Atopic Dermatitis Market Summary
Atopic Dermatitis Insights and Trends
- Atopic dermatitis, often referred to as eczema, is a chronic (long-lasting) disease that causes inflammation, redness, and irritation of the skin. It is a common condition that usually begins in childhood; however, anyone can get the disease at any age.
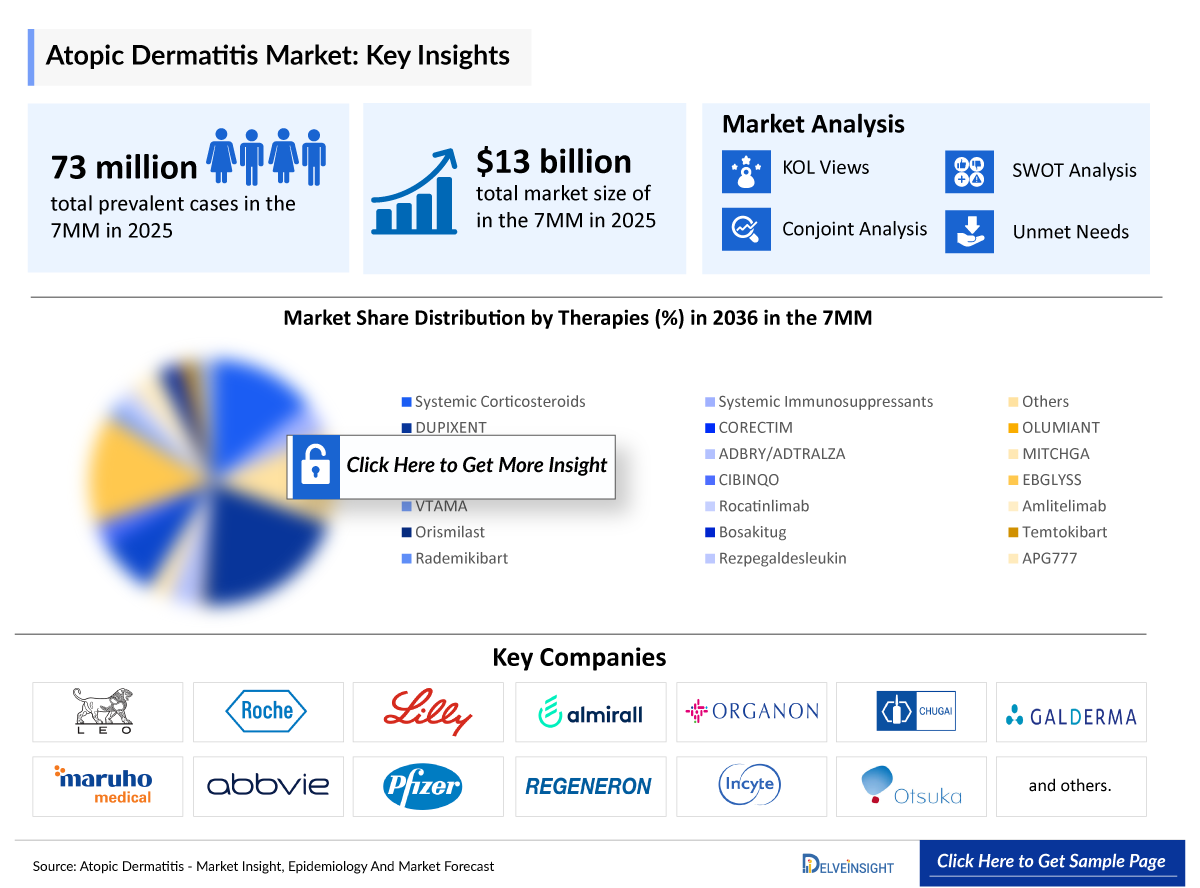
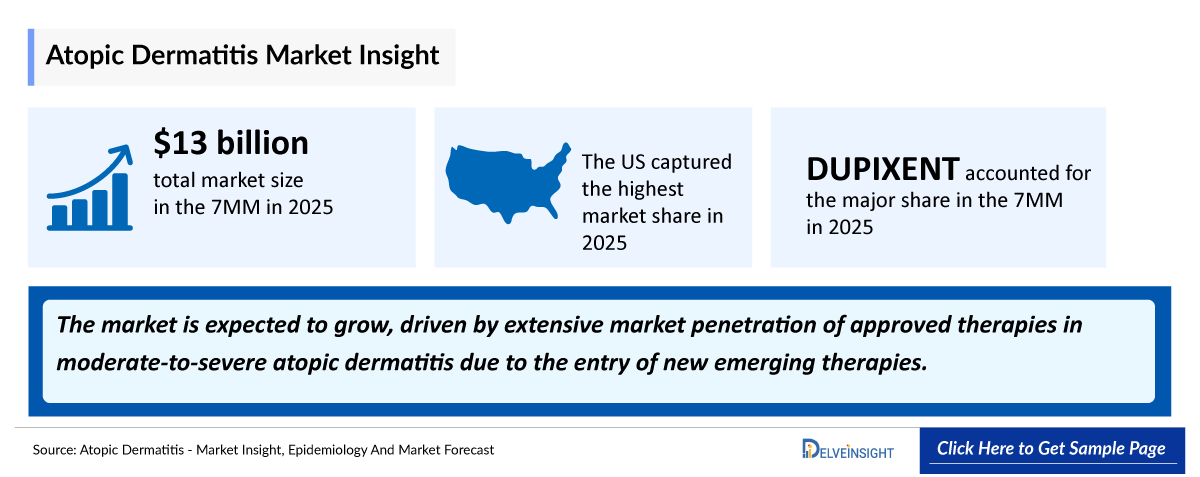
- According to DelveInsight’s analysis, atopic dermatitis market size was found to be ~USD 13,100 million in the leading markets (the United States, the EU4 (Germany, France, Italy, and Spain), the United Kingdom, and Japan) in 2025.
- The severity data from secondary findings confirms that most atopic dermatitis cases are mild to moderate in severity, regardless of age group.
- Accurate and timely diagnosis of atopic dermatitis remains critically important; however, approximately ~25% of cases are either undiagnosed or misdiagnosed, with significant delays in diagnosis being strongly associated with disease progression and burden.
- Currently, the treatment regimen of atopic dermatitis involves the use of topical treatment options such as emollients, topical corticosteroids (TCS), topical calcineurin inhibitors (TCIs), and systemic treatment such as immunosuppressant, corticosteroids, and others (phototherapy). For moderate-to-severe disease, systemic therapies are warranted, particularly in refractory cases or in cases with body surface area involvement that make topical application impractical.
- The approval of dupilumab (DUPIXENT) in 2017 marked a landmark change in the treatment landscape for atopic dermatitis patients. The drug has been progressively approved by the FDA for moderate-to-severe atopic dermatitis across various age groups: adults (2017), adolescents aged 12–17 (2019), children aged 6–11 (2020), and children aged 6 months–5 years (2022), specifically for patients inadequately controlled by or unsuitable for topical therapies.
