Spinal Fusion Devices in Healthcare: Stabilizing the Backbone of Modern Orthopedics
Apr 29, 2026
Table of Contents
Spinal disorders, ranging from degenerative disc disease to scoliosis, are a leading cause of disability worldwide. As populations age and lifestyles become increasingly sedentary, the burden of chronic back pain continues to rise. In this evolving healthcare landscape, spinal fusion devices have emerged as a cornerstone solution, enabling surgeons to restore stability, alleviate pain, and improve patient mobility.
What Are Spinal Fusion Devices?
Spinal fusion devices are a class of orthopedic implants and biomaterials used to permanently join two or more vertebral segments of the spine. The primary clinical goal is to eliminate painful motion at a diseased or unstable spinal level, whether caused by degenerative disc disease, spondylolisthesis, spinal stenosis, trauma, deformity (such as scoliosis), or post-tumor reconstruction.
Downloads
Click Here To Get the Article in PDF
Recent Articles
- Olympus’s Next Generation Electrosurgical Generator; Biocomposites Acquires Artoss GmbH; Artelon’...
- Neuspera Medical’s Nuvella System; Abbott Presented the Data for FreeStyle Libre 2 System; ZEISS ...
- OncoHost Granted New York State Lab Permit, Enabling PROphetNSCLC® Nationwide Access; Onkos Surgi...
- Alcon’s 510(k) Clearance From the FDA; Globus Medical Received FDA Clearance; UK Pioneers F...
- HR Pharmaceuticals Announced Collaboration with Poiesis Medical; Enovis Acquired Lima Corporate; ...
The principle behind fusion is straightforward: immobilize the target vertebrae so that bone grows across the disc space or facet joint over time, creating a solid bony bridge. Devices serve a mechanical role, holding the spine in correct alignment and providing structural support, while the body’s biology does the actual healing work, aided where needed by bone graft material.
Key Types of Spinal Fusion Devices
A complete fusion construct typically combines several device types working in concert: an interbody spacer restores disc height and alignment, pedicle screws anchor fixation into the vertebral body, rods or plates connect those anchors, and bone graft or synthetic substitutes provide the biological scaffold for new bone formation.
Interbody Fusion Devices
Interbody cages are implanted directly into the intervertebral disc space after the native disc material is removed. They restore disc height, decompress neural elements, and provide a structural load-sharing environment that promotes bony fusion. Access approaches determine the device geometry and surgical technique used.
The most common approaches include Anterior Lumbar Interbody Fusion (ALIF), Posterior Lumbar Interbody Fusion (PLIF), Transforaminal Lumbar Interbody Fusion (TLIF), Lateral Lumbar Interbody Fusion (LLIF/XLIF), and Oblique Lumbar Interbody Fusion (OLIF). Each approach has distinct anatomical access corridors, footprint sizes, and indications. Cervical fusion uses similar cage principles via the anterior cervical approach (ACDF).
Modern cages are manufactured from PEEK (polyether ether ketone), titanium, or titanium-coated PEEK. PEEK’s modulus of elasticity closely matches cortical bone, reducing stress shielding. Titanium’s osteoconductive surface promotes direct bone-implant contact. Surface texturing, 3D-printed porous structures, and lordotic or hyperlordotic angles represent key design differentiators among manufacturers.
Pedicle Screw Systems
Pedicle screws are the workhorse anchor of posterior spine fixation. Inserted through the pedicle, the bony bridge connecting the vertebral body to the posterior elements, they achieve purchase deep in cancellous and cortical bone. Polyaxial heads allow the surgeon to adjust rod angle after screw placement, simplifying construct assembly.
Cannulated designs accommodate guidewire placement under fluoroscopy or navigation. Fenestrated screws with side ports allow bone cement (PMMA) augmentation in severely osteoporotic patients, improving pullout strength. Expandable screws with deployable wings further address osteoporosis, which is increasingly relevant as the surgical population ages.
Rods and Plates
Posterior rods connect pedicle screws along the spinal column, completing the fixation construct. Standard 5.5 mm or 6.0 mm titanium or cobalt-chrome rods provide rigid stabilization. Titanium offers better MRI compatibility; cobalt-chrome provides higher stiffness. Pre-contoured rods for thoracolumbar deformity correction and hyperlordotic lumbar rods are now standard in deformity surgery.
Anterior cervical plates, low-profile titanium constructs, are fixed to the vertebral bodies in ACDF to supplement interbody cage fixation. Variable-angle screw holes and locking mechanisms prevent screw backout. Zero-profile interbody devices that eliminate a separate plate are increasingly popular, reducing dysphagia risk and surgical time.
