Neurovascular Devices Market Summary
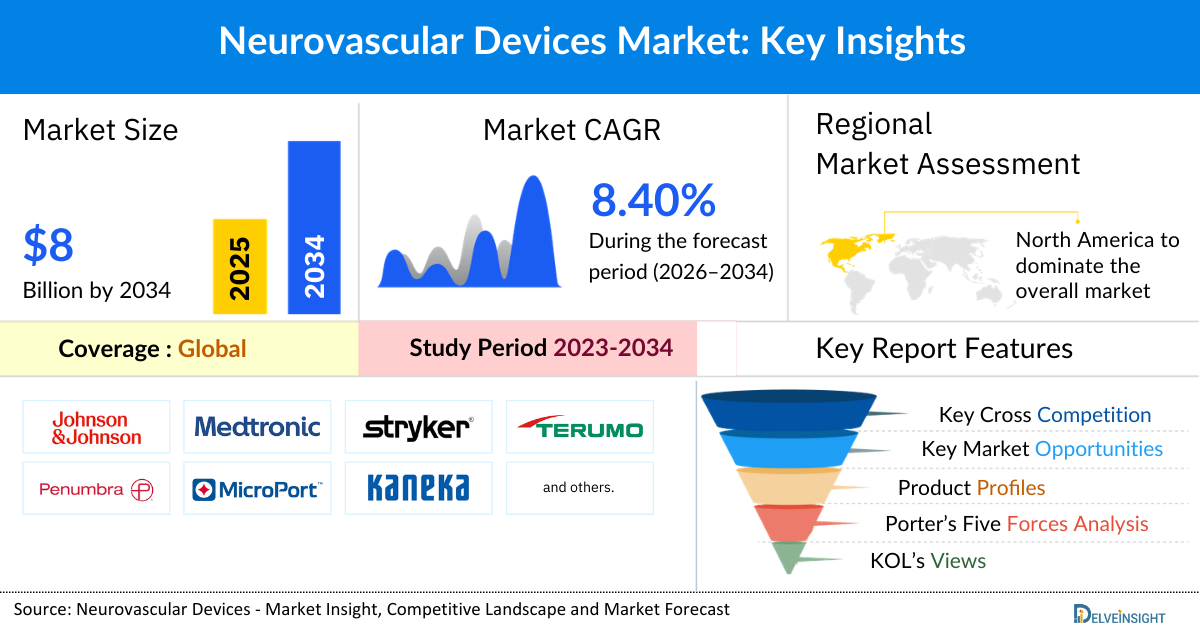
- The Global Neurovascular Devices Market Size is expected to increase from USD 3,904.74 million in 2025 to USD 8,044.66 million by 2034, reflecting strong and sustained growth.
- The Global Neurovascular Devices Market Size is growing at a CAGR of 8.40% during the forecast period from 2026 to 2034.
Global Neurovascular Devices Market Trends & Insights
- The Neurovascular Devices Market is being primarily driven by the rising prevalence of neurological disorders such as stroke, cerebral aneurysms, arteriovenous malformations, and other cerebrovascular diseases. The increasing incidence of ischemic and hemorrhagic strokes, particularly among the aging population and individuals with risk factors like Hypertension and Diabetes, is significantly boosting the demand for advanced neurovascular intervention devices. Additionally, growing awareness about early diagnosis, advancements in minimally invasive procedures, and improved healthcare infrastructure are further accelerating the adoption of neurovascular devices across hospitals and specialty neurology centers.
- The leading Neurovascular Devices Companies such as Johnson & Johnson Services Inc., Medtronic, Stryker, Terumo Corporation, Penumbra, Inc., Microport Scientific Corporation, Kaneka Corporation, Integer Holdings Corporation, phenox GmbH, Perflow Medical Ltd., Balt, Rapid Medical, ASAHI INTECC CO., LTD., Acandis GmbH, Cerus Endovascular Inc., and others.
- North America is expected to dominate the overall Neurovascular Devices Market due to the high prevalence of stroke and other neurovascular disorders, a well-established healthcare infrastructure, strong adoption of advanced and minimally invasive technologies, and supportive reimbursement policies. Additionally, the presence of leading medical device companies, continuous R&D investments, and growing awareness programs for stroke prevention and treatment further strengthen the region’s market leadership.
- In the product segment of the Neurovascular Devices Market, within cerebral balloon angioplasty and stenting systems category, the carotid artery stents category is estimated to account for the largest market share in 2025.