- The approved therapies for treating adults and adolescents with moderate-to-severe atopic dermatitis include lebrikizumab (EBGLYSS), upadacitinib (RINVOQ), abrocitinib (CIBINQO), tralokinumab (ADBRY/ADTRALZA), tapinarof (VTAMA), nemolizumab (MITCHGA), difamilast (MOIZERTO), delgocitinib ointment (CORECTIM), and others.
- In March 3, 2026, Kyowa Kirin announced the termination of all clinical trials for rocatinlimab.
Atopic Dermatitis Market Size and Forecast in the 7MM
- 2025 Atopic Dermatitis Market Size: ~USD 13,000 million
- 2036 Projected Atopic Dermatitis Market Size: ~USD 28,000 million
- Atopic Dermatitis Growth Rate (2026–2036): 7.0% CAGR
DelveInsight's ‘Atopic Dermatitis – Market Insights, Epidemiology and Market Forecast – 2036’ report delivers an in-depth understanding of the Atopic Dermatitis, historical and forecasted epidemiology, as well as the atopic dermatitis market trends in the United States, EU4 (Germany, Spain, Italy, and France) and the United Kingdom, and Japan.
The atopic dermatitis market report delivers a comprehensive analysis of the current treatment landscape, including standards of care, clinical practices, and evolving therapeutic algorithms. It evaluates atopic dermatitis patient burden trends, revenue & market share dynamics, peak patient share & therapy uptake analysis, and provides an in-depth market size assessment and growth rate projections (Historical & Forecast 2022–2036) across global regions. The report highlights key unmet medical needs in atopic dermatitis and maps the competitive and clinical landscape to uncover high‑value opportunities, providing a clear outlook on future market growth potential.
