabocemagene autoficel), an autologous gene therapy candidate to treat DEB, a progressive, devastatingly painful and debilitating, rare genetic skin disorder.
In October 2021, Castle Creek Biosciences announced that the US FDA’s Office of Orphan Products Development (OOPD) awarded the company a research grant of USD 1.825 million over four years. This funding supports a significant portion of the Phase III clinical development of FCX-007 for the treatment of RDEB.
ABCB5+ mesenchymal stem cells (ABCB5+ MSCs): RHEACELL
ABCB5+ mesenchymal stem cells (ABCB5+ MSCs) may represent a new, promising therapeutic approach to epidermolysis bullosa. ABCB5+ MSCs are infused into the bloodstreams and can migrate to the wounds, decreasing inflammation and promoting wound healing. ABCB5+ MSCs target all wounds (internal and external) and release and deposit collagen VII.
In October 2021, the biopharmaceutical company RHEACELL was honored by the organization ‘Dystrophic Epidermolysis Bullosa Research Association of America’ (DEBRA) with the ‘Partners in Progress Award’ to recognize RHEACELL’s research commitment and medical advancements in the development of a therapy to treat severe forms of epidermolysis bullosa.
|
Comparison of Emerging Drugs Under Development | |||||||
|
Drug Name |
Company |
Indication |
Highest Phase |
RoA |
MoA |
Molecule Type |
Anticipated Launch in the US |
|
D-Fi (dabocemagene autoficel) |
Castle Creek Biosciences |
RDEB |
III |
Intradermal injection |
Provides functional type VII collagen (COL7) locally to affected areas |
Autologous cell-based gene therapy |
2028 |
|
ABCB5+ mesenchymal stem cells (ABCB5+ MSCs) |
RHEACELL |
RDEB, JEB |
III |
IV infusion |
Form structural proteins collagen VII, laminin-322, and keratin14, and thus support wound healing |
Stem cell-based therapy |
Information is available in the full report |
|
RV-LAMB3-transduced epidermal stem cells |
Holostem Terapie Avanzate |
Generalized intermediate LAMB3-dependent JEB |
II/III |
Skin grafting (surgical application) |
Restoration of the epidermis |
Combined cell and gene therapy |
Information is available in the full report |
|
Note: Launch insights are provisional and may change with future report updates or the occurrence of major key catalysts. | |||||||
Note: A detailed emerging therapies assessment will be provided in the final report
Epidermolysis Bullosa Key Players, Market Leaders, and Emerging Companies
- Krystal Biotech
- Chiesi Farmaceutici
- Japan Tissue Engineering
- Abeona Therapeutics
- Castle Creek Biosciences
- RHEACELL
- Ishin Pharma
- Holostem Terapie Avanzate, and others
Epidermolysis Bullosa Market Recent Developments and Breakthroughs
- In September 2025, Krystal Biotech announced that the US FDA approved a label update for VYJUVEK that expands the VYJUVEK eligible patient population to include DEB patients from birth and provides patients full flexibility with respect to VYJUVEK application and managing wound dressings.
- In July 2025, Krystal Biotech announced that the Japanese Ministry of Health, Labour and Welfare (MHLW) granted marketing authorization to VYJUVEK for the treatment of wounds in patients with DEB, starting from birth. VYJUVEK is the first genetic medicine approved in Japan for the treatment of DEB. The Japanese approval allows for dosing at home or in a healthcare setting, with the option for administration by patients or their family members.
- In April 2025, Abeona Therapeutics announced the US FDA had approved ZEVASKYN gene-modified cellular sheets, also known as pz-cel, as the first and only autologous cell-based gene therapy for the treatment of wounds in adult and pediatric patients with RDEB.