Request for Unlocking the Sample Report of the @ Neurovascular Devices Market Insights
Neurovascular Devices Market Size and Forecasts
|
Report Metrics |
Details |
|
2025 Market Size | |
|
2034 Projected Market Size | |
|
Growth Rate (2026-2034) | |
|
Largest Market |
North America |
|
Fastest Growing Market |
Asia-Pacific |
|
Market Structure |
Moderately to Highly Concentrated |
Factors Contributing to the Growth of the Neurovascular Devices Market
-
Increasing prevalence of neurological disorders leading to a surge in neurovascular devices
The increasing prevalence of neurological disorders such as stroke, brain aneurysms, and arteriovenous malformations is a key factor boosting the overall market for neurovascular devices. Rising cases of these conditions, driven by aging populations, sedentary lifestyles, and associated risk factors like hypertension and diabetes, are creating higher demand for advanced diagnostic and treatment solutions. This is accelerating the adoption of neurovascular devices for minimally invasive procedures, thereby fueling market growth.
-
Increasing technological advancements in devices are escalating the Neurovascular Devices Market
Increasing technological advancements in neurovascular devices, such as next-generation stent retrievers, flow diverters, embolic coils, and AI-enabled imaging systems, are significantly escalating the market. These innovations enhance precision, safety, and success rates of minimally invasive procedures, reducing recovery time and complications. As a result, hospitals and clinicians are increasingly adopting these advanced solutions, driving strong neurovascular devices market growth.
-
Increase in product development activities among the key Neurovascular Devices Companies
The increase in product development activities among key market players is significantly boosting the neurovascular devices market by introducing more advanced, safer, and minimally invasive treatment options. Companies are continuously innovating and launching next-generation thrombectomy devices, flow diverters, embolization coils, and stent retrievers that improve procedural success rates and reduce complications. These advancements enhance treatment precision for conditions such as stroke and cerebral aneurysms, encouraging greater adoption among neurosurgeons and interventional radiologists. As a result, continuous R&D investments and new product approvals are accelerating market growth and strengthening competitive dynamics in the neurovascular devices sector.
Neurovascular Devices Market Report Segmentation
This neurovascular devices market report offers a comprehensive overview of the global neurovascular devices market, highlighting key trends, growth drivers, challenges, and opportunities. It covers detailed market segmentation by Product Type (Cerebral Embolization and Aneurysm Coiling Devices [Embolic Coils, Flow Diversion Devices, and Liquid Embolic Agents], Cerebral Balloon Angioplasty and Stenting Systems [Carotid Artery Stents and Embolic Protection Systems], Neurothrombectomy Devices [Clot Retrievers, Suction Devices, and Vascular Snare], and Support Devices [Micro Catheters and Micro Guidewires]), Application (Stroke, Cerebral Aneurysm, Carotid Artery Stenosis, and Others), End-User (Hospitals, Ambulatory Surgical Centers, and Others), and geography.
The Neurovascular Devices Market Report provides valuable insights into the competitive landscape, regulatory environment, and market dynamics across major markets, including North America, Europe, and Asia-Pacific. Featuring in-depth profiles of leading industry players and recent product innovations, this report equips businesses with essential data to identify market potential, develop strategic plans, and capitalize on emerging opportunities in the rapidly growing Neurovascular Devices Market.
Neurovascular devices are specialized medical devices used to diagnose and treat disorders affecting the blood vessels of the brain and spinal cord. These devices are primarily used in minimally invasive procedures to manage conditions such as stroke, cerebral aneurysms, arteriovenous malformations (AVMs), and other cerebrovascular diseases. Common examples include stent retrievers, flow diverters, embolization coils, thrombectomy devices, and microcatheters, which help restore blood flow, prevent vessel rupture, or block abnormal blood vessels in the brain.
The neurovascular devices market is being primarily driven by the rising prevalence of neurological disorders such as stroke, cerebral aneurysms, arteriovenous malformations, and other cerebrovascular diseases. The increasing incidence of ischemic and hemorrhagic strokes, particularly among the aging population and individuals with risk factors like hypertension and diabetes, is significantly boosting the demand for advanced neurovascular intervention devices. Additionally, growing awareness about early diagnosis, advancements in minimally invasive procedures, and improved healthcare infrastructure are further accelerating the adoption of neurovascular devices across hospitals and specialty neurology centers.
