Breaking the Cycle of Failure in AKI Drug Development: Opportunities in a High-Burden, Underserved Market
Apr 27, 2026
Table of Contents
Summary
- AKI continues to be associated with substantial morbidity and mortality, particularly in hospitalized and ICU-based settings where patients often present with severe underlying illness.
- TERLIVAZ (terlipressin) is the only approved pharmacologic therapy in the broader AKI space, receiving US FDA approval in September 2022 for HRS-AKI, a high-mortality but relatively narrow AKI segment.
- Drug development in AKI has faced numerous interconnected obstacles. Recent clinical setbacks, including the discontinuation of investigational candidates such as RMZ-035, ANG-3777, AP214, and GDC-8265, further highlight the complexity of these challenges.
- Despite past challenges, the AKI treatment landscape is advancing through ongoing scientific and technological progress. Emerging therapies, including AstraZeneca/Alexion Pharmaceuticals’s Ravulizumab, Alloksys Life Sciences’s RESCAP, AM-Pharma/Kyowa Kirin’s Ilofotase alfa, Arch Biopartners’s LSALT peptide, Novartis’s TIN816, and AstraZeneca/Mitsubishi Tanabe Pharma’s AZD4144, are currently under evaluation in multiple high-risk clinical settings.
AKI remains one of the most complex and high-impact conditions in modern medicine, characterized by a rapid decline in kidney function and associated with significant morbidity, mortality, and healthcare costs. Despite decades of clinical research and multiple late-stage development efforts, therapeutic innovation in AKI has been limited.
Downloads
Click Here To Get the Article in PDF
Recent Articles
- Exploring the Future of Acute Kidney Injury: Market Trends and Growth Opportunities
- What Does the Future Hold for Acute Kidney Injury? A Deep Dive into the Evolving Pipeline
- Everything You Need to Know About Acute Kidney Injury
- Profound Medical and Siemens Entered Into a Collaboration; CardiAI and Carleton University Forged...
- Breakthrough Therapies Shaping the Future of Acute Kidney Injury Treatment
The current treatment paradigm continues to rely predominantly on supportive care, reflecting a clear disconnect between disease burden and the availability of targeted therapies. This gap positions AKI as a high-risk yet high-reward opportunity, where successful innovation could deliver substantial clinical and economic value.
Disease Burden and Unmet Need
The burden of AKI is increasing globally, driven by rising hospitalizations, aging populations, and a higher prevalence of comorbidities. Incidence remains particularly elevated in critical care settings such as sepsis and major surgical procedures, where AKI significantly worsens outcomes and healthcare resource utilization. In 2025, AKI incident cases across the 7MM were estimated at ~8.7 million, with continued growth projected at a CAGR of 1.5% through 2036. High-burden subpopulations include sepsis-associated AKI (~875,000 cases) and cardiac surgery-associated AKI (~274,000 cases), both of which lack approved targeted therapies. Despite the substantial clinical burden, treatment options remain limited, with Terlipressin (TERLIVAZ) approved only for Hepatorenal Syndrome (HRS)–AKI, leaving a significant unmet need for disease-specific interventions.
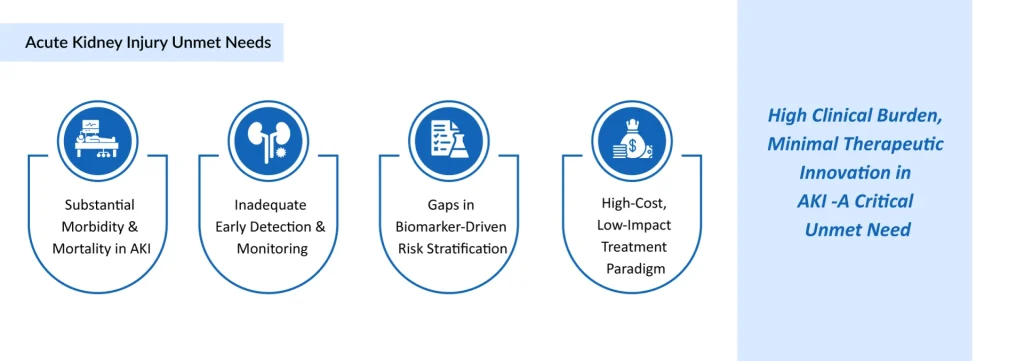
The AKI landscape is characterized by a significant imbalance between high clinical burden and limited therapeutic innovation. Persistent challenges, including substantial morbidity and mortality, delayed diagnosis, gaps in biomarker-driven risk stratification, and a high-cost yet low-impact treatment paradigm, continue to hinder effective disease management. Collectively, these factors highlight a critical and ongoing unmet need for targeted, innovative therapies in AKI.
Terlipressin (TERLIVAZ) Uptake and Evolving HRS–AKI Definition
HRS–AKI represents a severe and acute complication of cirrhosis, previously classified as Type I HRS (HRS-1). Recent updates in nomenclature and clinical criteria have shifted toward the term HRS-AKI, aligning it with broader AKI definitions to enable earlier and more dynamic diagnosis.
Terlipressin, approved by the US FDA in September 2022, remains the only pharmacological therapy for this high-mortality but relatively narrow AKI subset. Despite a sizable eligible population (~179,000 patients in 2025), its commercial uptake has been modest, with sales of USD 15.6 million in 2023, USD 24.4 million in 2024, and approximately USD 31.5 million in 2025. The therapy is administered at 0.85 mg every 6 h, with a WAC of ~USD 950 per dose, resulting in a total treatment cost of around USD 19,000 for a median 5-day course. While Terlipressin offers a targeted treatment option, uptake has been limited by factors such as high cost, monitoring requirements, and the evolving diagnostic framework, which continues to influence patient identification and treatment patterns in HRS-AKI.
