Acute Kidney Injury Market Summary
- The Acute Kidney Injury Market Size is anticipated to grow with a significant CAGR during the study period (2020-2034).
- The Acute Kidney Injury Market companies developing therapies include - Fresenius Medical Care, Baxter International, Medtronic, B. Braun Melsungen AG, Nikkiso Co., Ltd., Nipro Corporation, Asahi Kasei Medical, AM-Pharma, Atox Bio, Guard Therapeutics, Quark Pharmaceuticals, Angion Biomedica Corp., Pharmazz, Inc., Durect Corporation, Kringle Pharma, bioMérieux, BioPorto Diagnostics, Renalyx, Purespring Therapeutics, Maze Therapeutics, Sentien Biotechnologies, and others.
Acute Kidney Injury Market and Epidemiology Analysis
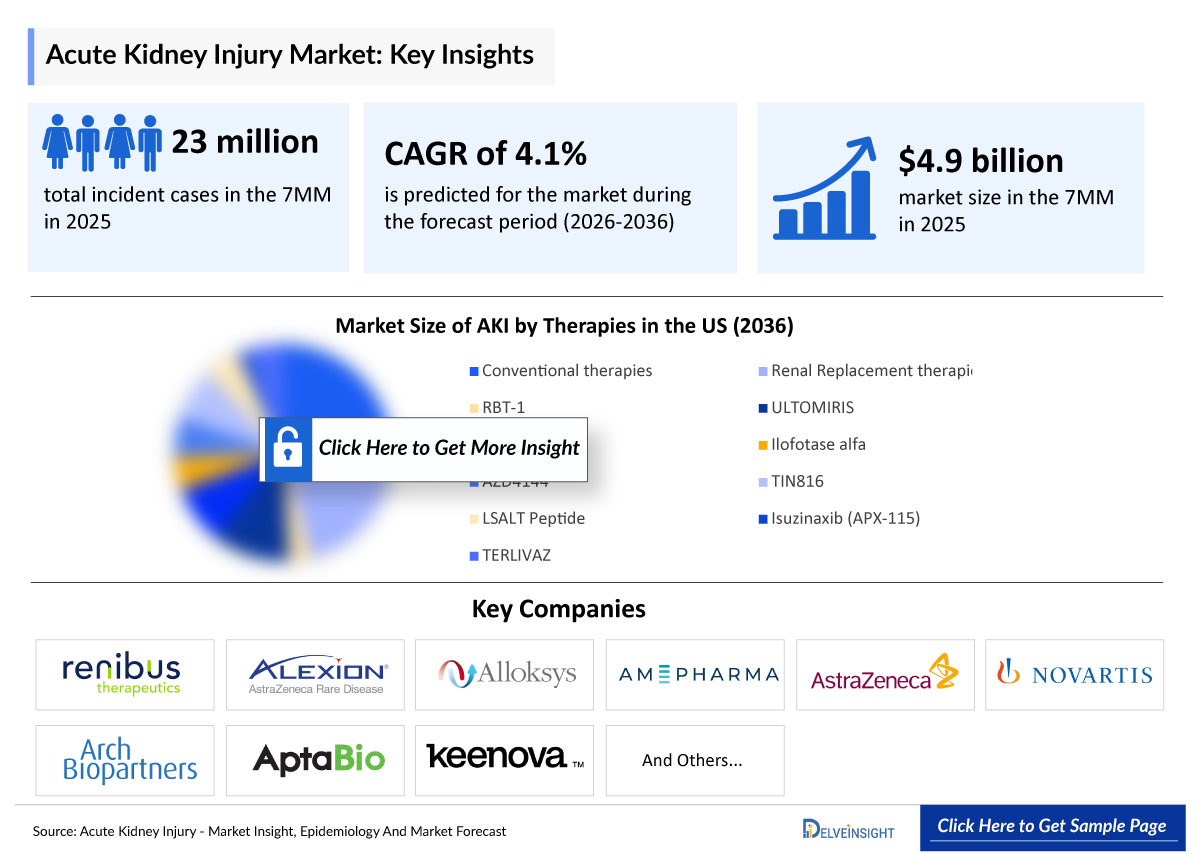
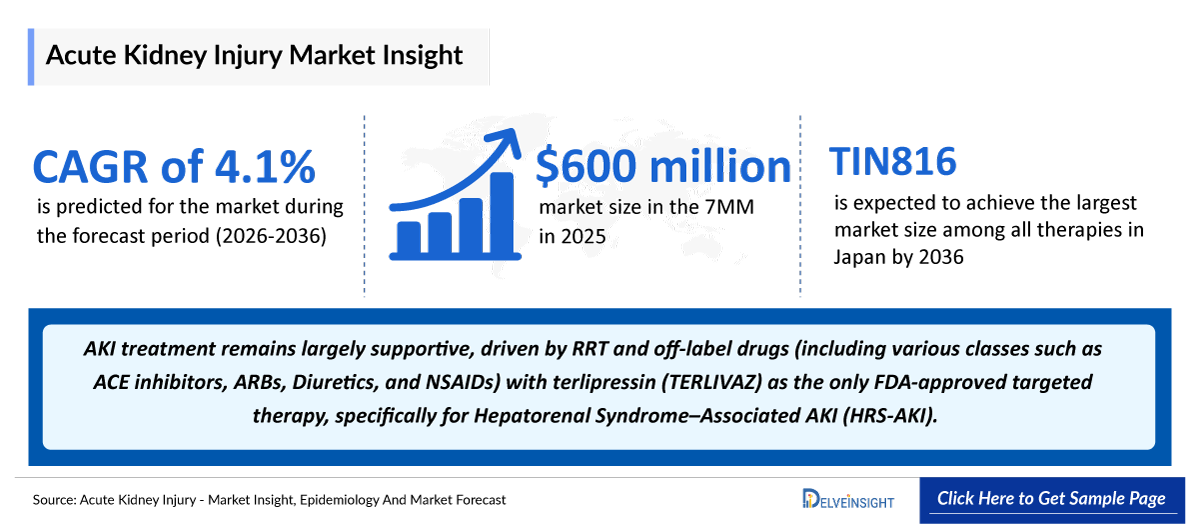