Get More Insights into the Report @ Neurovascular Devices Market Trends
What are the latest Neurovascular Devices Market Dynamics and Trends?
The Neurovascular Devices Market is gaining pace at present, owing to the increasing prevalence of Neurovascular diseases and their associated comorbidities such as ischemic strokes, hemorrhagic strokes, aging populations, hypertension, and arrhythmias.
According to recent analysis by DelveInsight, the 7MM (United States, Japan, Germany, France, Italy, Spain, and the United Kingdom) accounted for over 1.5 million cases of acute ischemic stroke. Globally, it is estimated that more than 3.5 million new cases of intracerebral hemorrhagic stroke occur each year, representing over 28.5% of all stroke incidents. In addition, approximately 1.3 million new subarachnoid hemorrhagic strokes are reported annually. Notably, a significant proportion of these cases, over 23.1% of intracerebral hemorrhagic strokes and 28% of subarachnoid hemorrhagic strokes, affect individuals aged 15-49, highlighting the impact on a younger population and the critical need for timely interventions.
These statistics directly translate to increased adoption of Neurovascular devices such as stent retrievers, embolic coils, flow diverters, and carotid stents, which are essential for minimally invasive management of ischemic and hemorrhagic strokes. As stroke awareness among clinicians and patients rises, the push for early diagnosis and intervention grows stronger, further driving demand for advanced, precise, and safe devices. Moreover, technological advancements in imaging, device design, and materials enhance procedural success and reduce complications, making these interventions more attractive to hospitals and healthcare providers. This combination of high patient burden, clinical need for effective treatment, and continuous innovation in device technology is creating a strong growth trajectory for the Neurovascular devices market.
The players are focusing their efforts on developing advanced Neurovascular devices. For instance, in June 2024, Penumbra announced the CE Mark and European launch of BMX™81 and BMX™96. These offerings, designed for the Neurovascular management of both ischemic and hemorrhagic strokes, represent Penumbra’s most advanced neuro access technologies to date, reflecting the emphasis on innovation and clinical efficacy in the neurovascular devices market.
Thus, the factors mentioned above are expected to boost the overall market of neurovascular devices during the forecast period from 2026 to 2034.
However, the growing number of product recalls, particularly those related to safety concerns, device malfunctions, or quality control issues, is expected to negatively impact the future growth of the neurovascular devices market. Recalls not only result in financial losses for manufacturers due to product withdrawals, legal liabilities, and corrective actions, but they also damage brand reputation and reduce physician and patient confidence in advanced neurovascular technologies. In addition, the stringent regulatory approval processes imposed by authorities such as the FDA and EMA further act as a limiting factor for market expansion.
Neurovascular devices, being high-risk and life-saving products used in conditions such as stroke and aneurysm, must undergo extensive clinical trials, rigorous safety evaluations, and detailed documentation before receiving approval. These requirements significantly increase development timelines and costs, delaying product launches and limiting the entry of new players into the market. As a result, while innovation in neurovascular devices continues to advance, regulatory complexities and recall-related challenges may restrain the overall growth potential of the market in the coming years.
Neurovascular Devices Market Segment Analysis
Neurovascular Devices Market by Product Type (Cerebral Embolization and Aneurysm Coiling Devices [Embolic Coils, Flow Diversion Devices, and Liquid Embolic Agents], Cerebral Balloon Angioplasty and Stenting Systems [Carotid Artery Stents and Embolic Protection Systems], Neurothrombectomy Devices [Clot Retrievers, Suction Devices, and Vascular Snare], and Support Devices [Micro Catheters and Micro Guidewires]), Application (Stroke, Cerebral Aneurysm, Carotid Artery Stenosis, and Others), End-User (Hospitals, Ambulatory Surgical Centers, and Others), and Geography (North America, Europe, Asia-Pacific, and Rest of the World).
By Product Type: Carotid Artery Stents Dominate the Neurovascular Devices Market
In the product type segment of the neurovascular devices market, within the cerebral balloon angioplasty and stenting systems category, the carotid artery stents segment is projected to dominate in 2025, capturing approximately 65% of the market share. Carotid artery stents are key drivers in the growth of the neurovascular devices market by offering less invasive, highly effective treatments for carotid artery disease, a leading cause of ischemic stroke. Compared to traditional surgery, stenting provides shorter recovery times, reduced procedural risk, and wider applicability for high-risk patients. Additionally, the technological innovations such as dual-layer micromesh, MicroNet mesh coatings, and integrated embolic protection enhance both safety and long-term outcomes, further bolstering clinician confidence and adoption.