Scope of the Atopic Dermatitis Market Insights Report | |
|
Study Period |
2022–2036 |
|
Historical Year |
2022–2025 |
|
Forecast Period |
2026–2036 |
|
Base Year |
2026 |
|
Geographies Covered |
|
|
Atopic Dermatitis Market CAGR (Study period/Forecast period) |
7.0% (2025 ̶ 2036) |
|
Atopic Dermatitis Epidemiology Segmentation Analysis |
Patient Burden Assessment
|
|
Atopic Dermatitis Companies |
|
|
Atopic Dermatitis Therapies |
|
|
Atopic Dermatitis Market |
Segmented by
|
|
Analysis |
|
Key Factors Driving the Atopic Dermatitis Market
Rising Atopic Dermatitis Prevalence
The overall prevalence of atopic dermatitis increasing, particularly among females, is a primary driver of atopic dermatitis market expansion. In the US, there were ~33,120, 000 diagnosed prevalent cases of atopic dermatitis in 2025, which is projected to increase by 2036.
Rising Opportunities in Atopic Dermatitis
Recent approvals of JAK inhibitors like RINVOQ, CIBINQO, and OPZELURA has given a ray of hope to other key players developing JAKs for this indication, as even though they come with a black box warning, these drugs could give fair competition to DUPIXENT, and take up a good share of the market.
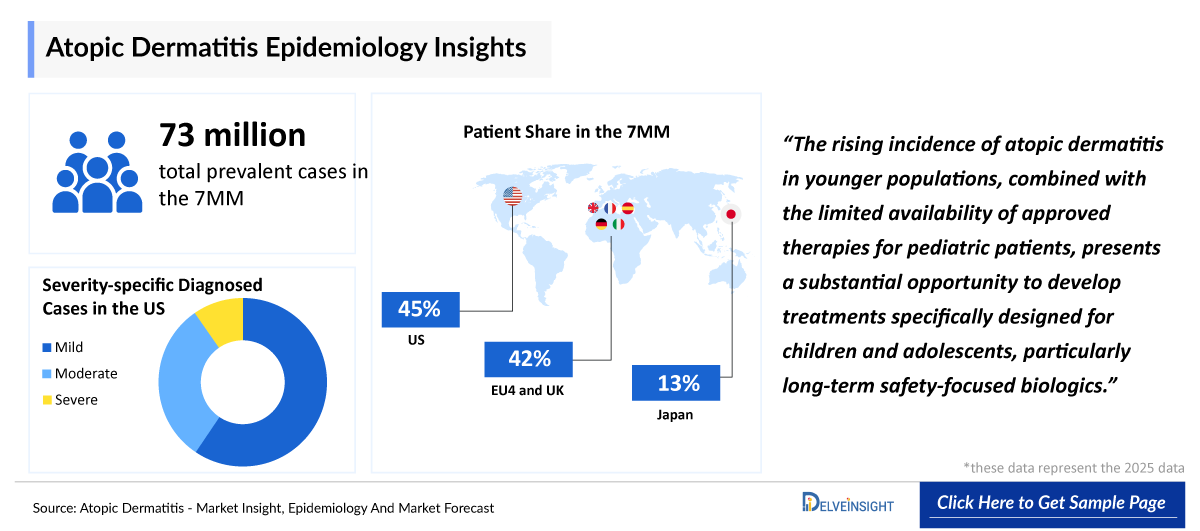
With increasing diagnoses in younger populations, there is significant potential for therapies tailored to children and adolescents, particularly long-term safety-focused biologics. Increased public awareness, improved access to health services, reimbursements, and financial support may boost the atopic dermatitis and pruritus in the market and improve the market accessibility of the emerging drugs.
Emerging Atopic Dermatitis Competitive Landscape
Some of the topic dermatitis drugs in clinical trials include Rocatinlimab, Amlitelimab, Orismilast, Ucenprubart, and others.
Atopic Dermatitis Understanding and Treatment Algorithm
Atopic Dermatitis Overview and Diagnosis
Atopic dermatitis, also called eczema, is a chronic condition and the most common type of skin inflammation that usually starts in early childhood but can occur at any age and can be recurrent or persistent throughout life. In ‘dermatitis,’ ‘derm’ means ‘skin,’ and ‘itis’ means ‘inflammation.’ Thus, dermatitis is a skin inflammation characterized by itchiness, redness, and a rash caused by genetics, an overactive immune system, infections, allergies, and irritating substances. Half of the patients with moderate-to-severe eczema also have asthma, hay fever (allergic rhinitis), and food allergies; it is the most common chronic skin disease in children.
Atopic Dermatitis Diagnosis
Atopic dermatitis is diagnosed clinically, as there is no specific laboratory test. Diagnosis is based on characteristic features especially pruritus (essential), chronic or relapsing eczematous lesions, typical distribution patterns (e.g., face/extensor areas in infants, flexural areas in older children and adults), and a personal or family history of atopy along with exclusion of other conditions.
