Acute Ocular Pain Market Outlook
- In 2023, the total Acute Ocular Pain market size in the US was approximately USD 260 million.
- The US FDA-approved Acute Ocular Pain therapies encompass DEXTENZA, a preservative-free intracanalicular insert placed within the lower lacrimal punctum and canaliculus of the eyelid to address ocular conditions. INVELTYS, combining loteprednol etabonate and KPI-121 in a nanoparticle corticosteroid formulation, targets postoperative ocular inflammation and pain. BROMSITE, utilizing a polymer-based formulation, enhances solubility, absorption, bioavailability, and duration of action compared to traditional topical treatments, among various other options available.
- Acute Ocular Pain medications, such as corticosteroids, carry a risk of adverse effects with prolonged use. These side effects may include elevated intraocular pressure, cataract formation, delayed wound healing, or increased susceptibility to infections. Thus, balancing the benefits of pain relief with the potential risks of systemic and ocular side effects is essential in treatment decision-making.
- As the population ages, the occurrence of conditions such as dry eye syndrome, glaucoma, and age-related macular degeneration increases. These conditions can cause ocular pain due to various mechanisms, including inflammation, elevated intraocular pressure, and corneal abrasions. Moreover, various additional elements contribute to the rising incidence of acute ocular pain in the 7MM. Exposure to environmental pollutants, allergens, and irritants has the potential to induce allergic reactions, chemical burns, or sensations of foreign bodies in the eyes, ultimately resulting in acute discomfort and pain.
- Current treatment for acute ocular pain is a multifaceted approach that may include the use of topical analgesics, anti-infective agents, anti-allergic, corticosteroids, artificial tears, and others. However, many topical treatments for acute ocular pain, provide temporary relief but may require frequent application due to their short duration of action. This inconvenience in patients may lead to decreased adherence to treatment regimens.
- Due to limited epidemiological data, there are gaps in the understanding of acute ocular pain in patients. This lack of comprehensive data makes it difficult to assess the full scope of the problem and allocate resources effectively. Furthermore, the lack of rapid and reliable diagnostic tools for identifying the underlying causes of acute ocular pain contributes to unmet needs in treatment.
- Several Acute Ocular Pain companies are working robustly on many new Acute Ocular Pain therapies, such as OCS-01 (Oculis SA), SURF (Surface Ophthalmics), APP13007 (Formosa Pharmaceuticals), SVT-15473 (Salvat Laboratories), SDN-037 (Visiox Pharma), DEXYCU (EyePoint Pharmaceuticals).
Factors affecting Acute Ocular Pain Market Growth
-
Rising Prevalence of Eye Disorders
Increasing cases of ocular infections, injuries, and post-surgical complications drive demand for acute ocular pain management.
-
Growing Awareness of Eye Health
Public education on eye care and early treatment encourages timely intervention.
-
Advancements in Pharmacological Therapies
Development of novel analgesics, anti-inflammatories, and topical formulations improves treatment outcomes.
-
Increasing Ophthalmic Surgeries
Growth in procedures like cataract, LASIK, and corneal surgeries increases postoperative pain management needs.
-
Technological Innovations in Drug Delivery
Eye drops, gels, and sustained-release ocular devices enhance efficacy and patient compliance.
-
Rising Geriatric Population
Elderly patients are more susceptible to ocular conditions, increasing the demand for acute ocular pain therapies.
-
Expansion of Eye Care Infrastructure
Better access to ophthalmology clinics and specialized care centers boosts market growth.
-
Integration of Digital Health Solutions
Teleophthalmology and online consultations improve accessibility and follow-up care.
-
Government & NGO Initiatives
Eye care programs and funding for ocular research promote awareness and treatment adoption.
-
Strategic Collaborations & Clinical Research
Partnerships between pharmaceutical companies and research institutes accelerate drug development for ocular pain management.
DelveInsight’s “Acute Ocular Pain Market Insights, Epidemiology, and Market Forecast – 2034” report delivers an in-depth understanding of acute ocular pain, historical and forecasted epidemiology, as well as the acute ocular pain market trends in the United States, EU4 (Germany, France, Italy, and Spain) and the United Kingdom, and Japan.
