cystic fibrosis
Aug 05, 2025
Anbogen’s ABT-301 Cleared by FDA for Phase I/II Colorectal Cancer Trial; Dyne’s DYNE-251 Gets FDA Breakthrough Tag for Duchenne Muscular Dystrophy; BMS’s BREYANZI Accepted for FDA Priority Review in MZL; Clarametyx’s CMTX-101 Earns FDA Fast Track and QIDP for Infections; Anixa Begins FDA-Approved IND Transfer for Phase II Breast Cancer Vaccine Trial
Anbogen Secures FDA Nod to Begin Phase I/II Trial of ABT-301 Triplet in Advanced Colorectal Cancer Anbogen Therapeutics has received FDA clearance to initiate a Phase I/II clinical trial of ABT-301 in combination with tislelizumab and bevacizumab for patients with metastatic colorectal cancer (mCRC). The study w...
Read More...
Dec 24, 2024
FDA Approves Vertex’s ALYFTREK for Cystic Fibrosis; ZEPBOUND Gets FDA Approval for Obstructive Sleep Apnea in Obese Adults; Tonix’s TNX-102 SL NDA Accepted for Fibromyalgia; Quoin’s QRX003 Clears FDA for Netherton Syndrome Study; FDA Approves First Mesenchymal Stromal Cell Therapy for Acute GvHD
Vertex Receives US FDA Approval for ALYFTREK: A Breakthrough CFTR Modulator for Cystic Fibrosis Vertex Pharmaceuticals Incorporated announced that the FDA had approved ALYFTREK (vanzacaftor/tezacaftor/deutivacaftor), a once-daily triple combination CFTR modulator for the treatment of cystic fibrosis. ALYFTREK is...
Read More...
Nov 06, 2024
6 Groundbreaking Applications of Organoids That Are Changing Healthcare
The advent of organoid technology has marked a revolutionary milestone in biomedical research and healthcare. Organoids, which are three-dimensional structures derived from stem cells that mimic the architecture and functionality of specific organs, have opened new avenues for understanding diseases, testing drugs,...
Read More...
Jan 12, 2024
Is Bacteriophage a Potential Game-Changer in the Realm of Cystic Fibrosis Treatment?
Cystic Fibrosis impacts approximately 1 in every 3,500 births, with around 70,000 affected individuals globally, and 31,000 in the United States alone. Those with the condition experience the development of dense, adhesive mucus in their lungs, making them prone to infections. Over time, this leads to inflammation ...
Read More...
Nov 28, 2023
Dupixent Significantly Reduced COPD Exacerbations; Sosei Heptares to Regain Ownership of GSK4381406; AstraZeneca Gains Rights to Usynova’s KRAS Inhibitor; FDA Approvs OGSIVEO for Desmoid Tumors; Orphan Drug Designation for ARCT-032 for the Cystic Fibrosis; FDA Fast Track Designation for ADP101 for Food Allergies
Dupixent Significantly Reduced COPD Exacerbations In Second Positive Phase III Trial The latest trial for Dupixent (dupilumab) in chronic obstructive pulmonary disease (COPD), called NOTUS, has delivered promising results. It showed a significant 34% reduction in COPD exacerbations, confirming earlier positive f...
Read More...
Dec 13, 2022
Amgen to Purchase Horizon Therapeutics; IND Clearance to Vertex’s VX-522; FDA Fast-Track Designation for Moleculin’s WP1122; Orphan Drug Designation to Brim’s WP1122; Eisai Presents Results of Lecanemab Phase 3 Confirmatory Clarity Ad Study; UCB Announces Phase 3 Studies for Bimekizumab
Amgen Inc. Agrees to Purchase Dublin-based Horizon Therapeutics Plc. for €24.7 Billion Amgen Inc. has agreed to buy Dublin-based Horizon Therapeutics Plc. for €24.7 billion ($26 billion), in a deal that could face further delays or a breakdown in negotiations. Following Sanofi's withdrawal from the race, c...
Read More...
May 26, 2021
Alarming Growth of Chronic Respiratory Diseases (CRDs) and Their Prolonged Impact on the Quality of Life
Today, the increasing prevalence of Chronic Respiratory Diseases (CRDs) is one of the leading health challenges worldwide. CRDs affect hundreds of millions of people and cause huge mortality, morbidity, immense socio-economic burden, and disability-adjusted life years (DALYs). Over the past few decades, si...
Read More...
Nov 26, 2020
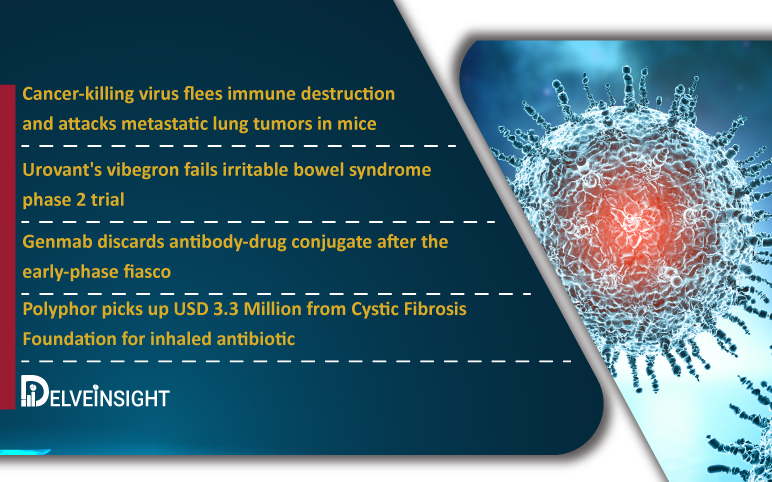
Cancer-killing virus flees immune destruction and attacks metastatic lung tumors; Urovant’s vibegron fails trial; Genmab discards antibody-drug conjugate; Polyphor picks up $3.3M for inhaled antibiotic
Cancer-killing virus flees immune destruction and attacks metastatic lung tumors in mice Several viruses have a natural ability to eliminate cancer; however translating them into treatments has proven difficult, partly due to the immune system tends to kill them before they can reach cancer cells. Researchers fr...
Read More...
Mar 26, 2020
FDA permits COVID-19 treatment, Roche’s antibiotics pact, Moderna’s vaccine, FDA permits Remdesivir, ReCode needs USD 80 Million
FDA permits COVID-19 treatments with blood from survivors The FDA revealed the convalescent plasma use that is derived from the donated blood of people that have recovered from COVID-19 and that might have beneficial antibodies. It is being used as an investigational treatment for patients with severe cases of t...
Read More...
Feb 10, 2020
Bridging the gap: Nontuberculous mycobacterium Infections Drug Pipeline
Slowly or rapidly growing bacteria- Nontuberculous mycobacteria (NTM), might appear innocuous, however, NTM infections have a notably higher total prevalence of 3,65,273 in the 7MM, estimates DelveInsight. Despite the global prevalence, clear from the facts and figures, the available therapies are entirely on an in...
Read More...








-Agonist.png)