Patients may also present with dry skin, excoriations, papules, and lichenification. Absence of itching should prompt reconsideration of the diagnosis. Atopic dermatitis must be differentiated from other scaly skin disorders (e.g., seborrheic dermatitis in infants). If treatment fails, alternative diagnoses, including nutritional, metabolic, or immunologic conditions, should be evaluated. Elevated IgE levels are common but are not diagnostic. Skin biopsy and microbiological tests may help rule out other or secondary conditions but are not definitive for atopic dermatitis.
Further details are provided in the report.
Atopic Dermatitis Treatment
The primary goals of atopic dermatitis treatment are to reduce inflammation and pruritus, restore and maintain the skin barrier, prevent flares, minimize infections, and improve quality of life and sleep. Management is stepwise and multidisciplinary, centered on pharmacologic and supportive care. Foundational therapy includes regular use of emollients to maintain skin hydration and topical corticosteroids as first-line anti-inflammatory treatment during flares, with topical calcineurin inhibitors (e.g., tacrolimus, pimecrolimus) or phosphodiesterase-4 inhibitors used for sensitive areas or long-term control.
For moderate-to-severe disease or inadequate response to topical therapy, systemic treatments are used, including biologics such as dupilumab and tralokinumab targeting type 2 inflammation, and oral Janus kinase (JAK) inhibitors (e.g., upadacitinib, abrocitinib) for rapid itch and inflammation control, though they require monitoring for safety risks. Adjunctive measures include antihistamines for itch relief (limited benefit), antimicrobial therapy for secondary infections, and phototherapy in appropriate patients.
Further details related to country-based variations are provided in the report.
Atopic Dermatitis Unmet Needs
The section “unmet needs of Atopic Dermatitis” outlines the critical gaps between the current state of patient care, diagnosis, and the ideal & effective management of the disease. It highlights the obstacles experienced by patients, clinicians, and researchers and identifies potential solutions for future progress.
- Disease burden and impact on the QoL (Quality of Life)
- Need for more effective treatment therapies
- Lack of personalized and age-specific treatment
- Lack of diagnostic guidelines for the management of atopic dermatitis and others…..
Comprehensive unmet needs insights in atopic dermatitis and their strategic implications are provided in the full report.
Atopic Dermatitis Epidemiology
Key Findings from Atopic Dermatitis Epidemiological Analysis and Forecast
- According to DelveInsight’s estimates, the total prevalent cases of atopic dermatitis in the 7MM were approximately 73,243,000 in 2025.
- It has been observed that the prevalence of atopic dermatitis is higher in children (<18 years); ~35% as compared to adults in the 7MM.
- In 2025, among severity of atopic dermatitis in the US, mild atopic dermatitis was the most prevalent subtype (~14,739,000 cases), followed by moderate atopic dermatitis.
- The atopic dermatitis is found to be more prevalent in females than in males.
Atopic Dermatitis Drug Chapters & Competitive Analysis
The Atopic dermatitis drug chapter provides a detailed, market-focused review of approved therapies and the emerging pipeline across Phase I–III clinical trials. It covers the mechanism of action, clinical trial data, regulatory approvals, patents, collaborations, and strategic partnerships for each therapy, along with their advantages, limitations, and recent developments. This section offers critical insights into the atopic dermatitis treatment landscape, supporting market assessment, competitive analysis, and growth forecasting for the atopic dermatitis therapeutics market.
Approved Therapies for Atopic Dermatitis
Upadacitinib (RINVOQ): AbbVie
RINVOQ/RINVOQ LQ is a Janus kinase (JAK) inhibitor indicated for the treatment of adults and pediatric patients 12 years of age and older with refractory, moderate to severe atopic dermatitis whose disease is not adequately controlled with other systemic drug products, including biologics, or when use of those therapies are inadvisable. Recently, the company announced topline results of LEVEL UP studies, in Phase III/IV and plans to present results from the LEVEL UP study at a future medical congress.
