Astellas Pharma
Oct 11, 2018
Copiktra receives approval; Lilly wins approval; Eisai’s Fycompa; Astellas’ Roxadustat; Janssen’s Esketamine Nasal Spray
SNAPSHOTS: Copiktra receives FDA approval for CLL and FL, Relief for tazemetostat developers after FDA lifted partial clinical hold, Conference highlights from 19th WCLC The United States Food and Drug Administration (USFDA) has granted approval to Verastem’s Copiktra (duvelisib), for the treatment of patients with...
Read More...
Feb 10, 2017
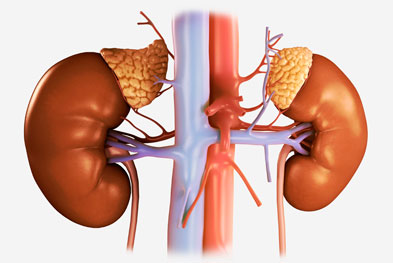
Chronic Kidney Disease: Complex Debilitating Condition
Chronic Kidney Disease (CKD) is a major global public health problem which progresses slowly and get worse over time. It is the 8th leading cause of death in United States, and is characterized by kidney damage and reduced kidney function. According to National Kidney Foundation, approximately 26 millions of adults ...
Read More...
Nov 03, 2016
Medivir & Karo Pharma’s deal; Astellas acquires Ganymed; Celldex Acquires Kolltan; Allergan to Acquire Motus Therapeutics
Medivir entered into an agreement with Karo Pharma to sell its subsidiary BioPhausia Medivir is set to sell is subsidiary, BioPhausia, to Karo Pharmafor for Skr908m ($100.9m). The agreement shows that this price will be on a cash and debt free basis, including a normalized working capital, with the transaction expec...
Read More...


-Agonist.png)