Why Drug Development has Failed in AKI
Drug development in AKI has been hindered by multiple interrelated challenges. The disease is highly heterogeneous, encompassing ischemic, inflammatory, and septic etiologies, which creates diverse patient populations and dilutes treatment effects in clinical trials. Delayed diagnosis, driven by reliance on traditional markers such as serum creatinine, often results in intervention after significant kidney damage has already occurred, limiting therapeutic efficacy. Additionally, conventional clinical endpoints, such as changes in creatinine levels or dialysis requirement, may not adequately capture early or meaningful clinical benefits, reducing the likelihood of demonstrating efficacy. Translational limitations further complicate development, as preclinical models frequently fail to reflect the complexity of human AKI, particularly in critically ill patients.
Recent clinical failures, including discontinuation of investigational therapies such as RMZ-035, ANG-3777, AP214, and GDC-8265, further underscore these challenges. However, these setbacks provide important insights, highlighting the need for biomarker-guided patient selection, earlier therapeutic intervention, and the use of more sensitive and composite endpoints. Collectively, these learnings are reshaping the design and execution of AKI clinical trials.
Targeted Clinical Development Focus on AKI
Clinical development of AKI is increasingly focused on specific, high-risk AKI subpopulations rather than broad, heterogeneous patient groups. Targeting these subsets improves patient stratification, increases the likelihood of demonstrating efficacy, and enhances regulatory success.
- Cardiac surgery-associated AKI (CSA-AKI): A well-defined setting with a predictable onset, enabling better trial design and endpoint assessment.
- Sepsis-associated AKI: Represents a high-burden population with significant unmet need, though biologically complex.
- Contrast-induced AKI: Offers a controlled clinical setting with identifiable triggers, supporting preventive and early intervention strategies.
Current Treatment Landscape: A Supportive Care Ceiling
AKI management remains largely unchanged, centered on supportive measures such as fluid optimization, hemodynamic stabilization, and avoidance of nephrotoxic agents. While essential, these interventions do not directly address the underlying pathophysiology of kidney injury. Renal replacement therapy remains a critical intervention in severe cases, but it is resource-intensive and does not alter disease progression. This “supportive care ceiling” highlights the urgent need for therapies capable of preventing injury, halting progression, or promoting renal recovery.
Despite historical challenges, the AKI treatment landscape is evolving with scientific and technological advances. Future success in AKI will likely depend less on broad-spectrum therapies and more on precision targeting of specific injury pathways in defined patient subsets.
Pipeline Momentum: Early Signs of a Turnaround
The AKI pipeline is gradually shifting toward targeted and mechanism-based approaches. Emerging therapies such as Ravulizumab (AstraZeneca/Alexion Pharmaceuticals), RESCAP (Alloksys Life Sciences), Ilofotase alfa (AM-Pharma/Kyowa Kirin), LSALT peptide (Arch Biopartners), TIN816 (Novartis), and AZD4144 (AstraZeneca/Mitsubishi Tanabe Pharma) are being evaluated across high-risk clinical settings.

While clinical success remains to be proven, these assets reflect a broader strategic shift.
- From symptomatic management to pathway-specific intervention
- From broad populations to stratified patient cohorts
- From late-stage intervention to earlier therapeutic targeting
This shift reflects cautious yet meaningful progress toward a more innovation-driven AKI landscape.
High Risk, High Reward: The Strategic Case for Investment
AKI represents a classic paradox in drug development. A condition with high prevalence, high mortality, and high economic burden, yet limited therapeutic success. This has historically discouraged investment. However, the landscape is shifting. Advances in biology, diagnostics, and trial design are gradually de-risking development pathways.
For industry stakeholders, AKI offers
- Large addressable patient populations: AKI affects millions of patients globally, particularly in hospital and critical care settings, offering a broad and scalable market opportunity for new therapies.
- Significant unmet clinical need: The absence of approved targeted treatments for most AKI types highlights a major gap in care, creating strong demand for effective disease-modifying therapies.
- Potential for premium, high-value therapies: Given the acute severity, high mortality risk, and hospitalization burden, innovative therapies that improve outcomes can command premium pricing and strong payer support.
A successful therapy would not only transform patient outcomes but also redefine standards of care and unlock substantial market value.
Conclusion
AKI remains one of the most challenging areas in modern medicine, defined by high clinical burden, limited therapeutic innovation, and repeated setbacks in drug development. However, the landscape is beginning to shift. Advances in disease understanding, biomarker development, and trial design are enabling a more targeted and precision-driven approach to therapy development. While historical failures have underscored the complexity of AKI, they have also provided critical insights that are reshaping future strategies. The increasing focus on well-defined patient subsets, earlier intervention, and mechanism-based therapies signals a transition toward a more rational and potentially successful development paradigm.
For industry stakeholders, AKI represents a compelling high-risk, high-reward opportunity. Bridging the gap between disease burden and therapeutic innovation has the potential not only to significantly improve patient outcomes but also to unlock substantial clinical and commercial value, ultimately redefining the standard of care in AKI.

Downloads
Article in PDF
Recent Articles
- Profound Medical and Siemens Entered Into a Collaboration; CardiAI and Carleton University Forged...
- What Does the Future Hold for Acute Kidney Injury? A Deep Dive into the Evolving Pipeline
- Exploring the Future of Acute Kidney Injury: Market Trends and Growth Opportunities
- Breakthrough Therapies Shaping the Future of Acute Kidney Injury Treatment
- Everything You Need to Know About Acute Kidney Injury