- According to DelveInsight’s analysis, the Acute Kidney Injury market size was found to be ~USD 4,900 million in the leading markets (the United States, the EU4 (Germany, France, Italy, and Spain), the United Kingdom, and Japan) in 2025.
- The incidence of AKI is on the rise, particularly in individuals with acute illnesses and potentially those undergoing significant surgical procedures.
- Acute Kidney Injury treatment remains largely supportive, driven by RRT and off-label drugs (including various classes such as ACE inhibitors, ARBs, Diuretics, and NSAIDs) with terlipressin (TERLIVAZ) as the only FDA-approved targeted therapy, specifically for Hepatorenal Syndrome–Associated AKI (HRS-AKI).
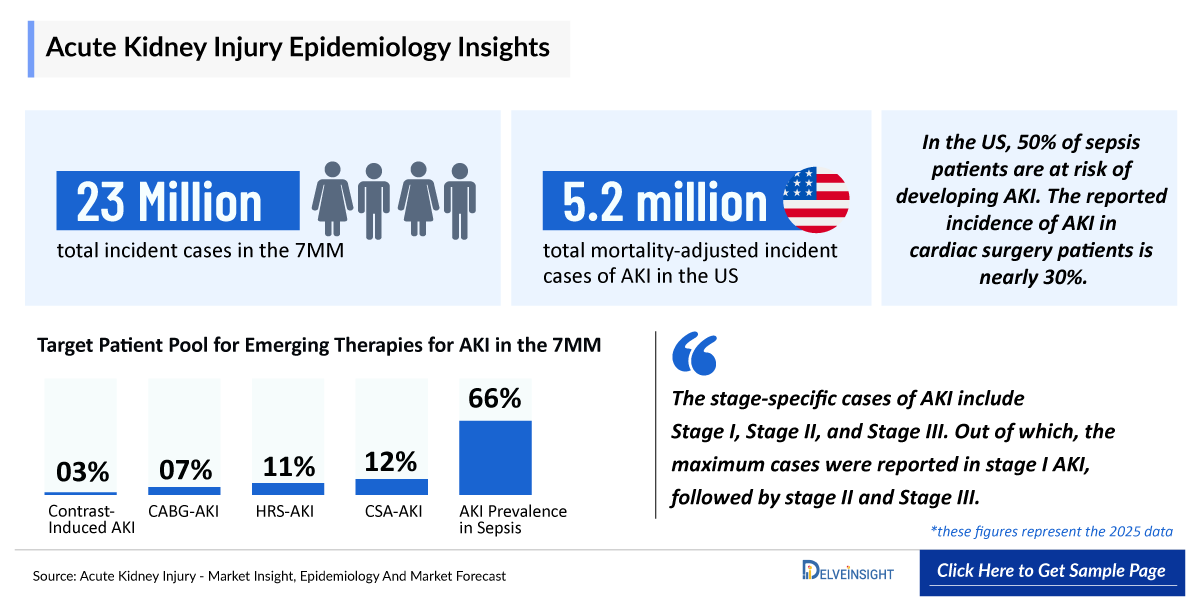
- Total mortality-adjusted incident cases of AKI in hospitalized patients in the 7MM accounted for approximately 13 million in 2025.