Epidermolysis Bullosa Market Outlook
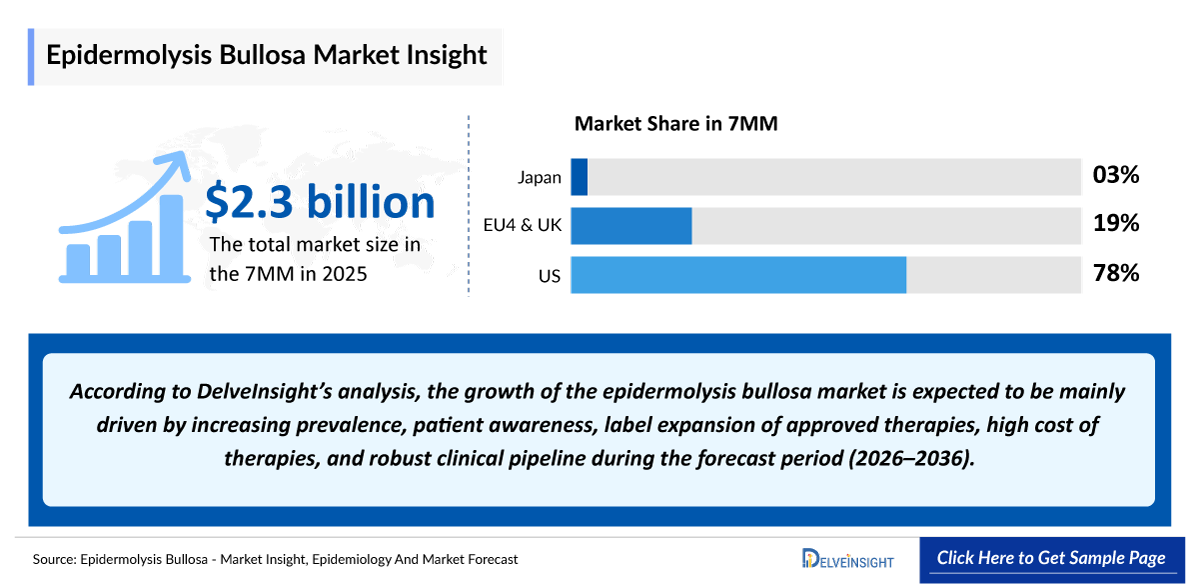
Significant advances in understanding the pathophysiology of epidermolysis bullosa have driven the development of repurposed drugs and novel targeted therapies, particularly disease-modifying approaches aimed at restoring defective proteins. However, no curative treatments exist for most epidermolysis bullosa subtypes, and current management remains largely supportive, focusing on wound care, pain control, and prevention of complications. Wound care is the cornerstone, but it is time-intensive, costly, and places a substantial burden on patients and caregivers, especially in severe forms such as recessive dystrophic epidermolysis bullosa (RDEB).
Supportive treatment commonly includes antibiotics, pain medications, and anti-pruritic therapies. Pruritus is a major concern and is managed with topical agents (ointments, corticosteroids, antihistamines) and systemic medications such as hydroxyzine and gabapentin. However, long-term use of some therapies, particularly corticosteroids, may lead to adverse effects.
Emerging therapeutic strategies include gene therapy, cell-based therapies, and protein replacement approaches. While early studies (e.g., fibroblast injections and bone marrow transplantation) have shown partial benefits, challenges related to safety, tolerability, and durability remain. Japan has led in cell therapy approvals with JACE, while Europe approved FILSUVEZ for wound treatment. A breakthrough occurred in 2023 with the US approval of VYJUVEK, the first topical gene therapy for DEB, which promotes wound healing and reduces blister formation.
Despite these advances, treatment availability remains limited across regions, highlighting significant unmet needs. Current research focuses on two main strategies: correcting the underlying genetic defect (e.g., COL7A1 gene therapies) and reducing inflammation to alleviate symptoms. A robust pipeline, including candidates such as D-Fi and ABCB5+ mesenchymal stem cells (ABCB5+ MSCs), as well as stem cell and combination gene-cell therapies, continues to evolve, with the potential to transform the treatment landscape. Continued innovation is critical given the high disease burden and impact on patient quality of life. Overall, the epidermolysis bullosa therapeutics market is expected to increase in the forecast period (2026–2036).