On the regulatory front, several impactful approvals have signaled robust advancements that further boost the market of the segment. For instance, in April 2025, Terumo Neuro’s Dual-Layer Micromesh Carotid Stent System received FDA Premarket Approval (PMA), the first of its kind in the U.S. This device targets carotid stenosis, including both de novo and post-endarterectomy restenotic lesions with vessel diameters from 3.5 mm to 9 mm, offering a significant new option for patients at higher risk following endarterectomy.
Collectively, these factors position carotid artery stents as a key growth driver within the neurovascular devices market, expanding their role in stroke prevention and reshaping treatment strategies worldwide.
By Application: Stroke Category Dominates the Neurovascular Devices Market
In the neurovascular devices market, strokes are projected to account for the largest share, representing 55% in 2025, driven by the high prevalence of ischemic strokes caused by blocked blood vessels in the brain. According to DelveInsight, the U.S. recorded nearly 754,000 diagnosed cases of acute ischemic stroke in 2023, the highest among the 7MM, a number that is projected to rise steadily by 2034. Thus, the timely and precise intervention is essential to restore blood flow and reduce brain damage, making advanced neurovascular devices critical in patient care.
Additionally, the minimally invasive thrombectomy procedures, performed using stent retrievers, aspiration catheters, and embolic protection systems, have become the standard of care for eligible patients. Continuous innovations in device design, imaging technologies, and AI-assisted procedural guidance have improved treatment efficacy, safety, and overall success rates, encouraging wider clinical adoption. A notable example is Johnson & Johnson’s April 2024 launch of the CEREGLIDE™ 71 Aspiration Catheter. Equipped with TruCourse™ technology, it offers enhanced flexibility and trackability, allowing clinicians to effectively access challenging anatomical regions and remove clots efficiently during acute ischemic stroke interventions.
Thus, the factors mentioned above are expected to boost the category, thereby boosting the overall market of neurovascular devices across the globe.
By End-Users: Hospitals Category Dominates the Market
Hospitals are boosting the overall Neurovascular devices market by increasingly adopting advanced, minimally invasive treatment solutions to improve patient outcomes and reduce procedural risks. Investments in state-of-the-art imaging systems, catheterization labs, and specialized stroke care units are driving higher demand for devices such as stent retrievers, embolic coils, and flow diverters. Additionally, hospitals’ focus on early diagnosis, rapid intervention, and comprehensive Neurovascular care is encouraging clinicians to use these devices more frequently, thereby accelerating neurovascular devices market growth.
Neurovascular Devices Market Regional Analysis
North America Neurovascular Devices Market Trends
North America is expected to account for the highest proportion of 38% of the neurovascular devices market in 2025, out of all regions, due to the high prevalence of stroke and other neurovascular disorders, a well-established healthcare infrastructure, strong adoption of advanced and minimally invasive technologies, and supportive reimbursement policies. Additionally, the presence of leading medical device companies, continuous R&D investments, and growing awareness programs for stroke prevention and treatment further strengthen the region’s market leadership.
According to DelveInsight analysis (2023), approximately 376,000 males and 378,000 females in the United States were affected by acute ischemic stroke in 2023. This significant patient population is driving strong demand for advanced neurovascular devices, including thrombectomy systems, embolic protection devices, clot retrieval catheters, and stent retrievers, as hospitals and healthcare providers seek to improve patient outcomes and reduce mortality rates.
Furthermore, the growing adoption of advanced and minimally invasive technologies is further accelerating the Neurovascular devices market. Procedures such as thrombectomy and endovascular coiling have become standard practice due to their ability to deliver favorable outcomes, lower complication rates, and shorter recovery times. In addition, increasing product development activities by key market players are expected to further boost neurovascular devices market growth. For example, in October 2024, Contego Medical received FDA premarket approval (PMA) for the Neuroguard IEP® System, featuring the company’s clinically proven Integrated Embolic Protection™ (IEP) technology, highlighting the ongoing innovation and expansion in the neuroprotection devices segment.
Therefore, the factors mentioned above are expected to drive the growth of the neurovascular devices market in North America.