Bone Grafts and Substitutes
No fusion occurs without a biological substrate. Autologous iliac crest bone graft (ICBG) remains the historical gold standard; it is osteogenic, osteoconductive, and osteoinductive, but harvesting it requires a second surgical site and carries donor-site morbidity. This limitation has driven strong growth in alternatives.
Demineralized bone matrix (DBM), processed allograft, synthetic calcium phosphate ceramics (hydroxyapatite, beta-tricalcium phosphate), and recombinant bone morphogenetic proteins (rhBMP-2, sold as INFUSE) are widely used. rhBMP-2 dramatically reduces autograft dependency but has faced scrutiny over ectopic bone formation, retrograde ejaculation (in ALIF), and potential cancer signal, leading to more cautious, evidence-based use. Newer peptide-based biologics and autologous platelet-rich plasma formulations are expanding the options.
Clinical Benefits and Challenges of Spinal Fusion Devices
Spinal fusion devices play a critical role in the surgical management of various spinal disorders, including degenerative disc disease, scoliosis, spinal instability, fractures, and spondylolisthesis. These devices, such as cages, rods, plates, screws, and biologics, are designed to stabilize the affected spinal segment and promote bone growth between adjacent vertebrae. One of the primary clinical benefits of spinal fusion devices is pain relief, particularly in patients suffering from chronic back pain caused by instability or nerve compression. By eliminating abnormal motion at the diseased segment, these devices can significantly improve patient mobility, posture, and overall quality of life.
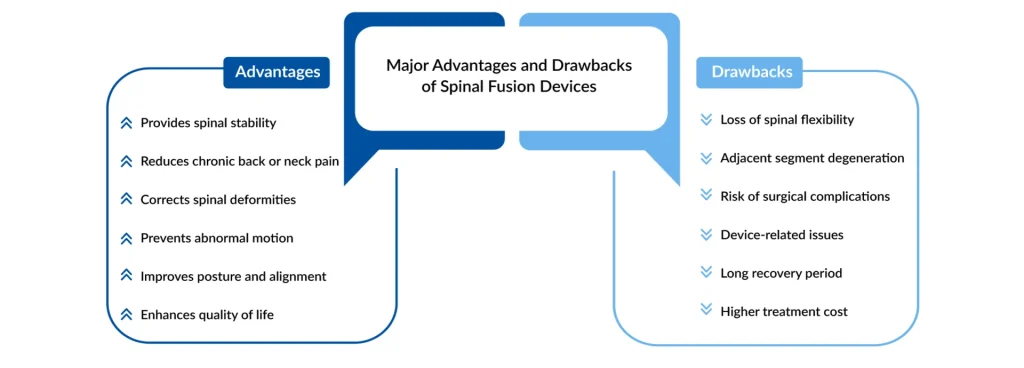
Another key advantage of spinal fusion devices is their ability to restore spinal alignment and structural integrity. In deformity correction procedures, such as those for scoliosis or kyphosis, advanced instrumentation systems help surgeons achieve precise correction while maintaining spinal balance. Minimally invasive spinal fusion techniques, supported by modern implant technologies, have also reduced operative trauma, shortened hospital stays, and accelerated recovery times. Additionally, innovations such as 3D-printed implants and expandable cages allow for better anatomical fit, enhanced load distribution, and improved fusion outcomes.
Despite these benefits, spinal fusion devices are associated with several clinical challenges. One of the most common concerns is nonunion or pseudarthrosis, where the bones fail to fuse completely, potentially leading to persistent pain and the need for revision surgery. Implant-related complications such as screw loosening, hardware breakage, cage migration, and infection can also occur, particularly in older patients or those with poor bone quality. Furthermore, adjacent segment disease remains a long-term challenge, as the fused segment may place additional stress on neighboring spinal levels, accelerating degeneration over time.
Patient selection and surgical complexity also significantly influence outcomes. Individuals with osteoporosis, obesity, diabetes, or smoking history may experience slower healing and higher complication rates. In addition, spinal fusion procedures can be costly and require considerable postoperative rehabilitation. As a result, clinicians must carefully weigh the benefits against the risks, considering conservative treatment options when appropriate. Continued advances in biomaterials, navigation systems, and regenerative therapies are expected to improve fusion success rates while minimizing complications in the future.