The acute ocular pain market report provides current treatment practices, emerging drugs, market share of individual therapies, and current and forecasted 7MM acute ocular pain market size from 2020 to 2034. The Acute Ocular Pain treatment market report also covers acute ocular pain treatment practices/algorithms and unmet medical needs to curate the best opportunities and assess the acute ocular pain market’s potential.
|
Study Period |
2020–2034 |
|
Forecast Period |
2024–2034 |
|
Geographies Covered |
US, EU4 (Germany, France, Italy, and Spain) the UK, and Japan |
|
Acute Ocular Pain Epidemiology |
|
|
Acute Ocular Pain Market |
|
|
Acute Ocular Pain Market Analysis |
|
|
Acute Ocular Pain Companies |
|
|
Acute Ocular Pain Market Barrier |
The considerable expenses associated with medications, diagnostic procedures, and potential long-term management options for acute ocular pain can present substantial hurdles to patients' ability to obtain and adhere to treatments. Furthermore, the absence of swift and dependable diagnostic tools for determining the root causes of acute ocular pain exacerbates the existing gaps in treatment. |
Acute Ocular Pain Treatment Market
Acute ocular pain overview
The International Association for the Study of Pain (IASP) defines pain as “an unpleasant sensory and emotional experience associated with actual or potential tissue damage,” and pain is commonly stratified into two broad groups—nociceptive and neuropathic pain. Nociceptive pain refers to the direct activation of nociceptors due to actual or threatened damage to non-neural tissue and implies an intact somatosensory nervous system. This is typically associated with transient pain following injury, surgery, or infection in the eye. In contrast, neuropathic pain is caused by a lesion or disease of the intrinsic somatosensory nervous system, often in the absence of other tissue injuries.
Ocular pain is a myriad of symptoms ranging from acute sharp pain in the eye to mild discomfort or itching in the eyes. As the spectrum of symptom presentation varies with eye pain, the cause of these pains may vary from straightforward refractive errors to potentially sight-threatening disorders like glaucoma and Acute Ocular Pain. Most ophthalmologic conditions producing ocular pains are associated with symptoms and signs like red eye, photophobia, vision loss, or diplopia. Sometimes it poses a challenge to physicians when the pain is associated with a quiet or “white eye.”