In January 2022, AbbVie announced that the US FDA approved RINVOQ (upadacitinib) for the treatment of moderate-to-severe atopic dermatitis in adults and adolescents aged 12 and older whose disease did not respond to previous treatment and is not well controlled with other pills or injections, including biologic medicines, or when the use of other pills or injections is not recommended
Atopic Dermatitis Marketed/Approved Therapies | ||||||
|
Drug/Therapy |
Company |
Indication |
Molecule Type |
RoA |
MoA |
Marketed Region |
|
Lebrikizumab (EBGLYSS) |
Roche/Eli Lilly and Company/Almirall |
Moderate-to-severe atopic dermatitis |
Monoclonal antibody |
SC |
IL-13 inhibitor |
US: 2024 EU: 2023 JP: 2024 |
|
Nemolizumab (MITCHGA/ NEMLUVIO) |
Chugai Pharmaceutical/ Galderma/ Maruho |
Pruritus associated with atopic dermatitis |
Monoclonal antibody |
SC |
IL-31 inhibitor |
US: 2024 EU: 2025 JP: 2022 |
|
Abrocitinib (CIBINQO) |
Pfizer |
Moderate-to-severe atopic dermatitis |
Small molecule |
Oral |
JAK-1 selective inhibitor |
US: 2022 EU: 2021 JP: 2021 |
Atopic Dermatitis Pipeline Analysis
Amlitelimab (KY1005): Sanofi
Amlitelimab (SAR445229, KY1005) is a fully human, non-T cell depleting monoclonal antibody that blocks OX40L, a key immune regulator. With its novel mechanism of action, amlitelimab aims to normalize the overactive immune system, without depleting T cells. It has the potential to be a first- or best-in-class treatment for a range of immune-mediated diseases and inflammatory disorders, including the anchor indication of moderate-to-severe atopic dermatitis.
As per the Q4 2026 presentation, the company plans to release the remaining Phase III data in H2 2026 and is also targeting regulatory submission within the same timeframe.
Comparison of Emerging Drugs Under Development | |||||||
|
Drug Name |
Company |
Highest Phase |
Indication |
RoA |
MoA |
Molecule Type |
Anticipated Launch in the US |
|
Amlitelimab (KY1005) |
Kymab/Sanofi |
III |
Moderate-to-severe-atopic-dermatitis |
IV/ SC |
OX40 inhibitor |
Monoclonal antibody |
2027 |
|
Bosakitug |
Biosion |
II |
Atopic dermatitis |
SC |
TSLP inhibitor |
Monoclonal antibody |
Information is available in the full report |
|
Orismilast |
UNION Therapeutics |
II |
Moderate-to-severe atopic dermatitis |
Oral |
PDE4 inhibitor |
Small molecule |
Information is available in the full report |
|
Note: Launch insights are provisional and may change with future report updates or the occurrence of major key catalysts. | |||||||
Atopic Dermatitis Key Players, Market Leaders and Emerging Companies
- Roche
- Eli Lilly and Company
- Chugai Pharmaceutical
- Galderma
- Maruho
- AbbVie
- Pfizer
- LEO Pharma
- Otsuka Pharmaceutical
- Incyte Corporation
- Torii Pharmaceutical
- Sanofi
- Regeneron Pharmaceuticals, and others
Atopic Dermatitis Drug Updates
- In March 2026, Sanofi presented new results from its Phase III amlitelimab studies in atopic dermatitis during a late-breaking research session at the American Academy of Dermatology Annual Meeting 2026.
- In March 2026, Eli Lilly announced positive Phase III results (ADorable-1 trial) for lebrikizumab in pediatric atopic dermatitis (≥6 months), demonstrating significant improvements in skin clearance and itch, with plans for regulatory submissions for label expansion.
- In January 2026, recent dermatology updates highlighted continued expansion of novel therapies for atopic dermatitis, including newer topical and biologic agents, reflecting rapid innovation and broader treatment options across age groups.
- In September 2025, the US Food and Drug Administration approved ruxolitinib cream (Opzelura) for pediatric patients aged 2–11 years, expanding access to a topical JAK inhibitor for atopic dermatitis.
Drug Class Insights
Atopic Dermatitis Market Outlook
For most people with atopic dermatitis, emollients and prescription topical therapies are sufficient to achieve control. In contrast, people with more severe or widespread atopic dermatitis, people with substantially impaired QoL, and individuals whose atopic dermatitis is refractory to optimized topical therapy may consider the use of phototherapy or systemic therapies to improve disease control and QOL.
Several effective treatments have become available in recent years for patients with atopic dermatitis, and more are in development. The current market uses several off-label therapies apart from pharmacological systemic treatment. Localized atopic itch is frequently treated with topical or intralesional therapeutic agents. Topical corticosteroids have been used for atopic dermatitis for decades. Pimecrolimus and tacrolimus, topical calcineurin inhibitors, are used as steroid-sparing agents for patients with atopic dermatitis. Phosphodiesterase-4 (PDE-4) inhibitors are an alternative option for the topical treatment of pruritis in atopic dermatitis. Oral antihistamines are commonly used for atopic itch. Additionally, phototherapy is efficacious in targeting atopic itch. Systemic pharmacologic treatments have proven to be beneficial in patients who are unresponsive to topical therapies.