- In 2025, the market size of AKI was highest in the US among the 7MM, accounting for approximately USD 54.9% million, which is further expected to increase by 2036.
- Treatment is currently dominated by supportive care such as dialysis and off-label therapies, while companies like AM-Pharma (ilofotase alfa), SeaStar Medical (QUELIMMUNE), Renibus Therapeutics (RBT-1), and Arch Biopartners (LSALT Peptide) are actively developing novel targeted treatments for AKI.
- In April 2025, Renibus Therapeutics announced that the topline results from the Phase III (PROTECT study) evaluating the effect of RBT-1 on reducing the risk of post-operative complications in patients undergoing cardiac surgery are anticipated in Q3 2025.
Key Factors Driving the Acute Kidney Injury (AKI) Market
Rising Prevalence of Acute Kidney Injury
The growing number of Acute Kidney Injury cases worldwide is one of the primary factors driving market growth. Acute Kidney Injury frequently occurs among hospitalized and critically ill patients, particularly those suffering from conditions such as sepsis, trauma, or cardiovascular diseases. As the patient pool expands, the demand for effective treatment and management solutions increases.
Increasing Burden of Chronic Diseases
Chronic conditions such as diabetes, hypertension, and chronic kidney disease significantly increase the risk of developing AKI. The rising global prevalence of these diseases contributes to higher incidence rates, thereby expanding the AKI treatment market.
Aging Global Population
Older individuals are more susceptible to kidney dysfunction and related complications. As the global population continues to age, the incidence of AKI is expected to increase, which in turn drives demand for dialysis, medications, and supportive care therapies.
Request a sample to unlock the CAGR for "Acute Kidney Injury Market Forecast"
DelveInsight's ‘Acute Kidney Injury Market Insights, Epidemiology and Market Forecast 2036’ report delivers an in-depth understanding of the Acute Kidney Injury (AKI), historical and forecasted epidemiology, as well as the Acute Kidney Injury (AKI) market trends in the United States, EU4 (Germany, Spain, Italy, and France) and the United Kingdom, and Japan.
The Acute Kidney Injury (AKI) market report delivers a comprehensive analysis of the current treatment landscape, including standards of care, clinical practices, and evolving therapeutic algorithms. It evaluates Acute Kidney Injury (AKI) patient burden trends, revenue & market share dynamics, peak patient share & therapy uptake analysis, and provides an in-depth market size assessment, and growth rate projections (Historical & Forecast 2022–2036) across global regions. The report highlights key unmet medical needs in Acute Kidney Injury (AKI) and maps the competitive and clinical landscape to uncover high‑value opportunities, providing a clear outlook on future market growth potential.