Continued in the report…..
Acute ocular pain diagnosis
Diagnosis of acute ocular pain is based on previous medical and medication history and physical examination of the eye, which is then confirmed by tests like computed tomography (CT), magnetic resonance imaging (MRI), histopathologic examination for the detection of cancers, and culture of the eye to check for a bacterial infection.
Continued in the report…..
Acute ocular pain treatment
Treatment for acute ocular pain aims to alleviate symptoms in the shortest period necessary while ensuring the patient's needs are met. A variety of management options include non-pharmacologic treatments, topical ocular cycloplegic agents, topical ocular nonsteroidal anti-inflammatory agents (NSAIDs), and oral pharmaceutical agents. The selection of oral analgesics, whether non-opioid medications like NSAIDs and acetaminophen or opioid medications is carefully tailored to minimize the duration of treatment and potential side effects, with a focus on addressing the underlying cause of the pain while ensuring patient safety and comfort.
Continued in the report…..
Acute Ocular Pain Epidemiology Analysis
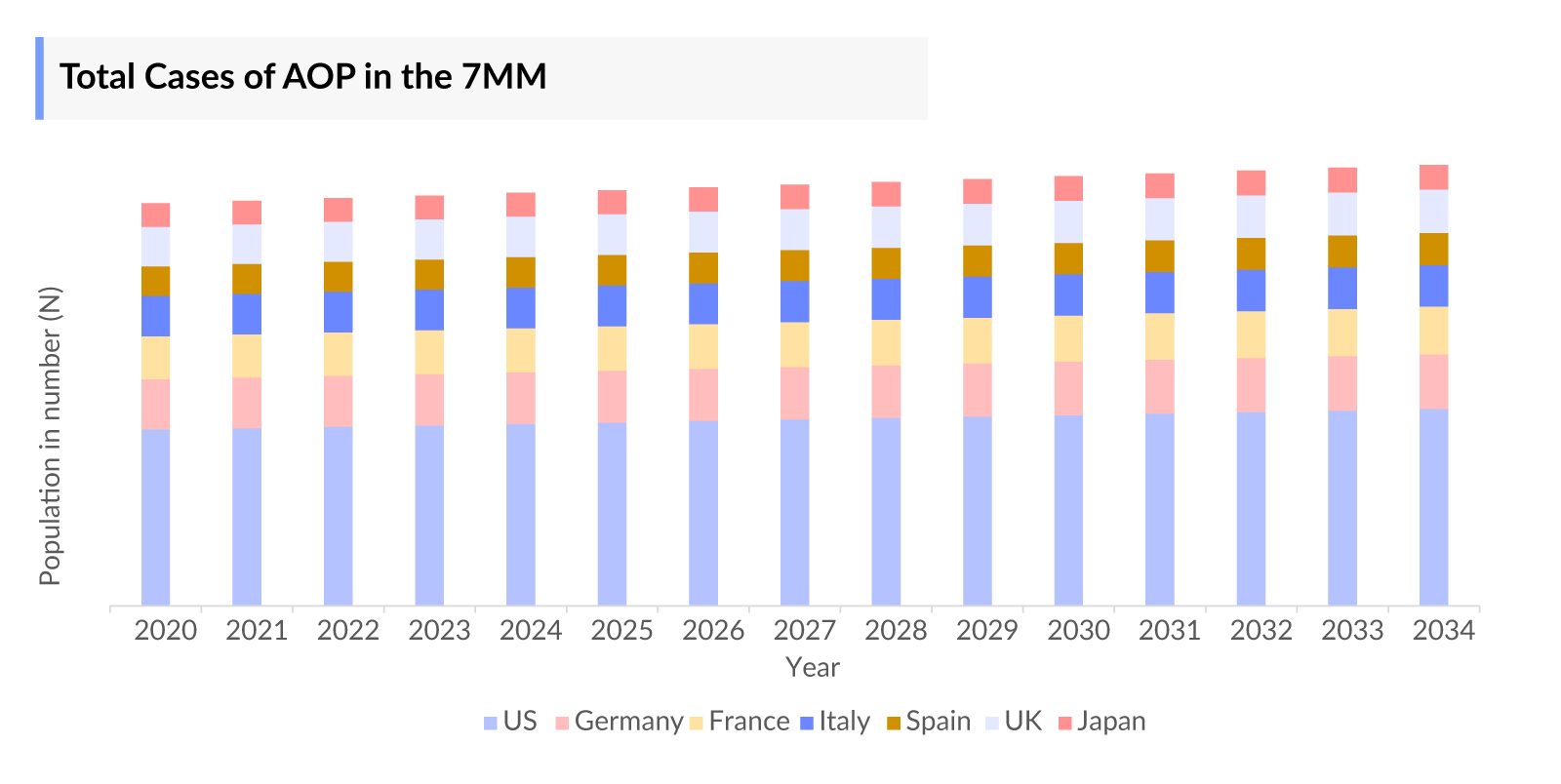
As the Acute Ocular Pain market is derived using a patient-based model, the acute ocular pain epidemiology chapter in the report provides historical as well as forecasted epidemiology segmented by number of cases of major eye disorders, and number of cases of acute ocular pain in major eye disorders in the 7MM covering the United States, EU4 countries (Germany, France, Italy, and Spain) and the United Kingdom, and Japan from 2020 to 2034.
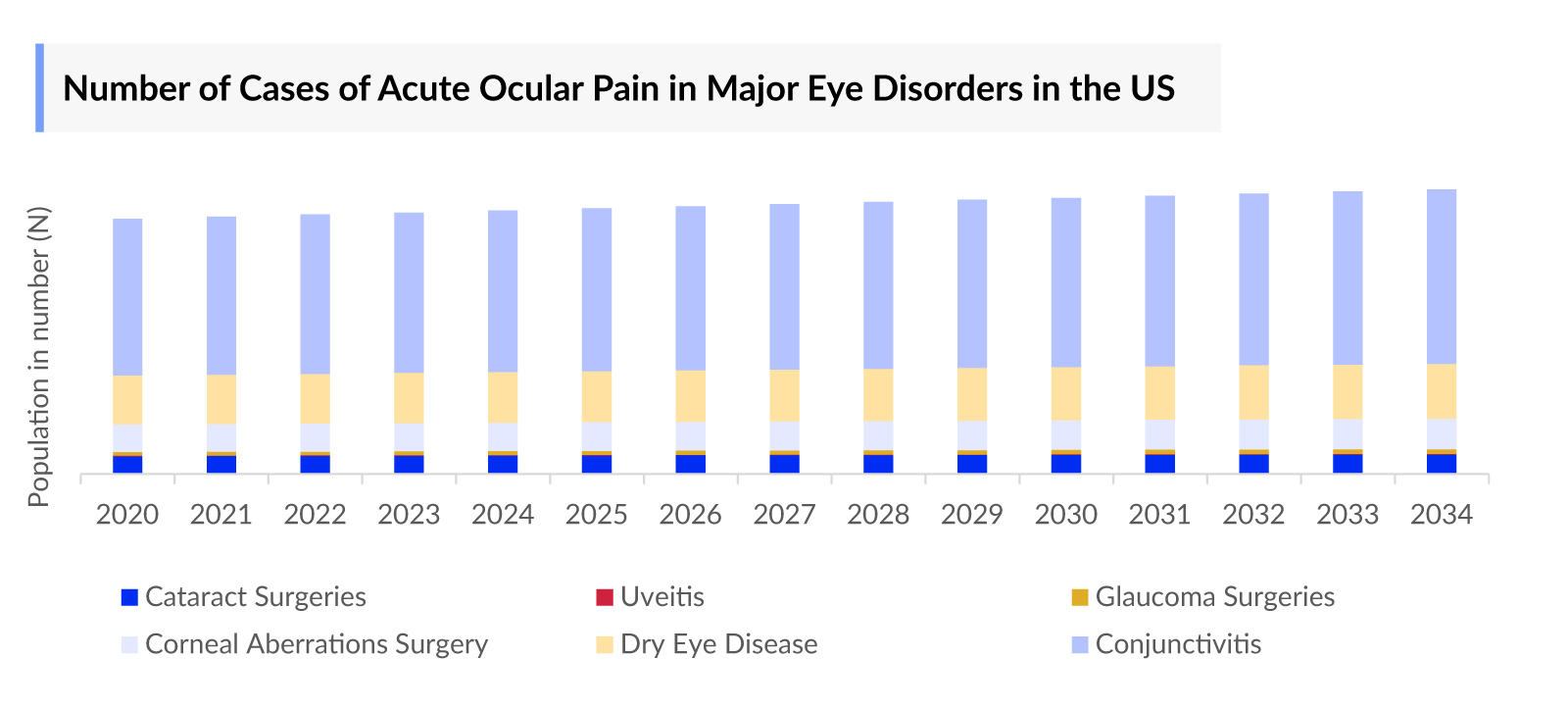
- As per DelveInsight’s analysis, in 2023, the US accounted for nearly 4 million cases of acute ocular pain in major eye disorders. These cases are anticipated to rise during the forecasted period (2024-2034) primarily due to increased awareness among healthcare providers and the public regarding acute ocular pain that will enhance its identification and diagnosis.
