GSK’s LYNAVOY Enters PBC Treatment Space, Targeting High-Burden Pruritus Segment
Mar 27, 2026
Summary
- LYNAVOY is an oral IBAT inhibitor approved to treat cholestatic pruritus in adults with PBC, making it the first medicine in the US specifically cleared for this indication.
- Up to 89% of people with PBC experience cholestatic pruritus, an internal, often unbearable itch that can disrupt sleep, aggravate fatigue, and severely diminish quality of life.
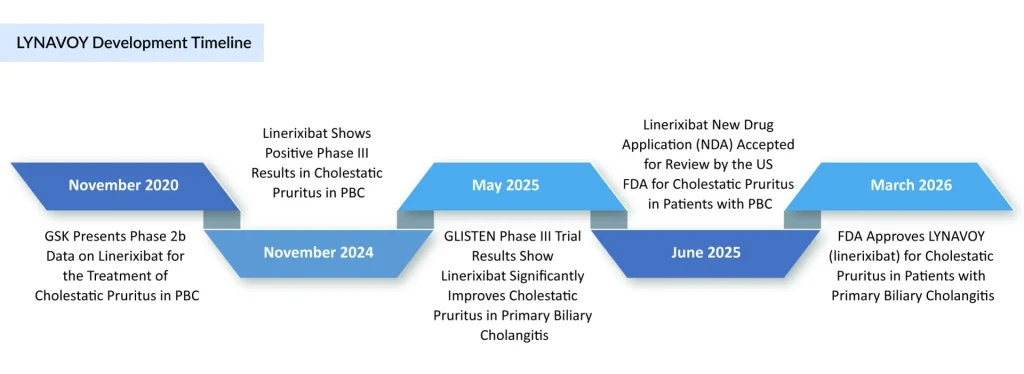
- Approval is underpinned by the phase III GLISTEN trial, where 238 PBC patients with moderate to severe itch achieved clinically meaningful itch reductions versus placebo on a 10‑point WI‑NRS scale, with significant improvements emerging as early as two weeks and sustained over 24 weeks.
The FDA has approved GSK’s LYNAVOY (linerixibat), ushering in a first-in-class breakthrough for cholestatic pruritus in adults with primary biliary cholangitis (PBC). This milestone signals a major leap in hepatology, finally delivering a targeted, mechanism-driven solution for a burdensome symptom that has long lacked effective treatment options.
Primary biliary cholangitis is a chronic, autoimmune cholestatic liver disease in which immune‑mediated destruction of the small intrahepatic bile ducts leads to progressive cholestasis, fibrosis, cirrhosis, and ultimately liver failure or transplantation if left untreated. PBC predominantly affects middle‑aged women and is typically suspected when cholestatic liver biochemistries are identified or when patients present with symptoms such as fatigue or pruritus. Pruritus in PBC, often described as an intense “internal itch”, is one of the most debilitating manifestations of the disease, severely impairing sleep, daily functioning, mental health, and overall quality of life. Epidemiological data cited by GSK indicate that up to 89% of individuals living with PBC experience cholestatic pruritus at some point in their disease course, underscoring its near‑ubiquitous impact in this population. In the United States alone, PBC is estimated to affect more than 100,000 adults, suggesting a substantial subset of patients with clinically significant itch and a high unmet need for targeted symptom control.
Downloads
Click Here To Get the Article in PDF
Recent Articles
- Merck and Moderna Initiate Study to Evaluate V940; FDA Approves Vertex and CRISPR Therapeutics’ C...
- Expanding Horizons in Chronic Pruritus Treatment: Emerging Therapies Target Diverse Mechanisms
- PPAR and NOX Agonist: Savior of Underserved Primary Biliary Cholangitis Patients
- GSK Receives FDA Fast Track Designation for Bepirovirsen; Gilead to Acquire CymaBay Therapeutics;...
- Sanofi’s Venglustat Gains US Breakthrough Therapy Nod, Boosting Hope for Type 3 Gaucher Disease; ...
Historically, management of PBC has focused on disease‑modifying therapy with UDCA as first‑line PBC treatment and additional agents such as obeticholic acid for patients with an inadequate biochemical response, but no therapy was specifically approved in the US to treat cholestatic pruritus itself. Off‑label approaches, including bile acid sequestrants, rifampin, opioid antagonists, and antidepressants, have been used with variable efficacy and tolerability, leaving many patients undertreated or unable to tolerate existing options. The approval of a targeted IBAT inhibitor such as LYNAVOY therefore addresses a highly visible gap in the PBC treatment armamentarium.
LYNAVOY is the brand name for linerixibat, an orally administered small‑molecule IBAT inhibitor. By blocking the reuptake of bile acids from the terminal ileum back into the enterohepatic circulation, IBAT inhibitors reduce circulating bile acid levels and downstream mediators thought to drive cholestatic itch. This mechanism provides a rational, targeted approach to pruritus in cholestatic liver disease, distinct from traditional systemic antipruritic drugs.
According to the FDA‑approved prescribing information, LYNAVOY is indicated for the treatment of cholestatic pruritus associated with primary biliary cholangitis in adult patients. It is, therefore, not a disease‑modifying agent for PBC per se, but a symptom‑focused therapy intended to be used alongside standard PBC treatments such as UDCA, rather than as a replacement. GSK highlights LYNAVOY as its first liver medicine to reach approval from its pipeline, emphasising the company’s broader strategy to expand into liver disease and rare disease portfolios.