Scope of the Acute Kidney Injury Market | |
|
Study Period |
2020 to 2036 |
|
Forecast Period |
2024-2036 |
|
Geographies Covered |
|
|
Acute Kidney Injury Market |
|
|
Acute Kidney Injury Market Size | |
|
Acute Kidney Injury Companies |
Fresenius Medical Care, Baxter International, Medtronic, B. Braun Melsungen AG, Nikkiso Co., Ltd., Nipro Corporation, Asahi Kasei Medical, AM-Pharma, Atox Bio, Guard Therapeutics, Quark Pharmaceuticals, Angion Biomedica Corp., Pharmazz, Inc., Durect Corporation, Kringle Pharma, bioMérieux, BioPorto Diagnostics, Renalyx, Purespring Therapeutics, Maze Therapeutics, Sentien Biotechnologies, and others. |
|
Acute Kidney Injury Market Unmet Need |
The Acute Kidney Injury (AKI) market faces significant unmet needs due to limited targeted therapies and heavy reliance on supportive care such as dialysis. Early diagnosis remains challenging, and effective treatments to prevent kidney damage or promote recovery are scarce. This highlights the need for innovative drugs, improved biomarkers, and advanced therapeutic approaches. |
Acute Kidney Injury (AKI) Understanding and Treatment Algorithm
Acute Kidney Injury Overview
Acute Kidney Injury (AKI) is a sudden decline in kidney function that occurs within hours or days, leading to the accumulation of waste products, electrolyte imbalance, and fluid overload in the body. It is commonly observed in hospitalized patients, particularly those in intensive care units (ICUs). AKI can develop due to several causes, including reduced blood flow to the kidneys, direct kidney damage from toxins or infections, and obstruction in the urinary tract. Major risk factors include sepsis, major surgery, dehydration, chronic diseases such as diabetes and hypertension, and exposure to nephrotoxic medications.
Acute Kidney Injury Market Diagnosis
The Acute Kidney Injury diagnosis of Acute Kidney Injury primarily focuses on identifying changes in kidney function through laboratory tests and clinical evaluation. Physicians commonly monitor serum creatinine levels and urine output, as sudden increases in creatinine or reduced urine production can indicate impaired kidney function. Blood tests are often combined with urinalysis to evaluate the presence of proteins, blood cells, or other abnormalities that may signal kidney damage. Imaging techniques such as ultrasound or CT scans are frequently used to detect structural abnormalities, urinary tract obstruction, or kidney swelling. In some cases, kidney biopsy may be performed to determine the exact cause of kidney injury, especially when intrinsic kidney diseases are suspected.
Acute Kidney Injury Market Treatment
The Acute Kidney Injury treatment of Acute Kidney Injury primarily focuses on addressing the underlying cause of kidney damage and preventing further complications. Initial management often involves stabilizing the patient by correcting fluid imbalances, maintaining blood pressure, and managing electrolyte levels. If AKI results from dehydration or blood loss, intravenous fluids may be administered to restore proper circulation to the kidneys. In cases caused by infections, antibiotics or other appropriate medications are prescribed.
Further details related to country-based variations are provided in the report.
Acute Kidney Injury (AKI) Epidemiology
The Acute Kidney Injury epidemiology highlights a significant and increasing healthcare burden worldwide, particularly among hospitalized and critically ill patients. AKI frequently occurs in intensive care units and is often associated with conditions such as sepsis, major surgeries, trauma, and cardiovascular diseases. The risk of AKI is higher among elderly individuals and patients with underlying disorders such as diabetes, hypertension, and chronic kidney disease. Advances in diagnostic tools and improved clinical awareness have led to better identification of AKI cases. Additionally, the growing use of nephrotoxic drugs and contrast agents has contributed to rising incidence rates across global healthcare settings.
Key Findings from Acute Kidney Injury Epidemiological Analyses and Forecast
- According to DelveInsight’s estimates, the total incident cases of AKI in the 7MM were approximately 23,076,000 in 2025.
- The total mortality-adjusted incident cases of AKI in the US were approximately 5.2 million in 2025.
- Among EU4 and the UK, UK accounted for the highest number of AKI cases, followed by Germany, whereas Spain accounted for the lowest number of cases in 2025.
- The stage-specific cases of AKI include Stage I, Stage II, and Stage III. Out of which, the maximum cases were reported in stage I AKI, followed by stage II and Stage III.
Acute Kidney Injury Market Recent Developments and Breakthroughs
- In November 2025, Mallinckrodt announced the completion of the planned spin-off of its Par Health generic pharmaceuticals and sterile injectable businesses. Moving ahead, Mallinckrodt will be known as Keenova Therapeutics and will focus on developing, manufacturing, and commercializing branded therapeutics that help patients with rare or unaddressed conditions live happier and healthier lives.