- In 2023, it was estimated that there were approximately 2 million cases of conjunctivitis associated with acute operative pain, making it the most prevalent condition among other disorders. This was followed by dry eye disease and corneal aberrations surgery, with nearly 1 million and 733 thousand cases, respectively, in the US. These cases are anticipated to increase by 2034 attributed to the aging population, characterized by a higher prevalence of age-related eye conditions like cataracts, macular degeneration, and glaucoma. These conditions are known to be significant contributors to acute ocular pain.
- Among the EU4 and the UK, Germany had the highest number of cases of acute ocular pain in major eye disorders, estimated to be nearly 1 million. Followed by France and Italy with approximately 830 thousand and 803 thousand cases, respectively, indicating substantial occurrences of acute ocular pain across these countries. According to DelveInsight’s analysis, these cases are expected to increase by 2034 driven by the exposure to environmental pollutants, allergens, and irritants that may exacerbate existing eye conditions or lead to new ones, resulting in acute ocular pain.
- In 2023, Japan accounted for nearly 870 thousand cases of acute ocular pain in major eye disorders. These cases are expected to increase by 2034 with the growing prevalence of an aging population in Japan, which is at a heightened risk of developing acute ocular pain.
Acute Ocular Pain Drug Analysis
The drug chapter segment of the acute ocular pain drugs market report encloses a detailed analysis of marketed Acute Ocular Pain drugs and late-stage (Phase III and Phase II) pipeline drugs. It also helps understand the acute ocular pain clinical trial details, expressive pharmacological action, agreements and collaborations, approval and patent details, advantages and disadvantages of each included drug, and the latest news and press releases.
Marketed Acute Ocular Pain Drugs
-
DEXTENZA (dexamethasone ophthalmic insert): Ocular Therapeutix
DEXTENZA is a preservative-free intracanalicular insert that is inserted in the lower lacrimal punctum, a natural opening in the eyelid, and into the canaliculus. It is designed to deliver a tapered dose of steroid (dexamethasone) to the ocular surface for up to 30 days. It acts as a corticosteroid that suppresses inflammation by inhibiting multiple inflammatory cytokines resulting in decreased edema, fibrin deposition, capillary leakage, and migration of inflammatory cells. The US FDA approved DEXTENZA in June 2019 for treating ocular inflammation following ophthalmic surgery as an additional indication. A single DEXTENZA releases a 0.4 mg dose of dexamethasone for up to 30 days following insertion.
-
INVELTYS (loteprednol etabonate ophthalmic suspension/KPI-121): Kala Pharmaceuticals
INVELTYS (loteprednol etabonate/KPI-121) is a nanoparticle ocular corticosteroid formulation for treating postoperative ocular inflammation and pain. INVELTYS, as with other ophthalmic corticosteroids, is contraindicated in most cornea and conjunctiva viral diseases, including epithelial Herpes simplex keratitis (dendritic keratitis), vaccinia, and varicella, and also in mycobacterial infection of the eye and fungal diseases of ocular structures.
Note: Further marketed drugs and their details will be provided in the report…
Emerging Acute Ocular Pain Drugs
-
OCS-01: Oculis
OCS-01 is a novel formulation of dexamethasone using Oculis’ proprietary Solubilizing Nanoparticle (SNP) technology. Additionally, SNP technology increases the solubility and residence time of the active drug on the eye’s surface, aiming to deliver an improved and clinically effective anti-inflammatory effect while reducing the frequency of topical administration. The drug candidate is currently in Phase III development in the US for evaluating the efficacy and safety of OCS-01 Eye drops in subjects following cataract surgery and has completed a Phase II trial. Additionally, OCS-01 is also being investigated for diabetic macular edema and Acute Ocular Pain-related cystoid macular edema.