Europe Neurovascular Devices Market Trends
Europe is playing a significant role in driving the growth of Neurovascular devices fueled The region has a high prevalence of Neurovascular disorders, including acute ischemic stroke, aneurysms, and arteriovenous malformations, which creates a substantial demand for advanced treatment options. European healthcare systems are increasingly focusing on early diagnosis and minimally invasive interventions, which has accelerated the adoption of Neurovascular devices such as thrombectomy systems, embolic protection devices, stent retrievers, and clot retrieval catheters. Moreover, the presence of key market players and continuous product innovation in the region, such as the development of next-generation thrombectomy and embolic protection devices, further strengthens market expansion. For instance, in June 2025, InspireMD announced that its CGuard® Prime Embolic Prevention System (EPS) received CE Mark approval under the European Medical Device Regulation (MDR) for use in stroke prevention.
Thus, the factors mentioned above are expected to boost the overall market of neurovascular devices in the Europe.
Asia-Pacific Neurovascular Devices Market Trends
Asia-Pacific is emerging as a major growth driver for the Neurovascular devices market due to its rapidly rising patient pool for Neurovascular disorders, including acute ischemic stroke and aneurysms, driven by aging populations. DelveInsight estimates indicated that in Japan, males were affected more with acute ischemic stroke, accounting for nearly 100,000 cases compared to females (around 90,000 cases) in 2023. Additionally, the growing awareness about early diagnosis and the benefits of minimally invasive procedures is fueling the adoption of advanced Neurovascular devices, including thrombectomy systems, stent retrievers, and embolic protection devices across the region.
Thus, the factors mentioned above are expected to boost the overall market of neurovascular devices in the Asia-Pacific region.
Who are the major players in the Neurovascular Devices Market?
The following are the leading companies in the Neurovascular Devices Market. These Neurovascular Devices Companies collectively hold the largest market share and dictate industry trends.
- Johnson & Johnson Services Inc.
- Medtronic
- Stryker
- Terumo Corporation
- Penumbra, Inc.
- Microport Scientific Corporation
- Kaneka Corporation
- Integer Holdings Corporation
- phenox GmbH
- Perflow Medical Ltd.
- Balt
- Rapid Medical
- ASAHI INTECC CO., LTD.
- Acandis GmbH
- Cerus Endovascular Inc., and others
How is the competitive landscape shaping the Neurovascular Devices market?
The competitive landscape of the neurovascular devices market is moderately consolidated, with a handful of global leaders holding substantial market share while regional players and emerging startups are steadily gaining traction. Major Neurovascular devices companies such as Medtronic, Stryker Corporation, Johnson & Johnson, Penumbra Inc., and Terumo Corporation dominate the market with comprehensive portfolios of neurovascular stents, coils, thrombectomy devices, and embolization systems, supported by advanced technologies and extensive global distribution networks. These companies are strengthening their positions through continuous innovation in minimally invasive procedures, integration of imaging and navigation technologies, and strategic acquisitions, exemplified by Stryker’s acquisition of Inari Medical to expand its Neurovascular offerings.
At the same time, emerging players, particularly in Asia-Pacific regions like China, Japan, and India, are intensifying competition by providing cost-effective devices and localized solutions, aiming to address the growing prevalence of strokes and other cerebrovascular disorders. The increasing focus on portable, efficient, and user-friendly neurovascular interventions, coupled with rising adoption in hospitals and specialized clinics, is opening avenues for new entrants. Overall, while the market remains concentrated among established leaders, the rise of regional innovators and strategic collaborations is gradually reshaping the competitive environment, making it more dynamic and innovation-driven.
Recent Developmental Activities in the Neurovascular Devices Market
- In May 2025, Terumo Interventional Systems announced the commercial launch of its FDA-approved ROADSAVER™ Carotid Stent System. Designed for use with the Nanoparasol® Embolic Protection System, the ROADSAVER Stent System is intended to treat carotid artery stenosis in patients at elevated risk of complications from carotid endarterectomy.
- In March 2025, Boston Scientific launched an updated FilterWire EZ embolic protection system in select European markets. The system features a wire with a 110-micron-pore filter at its tip, designed to capture embolic material while maintaining blood flow and reliably conforming to the artery walls.
- In September 2024, Minima Stent System received FDA clearance for its stent system, which is indicated for use in the treatment of intracranial aneurysms, offering a minimally invasive option for patients.