- In October 2025, AM-Pharma announced that the topline data of the Phase II clinical trial evaluating ilofotase alfa for the prevention of Cardiac Surgery-associated Renal Damage (CSA-RD) is expected in early 2026, and the full dataset will be presented thereafter at a scientific conference. The company is anticipated to initiate a pivotal Phase III trial for ilofotase alfa.
Acute Kidney Injury Drug Analysis
The Acute Kidney Injury (AKI) drug landscape remains limited, as there are currently few targeted therapies specifically approved to treat or reverse the condition. Most treatment strategies focus on supportive care and management of the underlying cause rather than direct pharmacological intervention. Medications are typically used to control contributing factors such as infections, inflammation, electrolyte imbalance, and fluid overload. Diuretics, vasopressors, and antibiotics are commonly administered depending on the patient’s clinical condition.
Acute Kidney Injury Marketed Drugs
Terlipressin (TERLIVAZ): Keenova Therapeutics (Mallinckrodt)
TERLIVAZ is a vasopressin analog developed for the treatment of HRS-AKI, a rapidly progressive and life-threatening form of renal failure seen in patients with advanced liver disease. It was approved by the US FDA in 2022 as the first pharmacological therapy specifically indicated for HRS-AKI, addressing a major unmet need in a condition historically associated with very high short-term mortality. TERLIVAZ offers meaningful value as a first-in-class approved therapy; its regulatory journey was notably prolonged, having faced multiple Complete Response Letters (CRLs) before eventual approval.
RBT-1: Renibus Therapeutics
RBT-1 by Renibus Therapeutics is an investigational drug that upregulates anti-inflammatory, antioxidant, and iron-scavenging pathways, activating a cytoprotective preconditioning response to reduce the risk of postoperative complications and improve patient outcomes following cardiothoracic surgery. In April 2025, Renibus Therapeutics announced that the topline results from the Phase III (PROTECT study) evaluating the effect of RBT-1 on reducing the risk of post-operative complications in patients undergoing cardiac surgery are anticipated in Q3 2025.
Ravulizumab: AstraZeneca
Ravulizumab (ULTOMIRIS), a long-acting C5 complement inhibitor, offers immediate, complete, and sustained complement inhibition. The medication works by inhibiting the C5 protein in the terminal complement cascade, a part of the body’s immune system. The company is currently conducting a Phase III trial of ravulizumab for CSA-AKI. As per the AstraZeneca Q4 2025 presentation, the data from the Phase III ARTEMIS trial for CSA-AKI is anticipated in H2 2026.
Acute Kidney Injury Market Outlook
The Acute Kidney Injury (AKI) market is expected to grow steadily due to rising incidence of kidney-related complications, increasing hospital admissions, and advancements in diagnostic technologies. Growing awareness about early detection and improved healthcare infrastructure are also supporting market expansion. In addition, ongoing research and development of novel therapies aimed at protecting kidney function are anticipated to create new growth opportunities in the coming years.
Key Findings from Acute Kidney Injury Market Forecast Report
- The total market size of AKI in the 7MM was ~USD 4,900 million in 2025 and is projected to grow during the forecast period (2026-2036).
- According to the estimates, the highest market size of AKI is from the United States in 2025 and is anticipated to grow at a CAGR of 5.1%.
- Among EU4 and the UK, UK has the maximum revenue share in 2025, while Spain has the lowest market share.
- TIN816 is expected to achieve the largest market size among all therapies in Japan by 2036.
Acute Kidney Injury Market Competitive Landscape
The Acute Kidney Injury (AKI) market is characterized by the presence of several global healthcare and biotechnology companies focusing on dialysis technologies, diagnostics, and emerging therapeutic solutions. Key players are investing in research and development to introduce innovative treatments that address the underlying causes of kidney injury. The market competition is also driven by advancements in renal replacement therapies, improved biomarker-based diagnostic tools, and strategic collaborations between pharmaceutical and medical device companies. In addition, increasing clinical trials and product development initiatives are strengthening the pipeline landscape, enabling companies to expand their market presence and improve treatment options for AKI patients.