-
APP13007: Formosa Pharmaceuticals
APP13007 is a nanoparticle formulation of the corticosteroid clobetasol in development for treating postoperative inflammation of the eye. Activus initially developed APP13007 by applying its patented proprietary Activus Pure Nanoparticle Technology (APNT) to the corticosteroid clobetasol propionate to create a novel nanoparticle formulation for treating postoperative inflammation of the eye. Formosa Pharmaceuticals, and AimMax Therapeutics, (United States) reported successful top-line results from CPN-302, the second of the two pivotal Phase III clinical studies of APP13007.
Formosa Pharma has entered licensing agreements with Eyenovia for the US and territories and China Grand Pharmaceutical for China, Hong Kong, and Macau. Moreover, APP13002 is in the IND-enablement stages and is available for licensing or co-development either regionally or globally.
Note: Further emerging therapies and their detailed assessment will be provided in the final report.
Acute Ocular Pain Drug Class Insights
Acute ocular pain treatment typically involves a combination of medications to address specific causes and symptoms. Nonsteroidal anti-inflammatory drugs (NSAIDs) like ketorolac or diclofenac are commonly used for their effective pain relief properties, although they carry the risk of adverse corneal reactions such as epithelial defects and impaired healing rates. Corticosteroids, while not typically used for acute pain relief, may be prescribed in certain cases to reduce inflammation associated with conditions like Acute Ocular Pain or allergic reactions, although prolonged use can lead to side effects like cataract formation and elevated intraocular pressure. Antibiotic eye drops or ointments may also be part of the treatment regimen if the ocular pain is caused by an infection, such as bacterial conjunctivitis or corneal ulcer. Fluoroquinolones (e.g., moxifloxacin, ciprofloxacin) and aminoglycosides (e.g., gentamicin) are common classes of antibiotics used for these purposes. Moreover, antihistamines such as olopatadine and ketotifen are utilized to manage ocular pain linked with allergic conjunctivitis or allergic reactions. These agents play a crucial role in alleviating itching, redness, and discomfort in the eyes.
Acute Ocular Pain Market Outlook
Therapeutic options for managing acute ocular pain include non-pharmacological treatment, such as bandage contact lenses, topical ocular cycloplegic agents, topical ocular nonsteroidal anti-inflammatory agents (NSAIDs), and oral pharmaceutical agents. For treating patients with acute pain, there are generally two categories of oral medications: non-opioid medications and opioid medications. Non-opioid oral medications include prescription or OTC NSAIDs and acetaminophen. Pain associated with surgery, injury, infection, or inflammation at the front of the eye is typically treated with a topical steroid, topical NSAID, systemic NSAID, lubricant ointment, gel or drops, bandage contact lens, or a few doses of oral opiate or topical anesthetic.
Recently, several Acute Ocular Pain drugs like DEXTENZA, INVELTYS, BROMSITE, OMIDRIA, NEVANAC, PROLENSA, and LOTEMAX have gained approval for managing ocular inflammation and pain post-ophthalmic or cataract surgery. DEXTENZA stands out as the pioneering FDA-approved intracanalicular insert, offering a novel administration route that bypasses the need for eye drops. It's a preservative-free, resorbable hydrogel insert delivering 0.4 mg of dexamethasone, effectively addressing post-surgical ocular inflammation and pain for up to 30 days with just one application. Despite their pain-relieving properties, these drugs are associated with well-known adverse effects such as cataract development and increased intraocular pressure or glaucoma.
However, several Acute Ocular Pain companies are working robustly on many new therapies, including OCS-01 (Oculis SA), APP13007 (Formosa Pharmaceuticals), SURF (Surface Ophthalmics), SVT-15473 (Salvat Laboratories), SDN-037 (Visiox Pharma), DEXYCU (EyePoint Pharmaceuticals), and others anticipated to positively influence the Acute Ocular Pain treatment market size during the forecast period [2024-2034].
According to DelveInsight, the overall dynamics of the acute ocular pain market are anticipated to change during the forecast period (2024-2034) owing to the expected launch of emerging therapies, increasing awareness among healthcare providers and patients, coupled with technological advancements in diagnostic techniques, is expected to contribute to a growing pool of diagnosed cases.