- In June 2024, Penumbra announced the CE Mark and European launch of BMX™81 and BMX™96. These offerings, designed for the Neurovascular management of both ischemic and hemorrhagic strokes, represent Penumbra’s most advanced neuro access technologies to date, reflecting the emphasis on innovation and clinical efficacy in the Neurovascular devices market.
Scope of the Neurovascular Devices Market | |
|
Study Period |
2023 to 2034 |
|
Base Year |
2025 |
|
Forecast Period |
2026 to 2034 |
|
Neurovascular Devices Market CAGR (2026-2034) | |
|
Neurovascular Devices Companies |
Johnson & Johnson Services Inc., Medtronic, Stryker, Terumo Corporation, Penumbra, Inc., Microport Scientific Corporation, Kaneka Corporation, Integer Holdings Corporation, phenox GmbH, Perflow Medical Ltd., Balt, Rapid Medical, ASAHI INTECC CO., LTD., Acandis GmbH, Cerus Endovascular Inc., and others. |
|
Neurovascular Devices Market Segments |
By Product Type, by Application, by End-Users, and by Geography |
|
Neurovascular Devices Regional Scope |
North America, Europe, Asia Pacific, Middle East, Africa, and South America |
|
Neurovascular Devices Country Scope |
U.S., Canada, Mexico, Germany, United Kingdom, France, Italy, Spain, China, Japan, India, Australia, South Korea, and key Countries |
Neurovascular Devices Market Segmentation
- Neurovascular Devices by Product Type Exposure
- Cerebral Embolization and Aneurysm Coiling Devices
- Embolic Coils
- Flow Diversion Devices
- Liquid Embolic Agents
- Cerebral Balloon Angioplasty and Stenting Systems
- Carotid Artery Stents
- Embolic Protection Systems
- Neurothrombectomy Devices
- Clot Retrievers
- Suction Devices
- Vascular Snare
- Support Devices
- Micro Catheters
- Micro Guidewires
- Cerebral Embolization and Aneurysm Coiling Devices
- Neurovascular Devices Application Exposure
- Stroke
- Cerebral Aneurysm
- Carotid Artery Stenosis
- Others
- Neurovascular Devices End-Users Exposure
- Hospitals
- Ambulatory Surgical Centers
- Others
Neurovascular Devices Geography Exposure
- North America Neurovascular Devices Market
- United States Neurovascular Devices Market
- Canada Neurovascular Devices Market
- Mexico Neurovascular Devices Market
- Europe Neurovascular Devices Market
- United Kingdom Neurovascular Devices Market
- Germany Neurovascular Devices Market
- France Neurovascular Devices Market
- Italy Neurovascular Devices Market
- Spain Neurovascular Devices Market
- Rest of Europe Neurovascular Devices Market
- Asia-Pacific Neurovascular Devices Market
- China Neurovascular Devices Market
- Japan Neurovascular Devices Market
- India Neurovascular Devices Market
- Australia Neurovascular Devices Market
- South Korea Neurovascular Devices Market
- Rest of Asia-Pacific Neurovascular Devices Market
- Rest of the World Neurovascular Devices Market
- South America Neurovascular Devices Market
- Middle East Neurovascular Devices Market
- Africa Neurovascular Devices Market
Neurovascular Devices Market Recent Industry Trends and Milestones (2022-2026)
|
Category |
Key Developments |
|
Neurovascular Devices Product Launch |
BMX™81 and BMX™96 by Penumbra, CEREGLIDE™ 71 Aspiration Catheter by Johnson & Johnson, Neuroguard IEP® System by Contego Medical |
|
Neurovascular Devices Regulatory Approvals |
Penumbra - BMX™81 and BMX™96 (CE), Contego Medical - Neuroguard IEP® System (FDA) |
|
Partnerships in the Neurovascular Devices Market |
Philips with Sim&Cure, Brainomix with Medtronic Neurovascular |
|
Acquisition in the Neurovascular Devices Market |
Stryker acquired Inari Medical, and Stryker Corporation acquired Boston Scientific's Neurovascular business. |
|
Company Strategy |
Medtronic: Planned to double the number of Neurovascular patients treated annually within three years. Philips: Strengthened its strategic partnership with Sim&Cure to expand innovation in Neurovascular therapy. |
|
Emerging Technology |
Artificial Intelligence (AI) and Machine Learning (ML), Advanced Imaging Technologies, Robotics and Navigation Systems, Remote Monitoring and Telemedicine |
Impact Analysis
AI-Powered Innovations and Applications:
Artificial intelligence (AI) is rapidly transforming the Neurovascular devices market by enhancing the accuracy, efficiency, and safety of cerebrovascular diagnosis and interventions. AI-powered tools are increasingly integrated into imaging platforms, enabling real-time analysis of complex Neurovascular structures through high-resolution MRI, CT, and angiography. These technologies assist clinicians in identifying vascular anomalies, planning interventions, and predicting procedural outcomes with higher precision. Machine learning algorithms also support stroke detection by rapidly analyzing imaging data to distinguish between ischemic and hemorrhagic strokes, which accelerates decision-making in critical care settings.