Key Acute Kidney Injury Market Companies
The Acute Kidney Injury Market companies developing therapies include -
- Fresenius Medical Care,
- Baxter International,
- Medtronic,
- B. Braun Melsungen AG,
- Nikkiso Co.,
- Nipro Corporation,
- Asahi Kasei Medical,
- AM-Pharma,
- Atox Bio,
- Guard Therapeutics,
- Quark Pharmaceuticals,
- Angion Biomedica Corp.,
- Pharmazz, Inc.,
- Durect Corporation,
- Kringle Pharma,
- bioMérieux,
- BioPorto Diagnostics,
- Renalyx,
- Purespring Therapeutics,
- Maze Therapeutics,
- Sentien Biotechnologies, and others.
Further details will be provided in the report….
Acute Kidney Injury (AKI) Drug Uptake
This section focuses on the uptake rate of potential drugs expected to be launched in the market during 2024–2034, which depends on the competitive landscape, safety, and efficacy data along with order of entry. It is important to understand that the key players evaluating their novel therapies in pivotal and confirmatory trials should remain vigilant when selecting appropriate comparators to stand the greatest chance of receiving a positive opinion from regulatory bodies, leading to approval, smooth launch, and rapid uptake.
Further detailed analysis of emerging therapies drug uptake in the report provides insights into expected adoption patterns, peak share potential, and factors influencing physician preference and patient access across key markets.
Acute Kidney Injury Clinical Trials Activities
The Acute Kidney Injury pipeline report insights into Acute Kidney Injury clinical trials across Phase III and Phase II stages. It highlights key pharmaceutical and biotechnology companies involved in developing targeted therapies aimed at addressing underlying mechanisms such as inflammation, oxidative stress, and tissue damage associated with AKI.
Acute Kidney Injury Pipeline Development Activities
The Acute Kidney Injury Clinical trial analysis report covers detailed information on collaborations, acquisitions, mergers, licensing agreements, and patent filings related to emerging Acute Kidney Injury therapies. It provides a strategic overview of how companies are strengthening their pipelines through partnerships and innovation-driven approaches.
Further detailed analysis of emerging therapies' drug uptake in the report…
Acute Kidney Injury Market Access and Reimbursement
TERLIVAZ was granted the new technology add-on payment (NTAP) by the Centers for Medicare & Medicaid Services (CMS) effective October 1, 2023. CMS created the NTAP to help ensure hospitals do not incur significant reimbursement shortfalls when adopting innovative new technologies. NTAP is an additional payment on top of the Medicare Severity Diagnosis-Related Group (MS-DRG) or TRICARE DRG reimbursement. Reimbursement is a crucial factor that affects the drug’s access to the market. Often, the decision to reimburse comes down to the price of the drug relative to the benefit it produces in treated patients. To reduce the healthcare burden of these high-cost therapies, many payment models are being considered by payers and other industry insiders.
Further details are provided in the final report….
Acute Kidney Injury Qualitative Analysis
We perform qualitative and market Intelligence analysis using various approaches, such as SWOT analysis and conjoint analysis.In the SWOT analysis of Acute Kidney Injury (AKI), strengths, weaknesses, opportunities, and threats in terms of disease diagnosis, patient awareness, patient burden, competitive landscape, cost-effectiveness, and geographical accessibility of therapies are provided.Conjoint analysis analyzes emerging therapies based on relevant attributes such as safety, efficacy, frequency of administration, route of administration, and order of entry. Scoring is given based on these parameters to analyze the effectiveness of therapy.
Scope of the Acute Kidney Injury Market Report
- The Acute Kidney Injury Market report covers a segment of key events, an executive summary, a descriptive overview of AKI, explaining its causes, signs and symptoms, pathogenesis, and currently available treatments.
- Comprehensive insight has been provided into the epidemiology segments and forecasts, the future growth potential of the diagnosis rate, and disease progression along treatment guidelines.
- Additionally, an all-inclusive account of both the current and emerging treatments, along with the elaborate profiles of late-stage and prominent therapies, will have an impact on the current treatment landscape.
