The Underactive Bladder Syndrome Treatment Landscape: Few Contenders, High Unmet Need
Mar 16, 2026
Summary
- The current treatment landscape for underactive bladder largely depends on pharmacological management and catheterization.
- Despite the significant unmet need, the clinical development pipeline for UAB remains relatively sparse.
- Notable candidates include TAC-302, being developed by Taiho Pharmaceutical and ZG-802 from Zeria Pharmaceutical, both currently in Phase II development, along with SFG-02 from SFG Sciences, which is in Phase I clinical evaluation. These emerging therapies highlight ongoing efforts to address the limited treatment options available for UAB.
Underactive bladder syndrome traps millions in a silent struggle with incomplete bladder emptying, straining daily life, and risking severe complications. Often misdiagnosed as overactive bladder (OAB) or bladder outlet obstruction (BOO) due to symptom overlap like urgency and frequency, it affects up to 40% of those over 70, with males more impacted by prostate issues.
Limited data exist on UAB prevalence, with current literature offering few estimates; existing studies highlight significant gaps and limitations in understanding the disease’s epidemiology. Among the 7MM + China, China accounted for the highest diagnosed prevalent cases of UAB with more than 11 million in 2024, which is expected to rise by 2034.
Downloads
Click Here To Get the Article in PDF
Recent Articles
As per DelveInsight’s estimations, the total diagnosed prevalent cases of LUTS in the 7MM + China were approximately 67 million cases in 2024 and are projected to increase by 2034. The anticipated rise in prevalent cases of moderate to severe LUTS in the US is attributed to an aging population, increased prevalence of obesity, and sedentary lifestyles.
There is no definitive UAB treatment to improve the quality of life and prevent complications significantly. The focus is on bladder emptying and patient compliance to avoid upper urinary tract damage.
Treatment Gaps Sustain Catheterization’s Dominance in the UAB Therapeutic Market
Management of underactive bladder currently involves a range of interventions that begin with conservative behavioral strategies and clean intermittent catheterization (CIC), and extend to pharmacologic therapies, surgical procedures, and emerging regenerative approaches such as stem cell and gene-based treatments.
At present, CIC remains the standard of care for patients who are unable to achieve effective bladder emptying. Intermittent and indwelling catheterization, along with sacral neuromodulation (SNM), constitute the primary treatment options. However, these approaches are associated with risks of infection, financial burden, and patient dissatisfaction, and they primarily address symptoms rather than the underlying disease pathology.
Pharmacologic management of UAB remains largely exploratory. Several UAB drug classes, including alpha-blockers, muscarinic agonists, cholinesterase inhibitors, and prostaglandin E2, have been investigated to enhance detrusor contractility or reduce bladder outlet resistance. Despite these efforts, clinical benefits have generally been limited, underscoring the persistent challenge of achieving meaningful therapeutic outcomes in UAB.
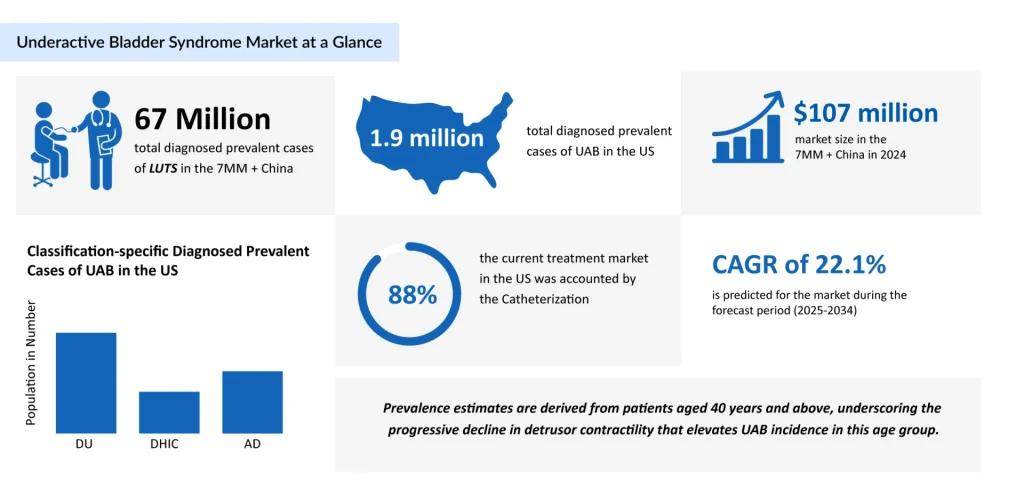
For several decades, catheterization has dominated the UAB therapeutic landscape. In 2024, catheter-based interventions accounted for nearly USD 874 million, representing approximately 88% of the total US UAB treatment market.
Pharmacotherapy, by contrast, covers roughly one-third of the U.S. UAB patient population, while catheterization accounts for the majority of treatment utilization. Among off-label pharmacologic options, alpha antagonists represent the largest segment, with an estimated market value of approximately USD 58 million in 2024. Clinicians primarily use this class to reduce bladder outlet resistance; however, long-term efficacy remains limited for many patients.