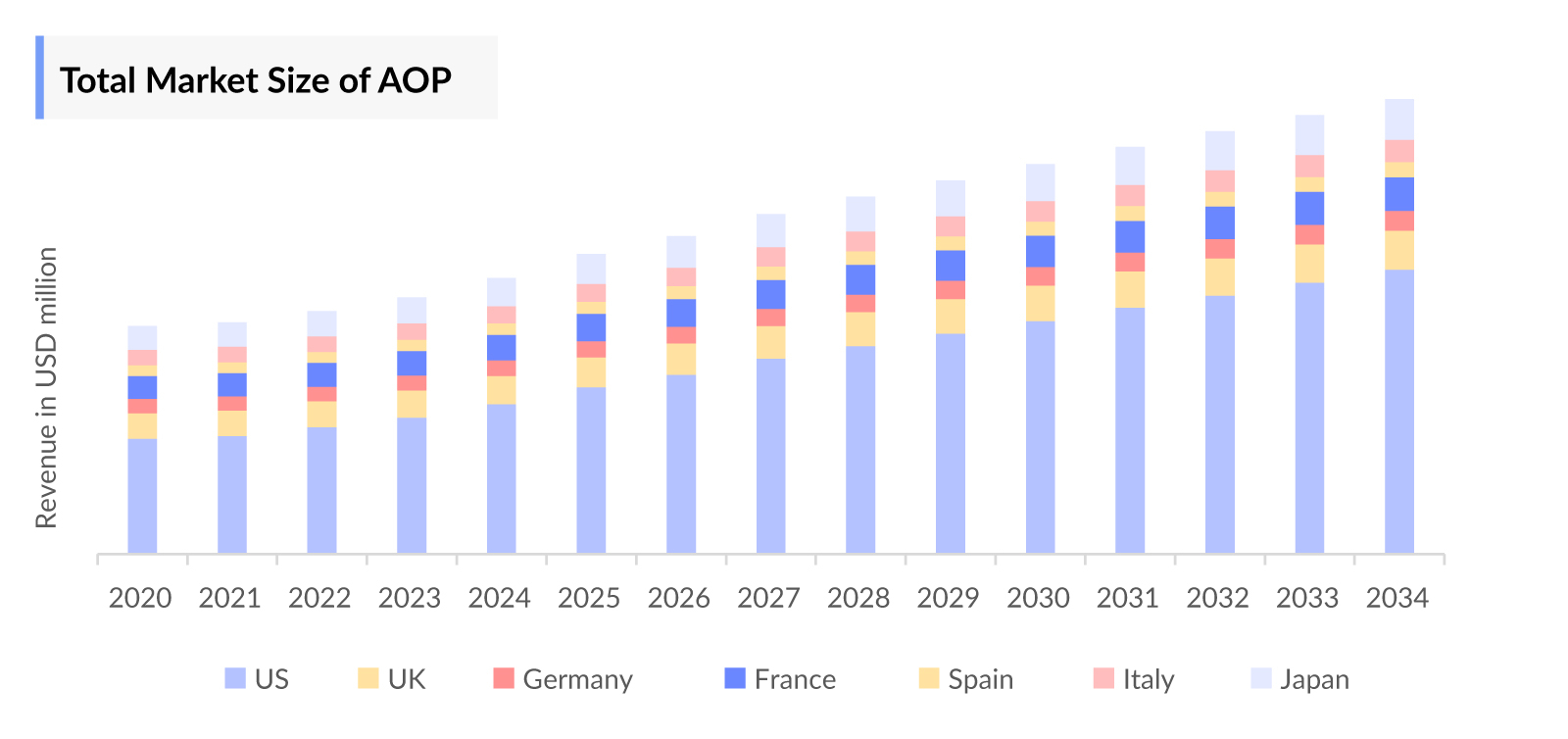
- In 2023, the total Acute Ocular Pain market size in the US was approximately USD 260 million.
- Among the EU4 and the UK, Germany holds the highest Acute Ocular Pain market size of around USD 19 million, followed by France and Italy with approximately USD 16 million and USD 15 million, respectively. Acute Ocular Pain treatment market numbers are expected to change during the forecast period (2024-2034), driven by the ongoing research and development efforts that may lead to the discovery of more effective medications and therapies for treating acute ocular pain.
Continued in report…
Acute Ocular Pain Drug Uptake
This section focuses on the uptake rate of potential Acute Ocular Pain drugs expected to be launched in the market during 2020–2034. For example, Oculis OCS-01, a novel formulation of dexamethasone based on a unique Oculis proprietary SNP technology, enhances drug solubility in the aqueous tear fluid and, thus, enhances bioavailability in eye tissues. Not only, does OCS-01 promise to be effective, but is a once-daily and preservative-free treatment to manage inflammation and pain.
Further detailed analysis of emerging therapies' drug uptake in the report…
Acute Ocular Pain Pipeline Drugs Market
The Acute Ocular Pain treatment market report provides insights into different therapeutic candidates in Phase III, Phase II, and Phase I. It also analyzes key acute ocular pain companies involved in developing targeted therapeutics.
Acute Ocular Pain Clinical Trial activities
The Acute Ocular Pain drugs market report covers information on collaborations, acquisitions and mergers, licensing, and patent details for emerging Acute Ocular Pain therapies.
