Molecular Diagnostics: Bridging the Gap Between Diagnosis and Treatment
Mar 25, 2026
Table of Contents
Molecular diagnostics is a rapidly advancing field within laboratory medicine that applies molecular biology techniques to detect and quantify specific DNA or RNA sequences. By enabling earlier detection, greater diagnostic accuracy, and more informed clinical decision-making, these technologies have become indispensable in modern healthcare. Their impact spans clinical medicine, public health, and biomedical research, particularly in the management of infectious diseases, inherited disorders, cancer, and personalized medicine.
Global health authorities underscore the critical role of diagnostics in improving health outcomes. The World Health Organization (WHO) emphasizes that limited access to reliable diagnostic tools contributes significantly to preventable morbidity and mortality worldwide, with millions of deaths linked to delayed or inaccurate diagnosis. Consequently, governments and health systems are increasingly prioritizing molecular diagnostics as a cornerstone of disease control, surveillance, and clinical management. Similarly, the Centers for Disease Control and Prevention (CDC) highlights molecular diagnostic technologies as foundational to outbreak detection and genomic surveillance, enabling more effective monitoring of pathogen evolution and antimicrobial resistance.
Downloads
Click Here To Get the Article in PDF
Recent Articles
- Cancer Diagnostic Market: Evaluating the Major Growth Factors and the Key Developments in the Domain
- Medtronic Receives FDA Approval for OmniaSecure™ Lumenless Defibrillation Lead and Presents Posit...
- Key Insights and Analysis Into the Point of Care Diagnostics Market
- Tempus Launches FDA-Approved xT CDx Test Nationwide; B. Braun Enhances Catheter Securement with t...
- Medtronic’s Hugo Robotic-Assisted Surgery System Trial; Insightec’s Pivotal LIBERATE Clinical Tri...
Alongside these public-health priorities, market analyses reflect strong commercial momentum. DelveInsight estimates the global molecular diagnostics market value at USD 9.56 billion in 2024, with projections indicating growth to around USD 16.12 billion by 2032, driven by rising infectious disease burden, expanding clinical applications, and continuous technological innovation.
Core Technologies Powering Molecular Testing
At the core of molecular diagnostics are highly sensitive technologies designed to detect nucleic acids with speed and precision. Polymerase chain reaction (PCR) and its quantitative variant (qPCR) remain the most widely used methods, capable of amplifying minute quantities of DNA or RNA to detectable levels. The CDC identifies PCR as a cornerstone of pathogen detection due to its exceptional sensitivity, specificity, and ability to quantify genetic material, making it essential for diagnosis, disease monitoring, and treatment evaluation.
The WHO strongly endorses nucleic acid amplification tests (NAATs) for priority diseases such as tuberculosis. WHO-recommended molecular assays can detect Mycobacterium tuberculosis and identify rifampicin resistance within hours, representing a major improvement over traditional culture methods that require weeks.
Next-generation sequencing (NGS) further expands diagnostic capability by enabling comprehensive genomic analysis rather than single-target detection. NGS supports detailed pathogen characterization, identification of drug-resistance mutations, and analysis of human genetic variation. The WHO recognizes targeted sequencing approaches as powerful tools for tuberculosis drug-resistance surveillance and for strengthening global infectious-disease monitoring systems. These methods are complemented by microarrays, hybridization-based assays, and increasingly sophisticated point-of-care molecular platforms, together forming a versatile and evolving diagnostic ecosystem.
Clinical Applications Across Medical Specialties
Molecular diagnostics has transformed clinical practice across numerous specialties. In infectious disease management, molecular assays consistently outperform conventional microscopy and culture methods in both sensitivity and turnaround time. The WHO reports that molecular testing is central to the diagnosis and management of HIV, tuberculosis, hepatitis, and emerging viral infections, enabling earlier treatment initiation, reduced transmission, and improved patient outcomes.
The scale of this need is substantial. WHO data indicate that approximately 39 million people were living with HIV globally in 2022, highlighting the importance of accurate molecular viral-load testing and resistance profiling. Similarly, hepatitis B and C infections affected an estimated 304 million people worldwide, reinforcing the critical role of molecular assays in screening, diagnosis, and treatment monitoring.
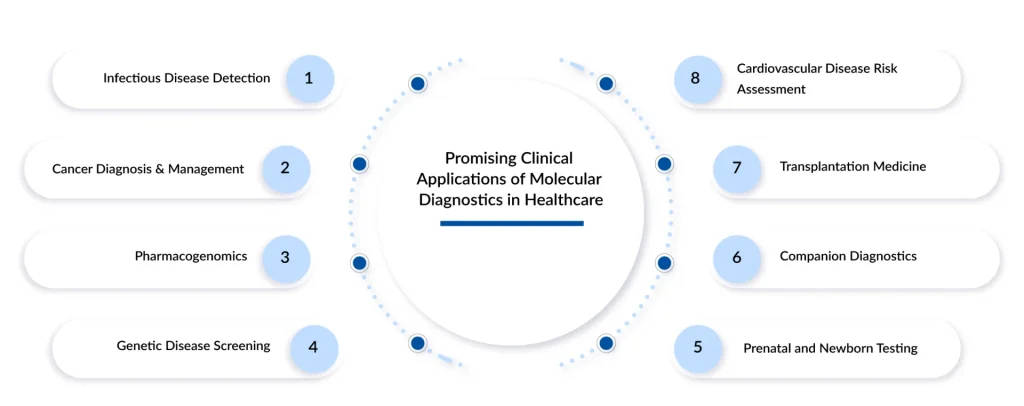
In oncology, molecular diagnostics underpins precision cancer care by identifying actionable genetic mutations and biomarkers that guide targeted therapies and immunotherapies. Regulatory authorities such as the FDA maintain formal lists of companion diagnostics, tests directly linked to specific drug therapies, demonstrating how molecular results now directly influence treatment selection and clinical outcomes.