Key findings from Epidermolysis Bullosa Market Forecast Report
- The market size of epidermolysis bullosa in the 7MM is USD 2,300 million in 2025, which is expected to rise during the forecast period (2026–2036).
- According to the estimates, the largest market size of epidermolysis bullosa is expected to be from the United States, i.e., USD 1,800 million in 2025.
Further details will be provided in the report….
Drug Class/Insights into Leading Emerging and Marketed Therapies in Epidermolysis Bullosa (2026–2036 Forecast)
The epidermolysis bullosa therapeutic landscape remains largely supportive; however, recent advances in gene and cell-based therapies have initiated a shift toward disease-modifying, mechanism-driven approaches aimed at correcting underlying genetic defects.
Gene therapies: Targeted gene therapies such as beremagene geperpavec (VYJUVEK) deliver functional copies of defective genes (e.g., COL7A1), promoting wound healing and reducing blister formation. These therapies represent a major advancement toward durable, disease-modifying treatment in DEB.
Cell and regenerative therapies: Approaches including autologous cultured epidermal grafts (e.g., JACE) and stem cell–based therapies aim to restore skin integrity and enhance healing. While promising, challenges related to scalability, tolerability, and long-term efficacy remain.
Further details will be provided in the report….
Epidermolysis Bullosa Drug Uptake
This section focuses on the uptake rate of potential drugs expected to be launched in the market during the forecast period (2026–2036). The analysis covers the epidermolysis bullosa market's uptake by drugs, patient uptake by therapy, and sales of each drug.
Drug uptake in epidermolysis bullosa is currently highest for ZEVASKYN (prademagene zamikeracel), driven by its disease-modifying potential and ability to address the underlying genetic defect rather than providing only symptomatic relief. Its targeted gene therapy approach and promising clinical outcomes have supported strong physician interest and early adoption.
Further detailed analysis of emerging therapies' drug uptake in the report…
Market Access and Reimbursement of Epidermolysis Bullosa
-
US
Reimbursement is a crucial factor that affects the drug’s access to the market. Often, the decision to reimburse comes down to the price of the drug relative to the benefit it produces in treated patients. To reduce the healthcare burden of these high-cost therapies, many payment models are being considered by payers and other industry insiders.
The price of gene and cell therapies depends mainly on their perceived value as defined by multiple factors, including their level of innovation, durable clinical benefit, treatable patient population size, and impact on health systems. The Institute for Clinical and Economic Review estimates that the cumulative budget impact for gene and cell therapies alone could rise to USD 3 trillion in the US when only about 10% of eligible patients are treated with these therapies.
To reduce the healthcare burden of these high-cost therapies, payers and other industry insiders are considering many payment models. The payment models are based on clinical outcomes, annuity payments, and expanded risk pools.
Further details are provided in the final report….
Epidermolysis Bullosa Therapies Price Scenario & Trends
Pricing and analogue assessment of epidermolysis bullosa therapies highlights evolving price dynamics structures. This section summarizes the cost of approved treatments, the closest and most appropriate analogue selection for emerging therapies, and understanding of how pricing influences market access, adherence, and long-term uptake.
Pricing of Epidermolysis Bullosa Approved Drugs
The recommended dose of FILSUVEZ is 10% birch triterpenes w/w supplied in 25 mL sterile tubes, with the total therapy cost being approximately USD 190,000.
Further details are provided in the final report….
Industry Experts and Physician Views for Epidermolysis Bullosa
To keep up with epidermolysis bullosa market trends, we take Key Opinion Leaders (KOLs) and Subject Matter Experts (SMEs) opinions working in the domain through primary research to fill the data gaps and validate our secondary research. Industry experts were contacted for insights on the epidermolysis bullosa emerging therapies, evolving treatment landscape, patient adherence to conventional therapies, therapy switching trends, drug adoption and uptake, accessibility challenges, and epidemiology and real-world prescription patterns in epidermolysis bullosa, including MD, PhD, Instructor, Postdoctoral Researcher, Professor, Researcher, and others.