Beyond diagnostics, AI is being applied in robotic-assisted Neurovascular interventions, guiding catheters, stents, and thrombectomy devices with improved precision and reduced procedural risks. Additionally, AI-driven platforms facilitate personalized treatment planning by integrating patient-specific anatomical and physiological data, optimizing device selection, and predicting clinical outcomes. The incorporation of cloud-based monitoring, predictive analytics, and decision-support systems further enhances post-procedural care and follow-up, improving overall patient outcomes. Overall, AI-powered innovations are redefining Neurovascular care, enabling minimally invasive procedures, enhancing procedural safety, and paving the way for more personalized and efficient treatment strategies.
U.S. Tariff Impact Analysis on Neurovascular Devices Market
The U.S. tariff impact on the neurovascular devices market is an important consideration for manufacturers, healthcare providers, and distributors, as tariffs on imported raw materials or finished components can increase production and procurement costs. Neurovascular devices, which include high-precision products such as thrombectomy systems, embolization coils, stents, and flow diverters, often rely on specialized metals, polymers, and electronic components sourced globally. When tariffs are imposed on these imports whether through broader trade policy shifts, anti-dumping measures, or changes in U.S. Customs duties manufacturers may experience higher input costs, which can compress profit margins or be passed downstream in the form of higher prices for healthcare providers and patients.
Additionally, tariffs can strain global supply chains, leading to delays and logistical challenges that further elevate operational costs. For U.S.-based companies that export neurovascular devices, retaliatory tariffs in foreign markets can reduce price competitiveness and market share internationally. Together, these factors can slow investment, influence strategic manufacturing decisions (such as reshoring or sourcing diversification), and potentially hinder the overall growth trajectory of the neurovascular devices market in the United States. Effective tariff impact analysis therefore remains critical for stakeholders to anticipate cost pressures, optimize supply chain resilience, and navigate evolving trade environments.
How This Analysis Helps Clients
- Cost Management: By understanding the tariff landscape, clients can anticipate cost increases and adjust pricing strategies accordingly, ensuring profitability.
- Supply Chain Optimization: Clients can identify alternative sourcing options and diversify their supply chains to reduce dependency on high-tariff regions, enhancing resilience.
- Regulatory Navigation: Expert guidance on navigating the evolving regulatory environment helps clients maintain compliance and avoid potential legal challenges.
- Strategic Planning: Insights into tariff impacts enable clients to make informed decisions about manufacturing locations, partnerships, and market entry strategies.
Startup Funding & Investment Trends:
|
Company Name |
Total Funding |
Main Products |
Stage of Development |
Core Technology |
|
Neuralink |
$650 million |
Brain-computer interface |
Series E |
Brain-computer interface for controlling digital and physical devices. |
|
MedTech Innovator 2025 Cohort |
Over $800,000 in non-dilutive funding |
Neurovascular devices |
Various (Seed to Series C) |
Diverse medical technologies, including Neurovascular innovations. |
Key Takeaways from the Neurovascular Devices Market Report Study
- Market size analysis for the current neurovascular devices market size (2025), and market forecast for 8 years (2026 to 2034)
- Top key product/technology developments, mergers, acquisitions, partnerships, and joint ventures happened over the last 3 years.
- Key companies dominating the neurovascular devices market.
- Various opportunities available for the other competitors in the neurovascular devices market space.
- What are the top-performing segments in 2025? How these segments will perform in 2034?
- Which are the top-performing regions and countries in the current neurovascular devices market scenario?
- Which are the regions and countries where companies should have concentrated on opportunities for the neurovascular devices market growth in the future?
Stay updated with us for Recent Articles @ New DelveInsight Blogs