Latest KOL Views on Acute Ocular Pain
To keep up with current Acute Ocular Pain market trends, we take KOLs and SMEs’ opinions working in the domain through primary research to fill the data gaps and validate our secondary research. Industry Experts contacted for insights on the acute ocular pain evolving treatment landscape, patient reliance on conventional therapies, patient therapy switching acceptability, and drug uptake, along with challenges related to accessibility, including Medical/scientific writers, Medical Professionals, Professors, Directors, and Others.
DelveInsight’s analysts connected with 50+ KOLs to gather insights; however, interviews were conducted with 15+ KOLs in the 7MM. Centers like the University of California, Johns Hopkins University School of Medicine, University Medical Center Schleswig Holstein, Kiel, Institut de la Vision, Sapienza University of Rome, University of Birmingham, Hospital Virgen De La Salud, were contacted. Their opinion helps understand and validate current and emerging therapy treatment patterns or acute ocular pain market trends. This will support the clients in potential upcoming novel treatments by identifying the overall scenario of the Acute Ocular Pain market and the unmet needs.
|
Region |
KOL Views |
|
The US |
No, patients rarely feel any sort of ocular pain before the surgery. Around 20% of patients suffer from pain after Glaucoma surgery. This percentage is specifically for patients suffering from Post-surgical pain. |
|
Italy |
In the case of cataract surgeries around 1-2% of patients suffer from postoperative Acute Ocular pain whereas the percentage is high in the case of Glaucoma surgeries (i.e., Around 8-10%). In the case of Dry eye disease and Acute Ocular Pain around 90% and 50% of patients suffer from pain respectively during the treatment. |
|
The UK |
Very few patients suffer from Ocular pain before the surgery. Most patients suffer from pain after the surgery. |
Physician’s View
According to physicians, a primary challenge in acute ocular pain treatment lies in the identification of the underlying cause. Many ocular conditions present with similar symptoms such as pain, redness, and irritation, making it challenging to differentiate between them without a comprehensive evaluation. Acute ocular pain can arise from a wide range of etiologies, including infections, inflammation, trauma, and systemic conditions. Determining the specific cause requires careful history-taking, clinical examination, and sometimes specialized diagnostic tests. Another challenge is that delayed or incorrect diagnosis can lead to complications or exacerbation of the underlying condition. Therefore, clinicians must accurately identify the cause of ocular pain to provide appropriate and timely treatment.
Qualitative Analysis
We perform Qualitative and market Intelligence analysis using various approaches, such as SWOT analysis and Attribute Analysis. In the SWOT analysis, strengths, weaknesses, opportunities, and threats in terms of disease diagnosis, patient awareness, patient burden, competitive landscape, cost-effectiveness, and geographical accessibility of therapies are provided. These pointers are based on the Analyst’s discretion and assessment of the patient burden, cost analysis, and existing and evolving treatment landscape.
Attribute Analysis analyzes multiple emerging Acute Ocular Pain therapies based on relevant attributes such as safety, efficacy, frequency of administration, route of administration, and order of entry. Scoring is given based on these parameters to analyze the effectiveness of therapy.
In efficacy, the trial’s primary and secondary outcome measures are evaluated; for instance, in Acute Ocular Pain trials, one of the most important primary outcome measures is complete eschar removal.
Further, the therapies’ safety is evaluated wherein the acceptability, tolerability, and adverse events are majorly observed, and it sets a clear understanding of the side effects posed by the drug in the trials. In addition, the scoring is also based on the route of administration, order of entry and designation, probability of success, and the addressable patient pool for each therapy. According to these parameters, the final weightage score and the ranking of the emerging therapies are decided.
Acute Ocular Pain Market Access and Reimbursement
The high cost of therapies for the treatment is a major factor restraining the growth of the Acute Ocular Pain drugs market. Because of the high cost, the economic burden is increasing, leading the patient to escape from proper treatment.
The reimbursement challenges related to medical care and treatment for individuals with acute ocular pain can be significant as they often require specialized medical attention, covering the costs of diagnosis, treatment, and ongoing care. Health insurance plans may not fully cover limited coverage of some medical treatments, and therapies specific to acute ocular pain. This can result in high out-of-pocket expenses for families seeking the best care for their loved ones. Moreover, it requires specialized care from healthcare providers with expertise. Finding and accessing such specialists may be challenging, and the associated costs may not always be fully reimbursed by insurance.
Further details will be provided in the report.
Scope of the Acute Ocular Pain Market Report
- The Acute Ocular Pain market report covers a segment of key events, an executive summary, descriptive overview of acute ocular pain, explaining its causes, signs and symptoms, pathogenesis, and currently available Acute Ocular Pain therapies.
- Comprehensive insight has been provided into the epidemiology segments and forecasts, the future growth potential of diagnosis rate, disease progression, and treatment guidelines.
- Additionally, an all-inclusive account of the current and emerging therapies, along with the elaborative profiles of late-stage and prominent therapies, will impact the current treatment landscape.