The first FDA-approved targeted systemic treatment for atopic dermatitis was DUPIXENT (dupilumab), a human monoclonal antibody that inhibits IL-4 and IL-13 signaling. Dupilumab is indicated for the treatment of adult and pediatric patients 6 months and older with moderate-to-severe atopic dermatitis whose disease is not adequately controlled with topical prescription therapies or when use of those therapies is not advisable. The drug was approved in the European Union in 2017, as well as in Japan in 2018.
In December 2021, tralokinumab (ADBRY/ADTRALZA) became the second biologic indicated for the treatment of atopic dermatitis and the first treatment to target IL-13 in the US specifically. Tralokinumab is indicated for the treatment of moderate-to-severe atopic dermatitis in patients 12 years and older whose disease is not adequately controlled with topical prescription therapies or when use of those therapies is not advisable.
AbbVie’s RINVOQ has gained strong traction in the atopic dermatitis market, supported by head-to-head trials versus DUPIXENT demonstrating statistically significant superiority on stringent endpoints such as EASI-90 and minimal itch. These results have positioned RINVOQ as a leading therapy, ranking second in the market.
- According to the estimates, the largest market size of atopic dermatitis was captured by the United States, i.e., ~USD 9,000 million in 2025.
- The atopic dermatitis market is evolving as treatment focus shifts from broad-spectrum biologics to more targeted therapies, with JAK inhibitors and IL-targeted agents driving adoption due to their favorable efficacy, safety, and expanded pediatric indications.
- Regional market dynamics show the US leading in revenue, while Japan and EU markets demonstrate steady growth, reflecting increasing diagnosis rates, broader pediatric access, and rising adoption of novel mechanisms.
- In 2025, among all the therapies for atopic dermatitis, the highest revenue is estimated to be generated by DUPIXENT, in the United States.
Further details will be provided in the report….
Drug Class/Insights into Leading Emerging and Marketed Therapies in Atopic Dermatitis (2022–2036 Forecast)
The atopic dermatitis market comprises targeted biologics, oral small molecules, topical therapies, and emerging immune-modulating approaches, each addressing distinct inflammatory pathways such as type 2 cytokine signaling, JAK-STAT pathways, and skin barrier dysfunction underlying atopic dermatitis.
Targeted biologics: Dupilumab (DUPIXENT) is a first-in-class IL-4 receptor alpha antagonist that inhibits IL-4 and IL-13 signaling, the central drivers of type 2 inflammation in atopic dermatitis. It has demonstrated strong efficacy and long-term safety across age groups, establishing biologics as the cornerstone of moderate-to-severe atopic dermatitis treatment. Newer biologics such as Tralokinumab (ADTRALZA) and Lebrikizumab further validate IL-13 as a critical therapeutic target and are expanding competition within this class.
Oral targeted small molecules (JAK inhibitors): Upadacitinib (RINVOQ) and Abrocitinib (CIBINQO) are selective JAK1 inhibitors that provide rapid itch relief and significant skin clearance by modulating multiple cytokine pathways involved in AD pathogenesis. These oral agents highlight the growing preference for fast-acting, convenient systemic therapies and are expected to drive significant uptake, particularly among patients seeking alternatives to injectables.
Emerging biologics and novel immune targets: Pipeline agents such as nemolizumab, targeting pruritus-specific pathways, and OX40/OX40L inhibitors are advancing the next wave of atopic dermatitis therapies. These agents aim to improve itch control and disease modification, addressing unmet needs in patients with inadequate response to current biologics.
Biologics and JAK inhibitors together define the core innovation landscape in atopic dermatitis, with biologics firmly established as first-line systemic therapies and oral small molecules driving future market expansion through convenience, rapid efficacy, and broader patient applicability during the 2022–2036 forecast period.
Further details will be provided in the report….
Atopic Dermatitis Drug Uptake
This section focuses on the uptake rate of potential drugs expected to be launched in the market during the forecast period (2026–2036). The analysis covers the atopic dermatitis market's uptake by drugs, patient uptake by therapy, and sales of each drug.
The uptake of therapies in atopic dermatitis is expected to vary across targeted biologics, oral small-molecule inhibitors, and topical agents. Recently approved and emerging targeted therapies such as dupilumab (DUPIXENT), tralokinumab (ADTRALZA), and upadacitinib (RINVOQ) demonstrate strong and sustained uptake, while lebrikizumab is anticipated to show progressive and competitive uptake, supported by their targeted mechanisms, robust efficacy in moderate-to-severe disease, and increasing physician confidence in biologic and JAK inhibitor use. Their adoption is primarily driven by the limitations of conventional topical corticosteroids and systemic immunosuppressant’s in achieving long-term disease control, as well as the growing demand for therapies that provide rapid itch relief and durable skin clearance.
