Companion Diagnostics in Healthcare: Powering the Era of Precision Medicine
Mar 11, 2026
Table of Contents
The landscape of modern medicine is undergoing a profound shift from a “one-size-fits-all” approach to a model that is deeply personalized and data-driven. At the heart of this transformation is the concept of precision medicine, which seeks to tailor medical treatment to the individual characteristics of each patient. Central to the success of this paradigm shift is the development and integration of companion diagnostics (CDx). These specialized tests act as the bridge between clinical diagnosis and targeted therapy, ensuring that patients receive the most effective treatments based on their unique biological makeup. By identifying specific biomarkers, companion diagnostics allow clinicians to move beyond trial-and-error prescribing, significantly improving patient outcomes and reducing the economic burden of ineffective treatments on global healthcare systems.
What Are Companion Diagnostics?
Companion diagnostic is a medical device, often an in vitro diagnostic (IVD), which provides information that is essential for the safe and effective use of a corresponding drug or biological product. The primary role of these tests is to identify patients who are most likely to benefit from a particular therapeutic, identify those at high risk for serious adverse reactions, and monitor the response to treatment to adjust dosing or strategy for improved safety. As outlined in official FDA guidance, the use of a companion diagnostic is often stipulated in the instructions for use in the labeling of both the diagnostic device and its corresponding therapeutic product. These diagnostics are distinct from general screening tests. While a standard blood test might measure glucose levels for general health monitoring, a companion diagnostic is intrinsically linked to a specific drug.
Downloads
Click Here To Get the Article in PDF
Recent Articles
- Promising Nephrotic Syndrome Treatments: A Look into the Future
- Geneseeq Technology Inc.’s GENESEEQPRIME® Assay Receives FDA 510(k) Clearance; Medtronic’s MiniMe...
- Molecular Diagnostics: Bridging the Gap Between Diagnosis and Treatment
- SIBIONICS’s Ground-breaking GS1 Continuous Glucose Monitoring System; Abbott’s HPV Test to Run on...
- Tempus Launches FDA-Approved xT CDx Test Nationwide; B. Braun Enhances Catheter Securement with t...
Companion Diagnostics: Transforming Cancer Care
Oncology has been the pioneer field for companion diagnostics, largely because cancer is a disease driven by genetic mutations and molecular alterations. The ability to “profile” a tumor’s DNA has revolutionized how oncologists approach treatment. Historically, cancer was treated based on its location in the body, such as the lungs, breast, or colon. Today, companion diagnostics allow for a molecular-level classification.
In the realm of lung cancer, for instance, companion diagnostics are used to detect EGFR mutations. If a patient’s tumor expresses these mutations, they can be treated with specific tyrosine kinase inhibitors (TKIs). The FDA maintains a comprehensive list of these approved pairings, which have moved the needle from general chemotherapy, which attacks both healthy and cancerous cells, to targeted therapies that specifically home in on the mechanisms driving the tumor’s growth. This transition has significantly extended the survival rates of patients with late-stage malignancies who previously had very limited options.
Key Benefits of Companion Diagnostics in Oncology
The integration of companion diagnostics into oncology brings several critical benefits, primarily centered on patient safety and therapeutic efficacy. One of the most significant advantages is the reduction of adverse drug reactions. By identifying patients who are genetically predisposed to toxicity or who lack the biological target the drug is designed to hit, clinicians can avoid prescribing treatments that would cause harm without providing benefit. This is particularly vital in oncology, where many treatments, such as certain immunotherapies or high-dose chemotherapies, carry the risk of severe side effects.
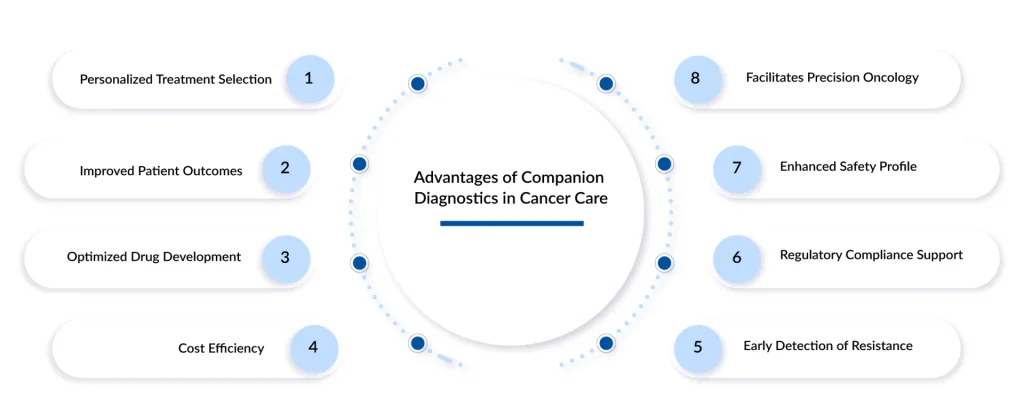
Furthermore, companion diagnostics enhance the cost-effectiveness of healthcare. Data from DelveInsight indicates that the rising focus on personalized medicine is driven by the need to make treatment regimens more efficient and to increase patient compliance by ensuring that the therapy chosen is the one most likely to work. When a drug is only given to those who will respond, the “waste” associated with expensive, ineffective therapies is minimized. This allows healthcare providers to allocate resources more effectively, ensuring that the high cost of modern biologics is justified by high success rates. Additionally, CDx tests can accelerate the drug development process for pharmaceutical companies by allowing them to conduct clinical trials in highly stratified, “biomarker-positive” populations, leading to clearer results and faster regulatory approvals.