Recent Developments in the Spinal Fusion Devices Segment
- In March 2026, Spinal Elements® announced that it had received 510(k) clearance from the FDA and had successfully performed the first clinical cases using its Ventana A Anterior Lumbar Interbody Fusion (ALIF) System. The launch strengthened the company’s portfolio of 3D-printed titanium interbody implants for both lumbar and cervical fusion procedures and was introduced through a limited market rollout.
- In February 2026, 4WEB Medical announced that it had received 510(k) clearance from the U.S. FDA to market its SI Joint Truss System™.
- In January 2026, CornerLoc™ announced the publication of 6-month interim results from the SPARTAN Study, a prospective, multicenter clinical trial designed to evaluate patient-reported pain and functional outcomes following lateral-oblique sacroiliac (SI) joint fusion using the TransLoc 3D™ System.
- In January 2026, Omnia Medical, a medical technology company focused on developing surgical solutions for spine and interventional pain physicians, announced that it had commercially launched its FDA-cleared PsiF DNA™ Sacroiliac Joint Stabilization System.
- In January 2026, Spine Innovation, LLC announced that it had received FDA 510(k) clearance to market the LOGIC™ Titanium Implant System. The LOGIC™ Implant System represented the next-generation version of the LOGIC™ platform and incorporated OsteoSync™ Ti, a patented pure titanium lattice material developed by Sites Medical that had been implanted in more than 250,000 patients since 2014.
Spinal Fusion Devices Market Landscape and Future Growth Outlook
The global spinal fusion devices market represents a significant segment within the broader orthopedic and spinal implants industry. As per DelveInsight, the global spinal fusion devices market size is expected to increase from USD 7 billion in 2025 to USD 10.7 billion by 2034, at a CAGR of 5%, driven by the increasing prevalence of degenerative disc disease, scoliosis, spinal stenosis, trauma-related injuries, and chronic lower back pain.
The market is highly consolidated, with major players such as Zimmer Biomet, Stryker, Medtronic, Orthofix Medical Inc., NuVasive Inc., B. Braun Melsungen AG, XTANT MEDICAL, Globus Medical, ATEC Spine, Inc., Medacta International, Johnson & Johnson Services, Inc., Captiva Spine, Inc., ZAVATION, and others competing through continuous innovation, strategic acquisitions, and geographic expansion. Product categories include cervical fixation devices, thoracolumbar systems, interbody fusion cages, biologics, and minimally invasive spinal fusion systems. North America currently leads the market due to advanced healthcare infrastructure, high procedure volumes, and early adoption of robotic-assisted spine surgery, while Asia-Pacific is emerging as a high-growth region due to expanding healthcare access and rising surgical demand.
Technological advancements are reshaping the competitive landscape, particularly the adoption of 3D-printed implants, navigation-enabled systems, robotic-assisted surgery platforms, and motion-preserving solutions that improve surgical precision and patient outcomes. Hospitals and ambulatory surgical centers are increasingly shifting toward minimally invasive fusion procedures because of reduced blood loss, shorter hospital stays, and faster recovery times. In addition, the growing use of biologics and customized implants is improving fusion success rates, creating new revenue opportunities for manufacturers. These innovations are expected to intensify competition as companies seek differentiation through clinically superior and cost-effective solutions.
Looking ahead, the spinal fusion devices market is poised for steady expansion over the next decade, supported by aging populations, sedentary lifestyles, obesity-related spinal disorders, and increasing awareness of surgical treatment options. Market estimates indicate mid-single-digit CAGR growth, with continued momentum in outpatient spine surgery and emerging markets such as India, China, and Southeast Asia. However, pricing pressures, reimbursement scrutiny, and the rise of non-fusion alternatives may moderate growth in mature markets. Overall, companies that combine innovation, surgeon training, and expansion into underserved regions are expected to capture the strongest long-term opportunities.

Downloads
Article in PDF
Recent Articles
- Sonomotion Secures FDA Clearance for Break Wave™ Lithotripsy System; Aidoc Earns FDA Clearance fo...
- Orthopedic Implants: Evaluating the Market Potential of the Unparalleled & Life-Saving Equip...
- MiniMed Secures FDA Clearance for MiniMed Flex; Spinal Elements Expands Portfolio with FDA-Cleare...
- Gore Secures FDA Approval for Its First Deep Venous Stent for IVC and Iliofemoral Veins; Stereota...
- Bioventus’s StimRouter Pain Management Device; FDA Approval for eCoin Therapy; SurGenTec Launches...