DelveInsight’s analysts connected with 10+ KOLs to gather insights; however, interviews were conducted with 6+ KOLs in the 7MM. Centers such as the University of North Carolina at Chapel Hill, the Berlin Institute of Health at Charité, and the University of Nottingham, etc. were contacted. Their opinion helps understand and validate current and emerging epidermolysis bullosa therapies, highlight unmet medical needs, provide epidemiological context, and support strategic decisions for market access, therapy adoption, and pipeline prioritization in epidermolysis bullosa.
What are the KOL Views on Epidermolysis Bullosa Market? | |
|
Region |
Key Opinion Leaders (KOLs) and Subject Matter Experts (SMEs) |
|
United States |
“I think the approach of B-VEC is exciting because it goes on to wounds and has an enduring effect for months, which is very helpful. As new wounds develop, you can just get on and treat them. And you can treat different anatomical sites, which again is helpful for people with lots of wounds in the axilla or around the neck and the flexors. Really hard areas to dress and areas that are not going to be suitable for ex vivo gene therapy grafting.” |
|
Germany |
“Diagnostic testing for epidermolysis bullosa is more common for patients with severe phenotypes; understanding the course of major clinical events may enable improved counseling on prognosis and management of epidermolysis bullosa.” |
Epidermolysis Bullosa Qualitative Analysis: SWOT and Conjoint Analysis
We perform qualitative and market Intelligence analysis using various approaches, such as SWOT analysis and conjoint analysis.
In the SWOT analysis of Epidermolysis Bullosa, strengths, weaknesses, opportunities, and threats in terms of disease diagnosis, patient awareness, patient burden, competitive landscape, cost-effectiveness, and geographical accessibility of therapies are provided.
Conjoint analysis analyzes emerging therapies based on relevant attributes such as safety, efficacy, frequency of administration, route of administration, and order of entry. Scoring is given based on these parameters to analyze the effectiveness of therapy.
The team of analysts analyzes promising emerging therapies based on relevant attributes such as safety, efficacy, frequency of administration, route of administration, and order of entry. In efficacy, the trial’s primary and secondary outcome measures are evaluated, whereas the therapies’ safety is evaluated, wherein the acceptability, tolerability, and adverse events are mainly observed. In addition, the scoring is also based on the route of administration, order of entry, probability of success, and the addressable patient pool for each therapy. According to these parameters, the final weightage score and the ranking of the emerging therapies are decided.
Scope of the Epidermolysis Bullosa Market Report
- The Epidermolysis Bullosa Market report covers a segment of key events, an executive summary, a descriptive overview of epidermolysis bullosa, explaining its causes, signs and symptoms, pathogenesis, and currently available treatments.
- Comprehensive insight has been provided into the epidemiology segments and forecasts, the future growth potential of the diagnosis rate, and disease progression along treatment guidelines.
- Additionally, an all-inclusive account of both the current and emerging treatments, along with the elaborative profiles of late-stage and prominent therapies, will have an impact on the current treatment landscape.
- A detailed review of the epidermolysis bullosa market, historical and forecasted market size, market share by therapies, detailed assumptions, and rationale behind our approach is included in the report, covering the 7MM drug outreach.
- The Epidermolysis Bullosa Market report provides an edge while developing business strategies by understanding trends through SWOT analysis and expert insights/KOL views, patient journey, and treatment preferences that help in shaping and driving the 7MM epidermolysis bullosa market.