Beyond Oncology: Expanding Applications of Companion Diagnostics
While oncology currently dominates the market, the application of companion diagnostics is rapidly expanding into other therapeutic areas. The same principles of genetic and molecular profiling are being applied to infectious diseases, cardiovascular conditions, and neurological disorders. For example, in the treatment of HIV, companion diagnostics are used to determine a patient’s co-receptor tropism before prescribing certain entry inhibitors. Similarly, in the field of cystic fibrosis, genetic testing is essential to identify patients with specific mutations, such as the G551D mutation, who will respond to specialized cystic fibrosis transmembrane conductance regulator (CFTR) modulators.
The rising prevalence of chronic and infectious diseases globally, as noted by the WHO, is a major driver for this expansion. In neurology, there is a growing interest in using companion diagnostics for Alzheimer’s disease. New therapies targeting amyloid-beta plaques require diagnostic confirmation, often through PET scans or cerebrospinal fluid analysis, to ensure the patient actually has the specific pathology the drug is designed to treat. As research into the human genome continues to advance, the potential for CDx to guide treatment for autoimmune diseases and rare genetic conditions grows, further embedding these tests into the fabric of general medical practice.
Key Challenges in Companion Diagnostics
Despite their promise, several challenges hinder the widespread adoption of companion diagnostics. One of the primary obstacles is the complexity of the regulatory landscape. To gain approval, a companion diagnostic must demonstrate both analytical validity (the test correctly identifies the biomarker) and clinical validity (the biomarker result reliably predicts the drug’s performance). This often requires the simultaneous development of both the drug and the diagnostic, known as “co-development,” which can be logistically and financially demanding for manufacturers.
Reimbursement is another significant hurdle. While the benefits of precision medicine are clear, many healthcare payers are slow to adopt reimbursement models for high-cost diagnostic tests. There is often a disconnect between the value a test provides in saving future costs and the immediate price tag of the test itself. Furthermore, as noted in reports by DelveInsight, the lack of standardized reimbursement rates across different regions can restrict market growth and patient access. Additionally, the technical infrastructure required for some advanced tests, such as Next-Generation Sequencing (NGS), is not available in all clinical settings, particularly in low- and middle-income countries, creating a “precision medicine gap” where only certain populations can benefit from these advancements.

Future Directions: From Single Markers to Systems-Level Guidance
The future of companion diagnostics lies in the move from single-biomarker tests to complex, systems-level diagnostic panels. Instead of looking for one specific mutation, new technologies like Next-Generation Sequencing allow for the simultaneous screening of hundreds of genes. This provides a “genomic signature” of a patient’s disease, offering a more comprehensive view of how they might respond to multiple different therapies. The FDA has already begun approving such “group labeling” or “class-based” diagnostics, which allow a single test to serve as a companion for an entire class of drugs, rather than just one specific product.
According to market analysis by DelveInsight, the global companion diagnostics market was valued at USD 5.91 billion in 2024 and is projected to reach USD 11.89 billion by 2032, growing at a CAGR of 12.32%. This growth is fueled by the exponential rise in cancer cases and the increasing integration of Artificial Intelligence (AI) in diagnostics to improve accuracy.
Key players driving this innovation include Agilent Technologies, Inc., F. Hoffmann-La Roche Ltd, Myriad Genetics, Inc., Abbott Laboratories, QIAGEN, BIOMÉRIEUX, Guardant Health Inc., Illumina, Inc., Pillar Biosciences, Inc., Leica Biosystems, Thermo Fisher Scientific Inc., Almac Group, NeoGenomics Laboratories, Invitae Corporation, 3D Medicines Corporation, ASURAGEN, INC. (Biotechne), Invivoscribe, Inc., Amoy Diagnostics Co., Ltd., Laboratory Corporation of America® Holdings, SAGA Diagnostics, and others.
These companies are increasingly focusing on liquid biopsies, non-invasive blood tests that detect circulating tumor DNA, which represent the next frontier in real-time treatment monitoring. As we move forward, the convergence of genomic data, AI-driven analytics, and sophisticated diagnostic tools will solidify companion diagnostics as the cornerstone of the precision medicine era, ensuring that the right patient receives the right treatment at the right time.

Downloads
Article in PDF
Recent Articles
- Edwards’s Pascal Precision System; Abbott’s New Spinal Cord Stimulation Device; Imagin to A...
- Precision Medicine: An emerging approach to conventional treatment
- Medtronic Receives FDA Approval for OmniaSecure™ Lumenless Defibrillation Lead and Presents Posit...
- SIBIONICS’s Ground-breaking GS1 Continuous Glucose Monitoring System; Abbott’s HPV Test to Run on...
- Companion Diagnostics