Regulatory recognition reflects the rarity and severity of the condition: linerixibat has been granted Orphan Drug Designation in the United States, European Union, and Japan for the treatment of cholestatic pruritus in patients with PBC, and has been awarded priority review status in China. These designations confer incentives such as market exclusivity and regulatory support that can materially enhance the product’s commercial attractiveness.
Beyond the US, regulatory submissions for linerixibat are under review in multiple jurisdictions, including the European Union, the United Kingdom, Canada, and China. In China, the therapy has been granted priority review for cholestatic pruritus in PBC, reflecting unmet need and the potential for accelerated access if efficacy and safety are confirmed.
The FDA’s decision is primarily supported by the GLISTEN Phase III trial, a global, randomised, placebo‑controlled study enrolling adults with PBC and moderate‑to‑severe cholestatic pruritus despite stable background therapy. Participants were randomised to receive oral linerixibat or placebo in addition to their existing PBC treatment, with follow‑up over 24 weeks.
GLISTEN met its primary endpoint, demonstrating a statistically significant and clinically meaningful reduction in itch intensity compared with placebo, as measured by patient‑reported instruments. Importantly for patient quality of life, itch relief was described as rapid, with improvements evident as early as week 2, and sustained across the 24‑week treatment period. Key secondary endpoints, including reductions in itch‑related sleep interference, were also achieved, supporting the broader impact of treatment beyond itch scores alone.
The safety profile observed in GLISTEN was consistent with the known class effects of IBAT inhibition, typically involving gastrointestinal events attributable to alterations in bile acid handling. No new major safety signals have been publicly highlighted in the approval communications; however, longer‑term data and post‑marketing experience will be important to fully characterise the risk–benefit balance in clinical practice.
Ahead of the US approval, GSK announced a global licensing agreement with Italian pharmaceutical company Alfasigma S.p.A., granting Alfasigma exclusive worldwide rights to develop, manufacture, and commercialise linerixibat. Under the terms of the deal, GSK stands to receive up to USD 690 million in aggregate payments, structured as a USD 300 million upfront payment, a USD 100 million milestone tied to anticipated US approval by late March, additional regulatory milestones for approvals in Europe and the UK, and up to USD 270 million in sales‑based milestones.
In addition to these milestone payments, GSK will receive tiered double‑digit royalties on global sales of LYNAVOY generated by Alfasigma, maintaining economic participation in the asset despite transferring operational control. The transaction remains subject to customary closing conditions and regulatory clearances, including review under the Hart‑Scott‑Rodino Act in the United States.
Alfasigma is one of the leading pharmaceutical companies in Italy, with a long‑standing commercial presence in gastroenterology and liver disease across more than 100 countries, positioning it as a logical commercial partner for a PBC‑focused therapy. For Alfasigma, LYNAVOY adds a differentiated, first‑in‑class IBS‑targeting medicine to its hepatology portfolio, expanding its reach in rare liver conditions and potentially strengthening its global footprint beyond its traditional markets.
Several emerging PBC therapies targeting bile acid transport and nuclear receptors are progressing through late‑stage development, including other IBAT inhibitors such as volixibat and PPAR agonists like saroglitazar, which have reported positive Phase III results and may seek regulatory approval in the coming years.
Sadaf Javed, Manager of Forecasting at DelveInsight, said that in this competitive but still underserved landscape, LYNAVOY differentiates itself by being the first therapy in the US with a specific label for cholestatic pruritus in PBC, rather than focusing primarily on biochemical disease markers.
Given its symptomatic focus, LYNAVOY is likely to be used in combination with standard PBC therapies, offering an add‑on option for patients who remain highly symptomatic despite biochemically controlled disease. This positioning may mitigate direct head‑to‑head competition with disease‑modifying agents and instead create opportunities for complementary use, particularly in patients whose primary complaint is relentless itching.
The approval of LYNAVOY also validates IBAT inhibition as a therapeutic strategy for cholestatic pruritus, which may catalyse further research into similar mechanisms for other cholestatic liver diseases, such as primary sclerosing cholangitis or intrahepatic cholestasis of pregnancy, although such indications would require dedicated clinical evaluation. As additional data accrue from long‑term extension studies, real‑world evidence, and potential head‑to‑head or combination trials, the understanding of where LYNAVOY fits best within the broader PBC management algorithm will continue to sharpen.

Downloads
Article in PDF
Recent Articles
- GSK Receives FDA Fast Track Designation for Bepirovirsen; Gilead to Acquire CymaBay Therapeutics;...
- FDA’s No to Intercept’s NASH Drug; Roche’s Phesgo gets approval; Gilead’s Remdesivir pricing
- Sanofi’s Venglustat Gains US Breakthrough Therapy Nod, Boosting Hope for Type 3 Gaucher Disease; ...
- Gilead’s Livdelzi FDA Approval for Primary Biliary Cholangitis; Incyte and Syndax’s Niktimv...
- PPAR and NOX Agonist: Savior of Underserved Primary Biliary Cholangitis Patients