In comparison, other next-generation therapies such as Abrocitinib (CIBINQO) and Baricitinib are expected to show gradual but steady uptake as long-term safety data matures and their positioning becomes clearer within treatment algorithms. Additionally, novel agents like Nemolizumab and emerging OX40 pathway inhibitors are projected to gain traction over the forecast period, particularly in patients with persistent pruritus or inadequate response to existing biologics, further diversifying the atopic dermatitis treatment landscape.
Further detailed analysis of emerging therapies' drug uptake in the report…
Market Access and Reimbursement of Atopic Dermatitis
The United States
|
The US Reimbursement for Atopic Dermatitis Therapies | |
|
Drug |
Access Program |
|
Upadacitinib (RINVOQ) |
|
|
Dupilumab (DUPIXENT) |
|
Reimbursement is a crucial factor that affects the drug’s access to the market. Often, the decision to reimburse comes down to the price of the drug relative to the benefit it produces in treated patients. To reduce the healthcare burden of these high-cost therapies, many payment models are being considered by payers and other industry insiders.
Atopic Dermatitis Therapies Price Scenario & Trends
Pricing and analogue assessment of atopic dermatitis therapies highlights evolving price dynamics structures. This section summarizes the cost of approved treatments, closest and most appropriate analogue selection for emerging therapies, and understanding of how pricing influences market access, adherence, and long-term uptake.
Pricing of Atopic Dermatitis Approved Drugs
The wholesale acquisition cost (WAC) of DUPIXENT subcutaneous solution 300 mg/2 mL in 2020 was USD 3,171, resulting in an annual cost of USD 42,809.
Further details are provided in the final report….
Industry Experts and Physician Views for Atopic Dermatitis
To keep up with atopic dermatitis market trends, we take Key Opinion Leaders (KOLs) and Subject Matter Experts (SMEs) opinions working in the domain through primary research to fill the data gaps and validate our secondary research. Industry experts were contacted for insights on the atopic dermatitis emerging therapies, evolving treatment landscape, patient adherence to conventional therapies, therapy switching trends, drug adoption and uptake, accessibility challenges, and epidemiology and real-world prescription patterns in atopic dermatitis, including MD, PhD, Instructor, Postdoctoral Researcher, Professor, Researcher, and others.
DelveInsight’s analysts connected with 10+ KOLs to gather insights; however, interviews were conducted with 6+ KOLs in the 7MM. Centers such as the University of North Carolina at Chapel Hill, Berlin Institute of Health at Charité, and the University of Nottingham, etc. were contacted. Their opinion helps understand and validate current and emerging atopic dermatitis therapies, highlight unmet medical needs, provide epidemiological context, and support strategic decisions for market access, therapy adoption, and pipeline prioritization in atopic dermatitis.
|
Region |
Key Opinion Leaders (KOLs) and Subject Matter Experts (SMEs) |
|
United States |
“There are still a ton of unmet needs for atopic dermatitis. I think one of the biggest areas right now, and a big focus, is primary prevention. We would love to figure out how to prevent this disease, and right behind that is what do we do for our littlest ones? For other patients, I think we still need a lot more in the ICH [International Conference on Harmonization] domain. We still need to understand the microbiome.” |
|
France |
“Patients go to alternative approaches because they are quite unsatisfied with current treatment; and because most healthcare professionals’ approach is ‘disease orientated.” |
Qualitative Analysis: SWOT and Conjoint Analysis
We perform qualitative and market Intelligence analysis using various approaches, such as SWOT analysis and conjoint analysis.
In the SWOT analysis of Atopic Dermatitis, strengths, weaknesses, opportunities, and threats in terms of disease diagnosis, patient awareness, patient burden, competitive landscape, cost-effectiveness, and geographical accessibility of therapies are provided.
Conjoint analysis analyzes emerging therapies based on relevant attributes such as safety, efficacy, frequency of administration, route of administration, and order of entry. Scoring is given based on these parameters to analyze the effectiveness of therapy.