Epidermolysis Bullosa Market Report Insights
- Epidermolysis Bullosa Patient Population Forecast
- Epidermolysis Bullosa Therapeutics Market Size
- Epidermolysis Bullosa Pipeline Analysis
- Epidermolysis Bullosa Market Size and Trends
- Epidermolysis Bullosa Market Opportunity (Current and forecasted)
Epidermolysis Bullosa Market Report Key Strengths
- Epidemiology‑based (Epi‑based) Bottom‑up Forecasting
- Artificial Intelligence (AI)-enabled Market Research Report
- 11-year forecast
- Epidermolysis Bullosa Market Outlook (North America, Europe, Asia-Pacific)
- Patient Burden Trends (by geography)
- Epidermolysis Bullosa Treatment Addressable Market (TAM)
- Epidermolysis Bullosa Competitive Landscape
- Epidermolysis Bullosa Major Companies Insights
- Epidermolysis Bullosa Price Trends and Analogue Assessment
- Epidermolysis Bullosa Therapies Drug Adoption/Uptake
- Epidermolysis Bullosa Therapies Peak Patient Share Analysis
Epidermolysis Bullosa Market Report Assessment
- Epidermolysis Bullosa Current Treatment Practices
- Epidermolysis Bullosa Unmet Needs
- Epidermolysis Bullosa Clinical Development Analysis
- Epidermolysis Bullosa Emerging Drugs Product Profiles
- Epidermolysis Bullosa Market Attractiveness
- Epidermolysis Bullosa Qualitative Analysis (SWOT and Conjoint Analysis)
Frequently Asked Questions from Epidermolysis Bullosa Market Report
Epidermolysis Bullosa Market Insights
- What was the epidermolysis bullosa market size, the market size by therapies, market share (%) distribution in 2025, and what would it look like by 2036? What are the contributing factors for this growth?
- What are the anticipated pricing variations among different geographies for the emerging therapies in the future?
- What can be the future treatment paradigm of epidermolysis bullosa?
- What are the disease risks, burdens, and unmet needs of epidermolysis bullosa? What will be the growth opportunities across the 7MM concerning the patient population with epidermolysis bullosa?
- Who is the major future competitor in the market, and how will the competitors affect their market share?
- What are the current options for the treatment of epidermolysis bullosa? What are the current guidelines for treating epidermolysis bullosa in the US, Europe, and Japan?
Reasons to Buy the Epidermolysis Bullosa Market Report
- The Epidermolysis Bullosa Market report will help in developing business strategies by understanding the latest trends and changing treatment dynamics driving the epidermolysis bullosa market.
- Bottom‑up forecasting builds from the affected population to product forecasts, delivering a robust, data‑driven approach ideal for new therapies and novel classes.
- Insights on patient burden/disease incidence, evolution in diagnosis, and factors contributing to the change in the epidemiology of the disease during the forecast years.
- Understand the existing Epidermolysis Bullosa Market opportunities in varying geographies and the growth potential over the coming years.
- Identifying strong upcoming Epidermolysis Bullosa companies in the Epidermolysis Bullosa market will help devise strategies to help get ahead of competitors.
- Detailed analysis and ranking of class-wise potential current and emerging therapies under the conjoint analysis section to provide visibility around leading classes.
- To understand KOLs’ perspectives on the accessibility, acceptability, and compliance-related challenges of existing treatment to overcome barriers in the future.
- Detailed insights into the unmet needs of the existing Epidermolysis Bullosa Market so that the upcoming Epidermolysis Bullosa companies can strengthen their development and launch strategy.
- This Artificial Intelligence (AI)‑enabled report summarize and simplify complex datasets within the report into clear, actionable insights for stakeholders, investors, and healthcare providers, enabling faster, data‑driven decisions.
Stay Updated with us for Recent Articles
- Gene Therapies for Epidermolysis Bullosa Treatment: The Next Frontier in Dermatology
- Will The Burgeoning Gene Therapies Make a Difference in Dystrophic Epidermolysis Bullosa Patients’ Lives?
- Which Pipeline Therapy Has The Potential To Revolutionize The Dystrophic Epidermolysis Bullosa Treatment Market?
- Pharma Players Line Up To Launch Novel Therapies Fueling Epidermolysis Bullosa Market Size
- Epidermolysis Bullosa Market Infographics