Muscarinic agents, including bethanechol and carbachol, are also utilized, though less frequently than alpha antagonists, contributing approximately USD 36 million to the market in 2024. Overall, pharmacotherapy represents about USD 114 million of the current UAB market, including approximately USD 20 million attributed to other drug classes.
Sadaf Javed, Manager of Forecasting and Analytics at DelveInsight, highlighted that the discontinuation of ASP8302 following inadequate Phase II efficacy, coupled with Ono Pharmaceutical’s decision to stop developing ONO-8055, reflects the high development risks in underactive bladder (UAB) research and underscores the urgent need for therapies with proven clinical benefits.
Javad further emphasized that significant growth opportunities exist in emerging markets, where the increasing prevalence of UAB and expanding healthcare access are expected to drive market expansion.
New Candidates Begin to Shape the Underactive Bladder Pipeline
Despite the significant unmet need, the UAB therapeutic pipeline remains limited, with only a handful of investigational candidates advancing through early clinical development. Currently, assets such as TAC-302 from Taiho Pharmaceutical, ZG-802 (acotiamide) from Zeria Pharmaceutical, and SFG-02 developed by SFG Sciences represent the few active efforts to address this challenging condition.
TAC-302 is an orally bioavailable small molecule designed to stimulate neurite outgrowth in peripheral neurons, a mechanism aimed at restoring bladder nerve function. Preclinical studies demonstrated that the therapy could improve bladder denervation and alleviate both storage and voiding dysfunctions, even when treatment was initiated after symptoms had already developed. The candidate is currently being investigated in Phase II clinical trials for patients with detrusor underactivity associated with overactive bladder (OAB). While early clinical findings suggested potential benefits, the results have yet to establish definitive efficacy, and limited recent updates on its development underscore the need for further studies to fully validate its therapeutic potential.
Another promising candidate, ZG-802 (acotiamide), is being developed as an oral small-molecule therapy that modulates lower urinary tract function. By targeting impaired bladder contractility, one of the central drivers of UAB, ZG-802 aims to improve bladder emptying and overall urinary function. Its progression into Phase II development indicates encouraging early-stage safety and efficacy signals, positioning it as one of the more notable contenders in the evolving UAB pipeline.
Meanwhile, SFG-02 represents a novel therapeutic approach currently in Phase I development. Unlike conventional oral therapies, SFG-02 is administered via a transdermal patch, offering a potentially convenient and non-invasive treatment option for patients. The candidate is expected to enter proof-of-concept studies in the United States and Japan by 2026, although its development has yet to expand into Europe.

Even though there are limited number of candidates and geographic concentration of clinical trials, the anticipated launch of these therapies will bring a wave of change in the UAB therapeutic segment.
What Lies Ahead in the UAB Therapeutic Space?
The treatment landscape for underactive bladder reflects a challenging and evolving market, marked by a considerable demand for more effective therapeutic options. UAB is characterized by diminished or inadequate bladder contractions, resulting in delayed or incomplete bladder emptying. Proper management requires comprehensive neurologic and urodynamic assessments, with treatment strategies focused on protecting the upper urinary tract, preventing bladder overdistension, and minimizing residual urine volume.
Innovative and emerging treatment approaches are showing potential in addressing UAB. Neuromodulation strategies, particularly sacral neuromodulation, are being investigated for their ability to enhance bladder activity by influencing neural signaling pathways. In addition, botulinum toxin injections may be utilized to relax the external urethral sphincter in patients with functional bladder outlet obstruction, thereby lowering resistance to urine flow. Although the development pipeline remains limited, several investigational therapies, such as TAC-302, ZG-802, and SFG-02, are currently undergoing clinical evaluation as prospective UAB treatments.
Market estimates indicate that the combined 7MM and China UAB market was valued at approximately USD 107 million in 2024, and it is projected to grow throughout the forecast period from 2025 to 2034. At present, first-line pharmacologic treatment largely involves oral parasympathomimetic agents like bethanechol, which aim to stimulate detrusor muscle contraction. However, their clinical benefits are often constrained by limited efficacy and cholinergic adverse effects.
Similarly, oral alpha-adrenergic blockers such as tamsulosin and silodosin are prescribed to decrease urethral resistance and facilitate bladder emptying, though they do not directly address impaired detrusor contractility. Looking ahead, TAC-302 is expected to emerge as a leading therapy and is projected to capture the largest share of the UAB market across the 7MM and China by 2034.
In summary, the rising global geriatric population, increasing prevalence of neurogenic conditions such as diabetes and spinal cord injury, and improvements in diagnostic awareness are expected to expand the identified patient pool. As these factors converge with the anticipated introduction of novel therapies and neuromodulation-based interventions, the UAB treatment market is projected to witness robust expansion over the forecast period, with substantial opportunities for innovation and commercial growth.

Downloads
Article in PDF