The team of analysts analyzes promising emerging therapies based on relevant attributes such as safety, efficacy, frequency of administration, route of administration, and order of entry. In efficacy, the trial’s primary and secondary outcome measures are evaluated, whereas the therapies’ safety is evaluated, wherein the acceptability, tolerability, and adverse events are majorly observed. In addition, the scoring is also based on the route of administration, order of entry, probability of success, and the addressable patient pool for each therapy. According to these parameters, the final weightage score and the ranking of the emerging therapies are decided.
Scope of the Atopic Dermatitis Market Report
- The report covers a segment of key events, an executive summary, a descriptive overview of atopic dermatitis, explaining its causes, signs and symptoms, pathogenesis, and currently available treatments.
- Comprehensive insight has been provided into the epidemiology segments and forecasts, the future growth potential of the diagnosis rate, and disease progression along treatment guidelines.
- Additionally, an all-inclusive account of both the current and emerging treatments, along with the elaborative profiles of late-stage and prominent therapies, will have an impact on the current treatment landscape.
- A detailed review of the atopic dermatitis market, historical and forecasted market size, market share by therapies, detailed assumptions, and rationale behind our approach is included in the report, covering the 7MM drug outreach.
- The report provides an edge while developing business strategies by understanding trends through SWOT analysis and expert insights/KOL views, patient journey, and treatment preferences that help in shaping and driving the 7MM atopic dermatitis market.
Atopic Dermatitis Market Report Insights
- Atopic Dermatitis Patient Population Forecast
- Atopic Dermatitis Therapeutics Market Size
- Atopic Dermatitis Pipeline Analysis
- Atopic Dermatitis Market Size and Trends
- Atopic Dermatitis Market Opportunity (Current and forecasted)
Atopic Dermatitis Market Report Key Strengths
- Epidemiology‑based (Epi‑based) Bottom‑up Forecasting
- Artificial Intelligence (AI)-enabled Market Research Report
- 11-year forecast
- Atopic Dermatitis Market Outlook (North America, Europe, Asia-Pacific)
- Patient Burden Trends (by geography)
- Atopic Dermatitis Treatment Addressable Market (TAM)
- Atopic Dermatitis Competitive Landscape
- Atopic Dermatitis Major Companies Insights
- Atopic Dermatitis Price Trends and Analogue Assessment
- Atopic Dermatitis Therapies Drug Adoption/Uptake
- Atopic Dermatitis Therapies Peak Patient Share analysis
Atopic Dermatitis Market Report Assessment
- Atopic Dermatitis Current Treatment Practices
- Atopic Dermatitis Unmet Needs
- Atopic Dermatitis Clinical Development Analysis
- Atopic Dermatitis Emerging Drugs Product Profiles
- Atopic Dermatitis Market Attractiveness
- Atopic Dermatitis Qualitative Analysis (SWOT and Conjoint Analysis)
Key Questions Answered in the Atopic Dermatitis Market Report
Market Insights
- What was the atopic dermatitis market size, the market size by therapies, market share (%) distribution in 2025, and what would it look like by 2036? What are the contributing factors for this growth?
- What are the anticipated pricing variations among different geographies for the emerging therapies in the future?
- What can be the future treatment paradigm of atopic dermatitis?
- What are the disease risks, burdens, and unmet needs of atopic dermatitis? What will be the growth opportunities across the 7MM concerning the patient population with Atopic Dermatitis?
- Who is the major future competitor in the market, and how will the competitors affect their market share?
- What are the current options for the treatment of atopic dermatitis? What are the current guidelines for treating atopic dermatitis in the US, Europe, and Japan?
Reasons to Buy Atopic Dermatitis Market Report
- The report will help in developing business strategies by understanding the latest trends and changing treatment dynamics driving the atopic dermatitis market.
- Bottom up forecasting builds from the affected population to product forecasts, delivering a robust, data driven approach ideal for new therapies and novel classes.
- Insights on patient burden/disease incidence, evolution in diagnosis, and factors contributing to the change in the epidemiology of the disease during the forecast years.
- Understand the existing market opportunities in varying geographies and the growth potential over the coming years.
- Identifying strong upcoming players in the market will help devise strategies to help get ahead of competitors.
- Detailed analysis and ranking of class-wise potential current and emerging therapies under the conjoint analysis section to provide visibility around leading classes.
- To understand KOLs’ perspectives on the accessibility, acceptability, and compliance-related challenges of existing treatment to overcome barriers in the future.
- Detailed insights on the unmet needs of the existing market so that the upcoming players can strengthen their development and launch strategy.
- This Artificial Intelligence (AI) enabled report summarize and simplify complex datasets within the report into clear, actionable insights for stakeholders, investors, and healthcare providers, enabling faster, data driven decisions.




-market.png&w=256&q=75)