Genetic testing and prenatal screening also rely heavily on molecular techniques. National healthcare systems, including the United Kingdom’s National Health Service (NHS), integrate genomic and molecular testing into routine care to diagnose inherited conditions and support informed reproductive decision-making. In transplantation medicine and hematology, quantitative molecular assays are used to monitor viral reactivation and minimal residual disease, enabling early intervention and improved long-term prognosis.
Role in Personalized and Precision Medicine
Precision medicine seeks to tailor healthcare interventions to individual patient characteristics, and molecular diagnostics is central to this approach. By identifying the molecular drivers of disease, these tests allow clinicians to select therapies that are more likely to be effective for specific patient subgroups. The FDA emphasizes that companion diagnostics ensure targeted therapies are prescribed only when supported by appropriate molecular evidence, improving both safety and therapeutic efficacy.
In oncology, molecular profiling has fundamentally shifted treatment paradigms away from tissue-based classification toward genomics-driven decision-making. Healthcare systems such as the NHS Genomic Medicine Service report that integrating molecular diagnostics into cancer pathways enhances diagnostic accuracy, supports personalized interventions, and reduces unnecessary or ineffective treatments.
Pharmacogenomics represents another growing application area. By identifying genetic variants that influence drug metabolism and response, molecular diagnostics can guide medication choice and dosing, helping to prevent adverse drug reactions, a significant source of preventable harm in healthcare systems worldwide.
Challenges and Considerations for Healthcare Systems
Despite its clear clinical value, expanding access to molecular diagnostics presents notable challenges. High upfront costs for instruments, reagents, and maintenance can strain healthcare budgets, particularly in low- and middle-income countries. The WHO advocates for careful selection of molecular platforms based on local disease prevalence, infrastructure, and workforce capacity to ensure sustainability and maximize public-health impact.
Regulatory oversight and quality assurance are equally critical. The FDA requires molecular diagnostic tests to demonstrate analytical validity, clinical validity, and clinical utility before approval, ensuring reliable and safe use in patient care. Laboratories must participate in external quality-assessment programs and maintain accreditation to ensure consistent performance.

Workforce limitations also pose a significant barrier. Advanced molecular testing requires specialized expertise in molecular biology, data analysis, and bioinformatics. The CDC highlights the need for sustained investment in laboratory training and workforce development to address skill shortages. Additionally, the expanding use of genetic data raises ethical, legal, and privacy concerns, underscoring the importance of robust governance frameworks to protect patient rights and maintain public trust.
Emerging Trends and Future Directions
Molecular diagnostics continues to evolve rapidly, with genomic sequencing increasingly used for real-time surveillance of emerging infectious diseases. The CDC reports that these approaches allow health authorities to detect outbreaks earlier, monitor pathogen mutations, and respond more effectively to public-health threats. The WHO similarly promotes sequencing technologies for antimicrobial-resistance monitoring and global health-security initiatives.
At the same time, advances in point-of-care molecular diagnostics are bringing highly accurate testing closer to patients, including in remote and resource-limited settings. WHO guidance emphasizes the potential of these platforms to reduce diagnostic delays and improve equity in access to essential health services.
In conclusion, molecular diagnostics has fundamentally reshaped modern healthcare, transforming the detection, monitoring, and management of disease across infectious medicine, oncology, genetics, and precision therapeutics. Public-health institutions and regulatory bodies provide essential guidance, standards, and oversight to ensure safe and effective implementation, while market dynamics signal strong and sustained demand. By aligning technological innovation with evidence-based policy, workforce development, and ethical governance, healthcare systems can fully harness the potential of molecular diagnostics to improve outcomes for individuals and populations alike.

Downloads
Article in PDF
Recent Articles
- Noninvasix’s LIVOx™ Central Venous Oxygenation Monitor; LivaNova’s aura6000 System; Oxford...
- AbbVie & iSTAR’S Strategic Alliance; Baxter Launches Welch Allyn RetinaVue 100 Imager PRO; I...
- Stryker’s LIFEPAK 35 Monitor/Defibrillator; Qiagen’s QIAstat-Dx Respiratory Panel; Evolution Opti...
- Polymerase Chain Reaction (PCR) Market: Evaluating the Major Market Dynamics and Key Trends Drivi...
- CereVasc’s eShunt System Study; FDA Approves NGS-Based CDx for Trastuzumab Deruxtecan; Nanopath S...