- A detailed review of the acute ocular pain therapeutics market, historical and forecasted market size, market share by therapies, detailed assumptions, and rationale behind our approach is included in the report, covering the 7MM drug outreach.
- The Acute Ocular Pain therapeutics market report provides an edge while developing business strategies, by understanding trends, through SWOT analysis and expert insights/KOL views, patient journey, and treatment preferences that help shape and drive the 7MM acute ocular pain market.
Acute Ocular Pain Market Report Insights
- Acute Ocular Pain Patient Population
- Acute Ocular Pain Therapeutic Approaches
- Acute Ocular Pain Pipeline Analysis
- Acute Ocular Pain Market Size
- Acute Ocular Pain Market Trends
- Existing and Future Acute Ocular Pain Market Opportunity
Acute Ocular Pain Market Report Key Strengths
- 11 years Forecast
- The 7MM Coverage
- Acute Ocular Pain Epidemiology Segmentation
- Key Cross Competition
- Attribute analysis
- Acute Ocular Pain Drugs Uptake
- Key Acute Ocular Pain Market Forecast Assumptions
Acute Ocular Pain Market Report Assessment
- Current Acute Ocular Pain Treatment Practices
- Acute Ocular Pain Unmet Needs
- Acute Ocular Pain Pipeline Product Profiles
- Acute Ocular Pain Market Attractiveness
- Qualitative Analysis (SWOT and Conjoint Analysis)
Key Questions Answered in the Acute Ocular Pain Report
Acute Ocular Pain Market Insights
- What was the acute ocular pain market share (%) distribution in 2020 and what it would look like in 2034?
- What was the total Acute Ocular Pain market size by therapies, and market share (%) distribution in 2020, and what would it look like by 2034? What are the contributing factors for this growth?
- How will OCS-01, and APP13007 affect the treatment paradigm of acute ocular pain?
- How will APP13007 compete with other upcoming products?
- Which drug is going to be the largest contributor by 2034?
- What are the pricing variations among different geographies for approved and marketed Acute Ocular Pain therapies?
- How would future opportunities affect the Acute Ocular Pain market dynamics and subsequent analysis of the associated trends?
Acute Ocular Pain Epidemiology Insights
- What are the disease risks, burdens, and unmet needs of acute ocular pain? What will be the growth opportunities across the 7MM concerning the patient population with acute ocular pain?
- What is the historical and forecasted acute ocular pain patient pool in the United States, EU4 (Germany, France, Italy, and Spain) the United Kingdom, and Japan?
- Out of the above-mentioned countries, which country would have the highest diagnosed prevalent acute ocular pain population during the forecast period (2023–2034)?
- What factors are factors contributing to the growth of acute ocular pain cases?
Current Treatment Scenario, Marketed Drugs, and Emerging Therapies
- What are the current options for the treatment of acute ocular pain? What are the current guidelines for treating acute ocular pain in the US and Europe?
- How many Acute Ocular Pain companies are developing therapies for the treatment of acute ocular pain?
- How many emerging Acute Ocular Pain therapies are in the mid-stage and late stage of development for treating acute ocular pain?
- What are the recent novel therapies, targets, Acute Ocular Pain mechanisms of action, and technologies developed to overcome the limitations of existing therapies?
- What is the Acute Ocular Pain cost burden of current treatment on the patient?
- Patient acceptability in terms of preferred treatment options as per real-world scenarios?
- What are the country-specific accessibility issues of approved Acute Ocular Pain therapies?
- What is the 7MM historical and forecasted Acute Ocular Pain market?
Reasons to Buy the Acute Ocular Pain Report
- The Acute Ocular Pain treatment market report will help develop business strategies by understanding the latest trends and changing treatment dynamics driving the acute ocular pain market.
- Insights on patient burden/ Acute Ocular Pain prevalence, evolution in diagnosis, and factors contributing to the change in the epidemiology of the disease during the forecast years.
- Understand the existing Acute Ocular Pain market opportunities in varying geographies and the growth potential over the coming years.
- Distribution of historical and current Acute Ocular Pain patient share based on real-world prescription data in the US, EU4 (Germany, France, Italy, and Spain) the United Kingdom, and Japan.
- Identifying strong upcoming Acute Ocular Pain companies in the Acute Ocular Pain market will help devise strategies to help get ahead of competitors.
- Detailed analysis and ranking of class-wise potential current and emerging therapies under the conjoint analysis section to provide visibility around leading classes.
- Highlights of access and reimbursement policies for acute ocular pain, barriers to accessibility of approved therapy, and patient assistance programs.
- To understand Key Opinion Leaders’ perspectives around the accessibility, acceptability, and compliance-related challenges of existing treatment to overcome barriers in the future.
- Detailed insights on the unmet needs of the existing Acute Ocular Pain treatment market so that the upcoming Acute Ocular Pain companies can strengthen their development and launch strategy.
Related Insights:



.png&w=256&q=75)