- A detailed review of the AKI market, historical and forecasted market size, market share by therapies, detailed assumptions, and rationale behind our approach is included in the report, covering the 7MM drug outreach.
- The report provides an edge while developing business strategies by understanding trends through SWOT analysis and expert insights/KOL views, patient journey, and treatment preferences that help in shaping and driving the 7MM AKI market.
Acute Kidney Injury Market Report Insights
- Acute Kidney Injury (AKI) Patient Population Forecast
- Acute Kidney Injury (AKI) Therapeutics Market Size
- Acute Kidney Injury (AKI) Pipeline Analysis
- Acute Kidney Injury (AKI) Market Size and Trends
- Acute Kidney Injury (AKI) Market Opportunity (Current and forecasted)
Acute Kidney Injury Market Report Key Strengths
- Epidemiology‑based (Epi‑based) Bottom‑up Forecasting
- Artificial Intelligence (AI)-enabled Market Research Report
- 11-year forecast
- Acute Kidney Injury (AKI) Market Outlook (North America, Europe, Asia-Pacific)
- Patient Burden Trends (by geography)
- Acute Kidney Injury (AKI) Treatment Addressable Market (TAM)
- Acute Kidney Injury (AKI) Competitive Landscape
- Acute Kidney Injury (AKI) Major Companies Insights
- Acute Kidney Injury (AKI) Price Trends and Analogue Assessment
- Acute Kidney Injury (AKI) Therapies Drug Adoption/Uptake
- Acute Kidney Injury (AKI) Therapies Peak Patient Share analysis
Acute Kidney Injury Market Report Assessment
- Acute Kidney Injury (AKI) Current Treatment Practices
- Acute Kidney Injury (AKI) Unmet Needs
- Acute Kidney Injury (AKI) Clinical Development Analysis
- Acute Kidney Injury (AKI) Emerging Drugs Product Profiles
- Acute Kidney Injury (AKI) Market Attractiveness
- Acute Kidney Injury (AKI) Qualitative Analysis (SWOT and Conjoint Analysis)
FAQ's related to the Acute Kidney Injury Market Report
Acute Kidney Injury Market Insights
- What was the Acute Kidney Injury (AKI) market size, the market size by therapies, market share (%) distribution in 2025, and what would it look like by 2036? What are the contributing factors for this growth?
- What are the anticipated pricing variations among different geographies for the emerging therapies in the future?
- What can be the future treatment paradigm of Acute Kidney Injury (AKI)?
- What are the disease risks, burdens, and unmet needs of Acute Kidney Injury (AKI)? What will be the growth opportunities across the 7MM concerning the patient population with Acute Kidney Injury (AKI)?
- Who is the major future competitor in the market, and how will the competitors affect their market share?
- What are the current options for the treatment of Acute Kidney Injury (AKI)? What are the current guidelines for treating Acute Kidney Injury (AKI) in the US, Europe, and Japan?
Reasons to Buy the Acute Kidney Injury Market Forecast Report
- The Acute Kidney Injury Market report will help in developing business strategies by understanding the latest trends and changing treatment dynamics driving the Acute Kidney Injury (AKI) market.
- Bottom up forecasting builds from the affected population to product forecasts, delivering a robust, data driven approach ideal for new therapies and novel classes.
- Insights on patient burden/disease incidence, evolution in diagnosis, and factors contributing to the change in the epidemiology of the disease during the forecast years.
- Understand the existing market opportunities in varying geographies and the growth potential over the coming years.
- Identifying strong upcoming players in the market will help devise strategies to help get ahead of competitors.
- Detailed analysis and ranking of class-wise potential current and emerging therapies under the conjoint analysis section to provide visibility around leading classes.
- To understand KOLs’ perspectives on the accessibility, acceptability, and compliance-related challenges of existing treatment to overcome barriers in the future.
- Detailed insights into the unmet needs of the existing market so that the upcoming players can strengthen their development and launch strategy.
- This Artificial Intelligence (AI) enabled report summarizes and simplifies complex datasets within the report into clear, actionable insights for stakeholders, investors, and healthcare providers, enabling faster, data driven decisions.
-market-report.png&w=256&q=75)


.png&w=3840&q=75)
