The Next Wave of Radioligand Therapies: 5 Candidates to Watch
Mar 20, 2026
Summary
- Radioligand therapy is quickly establishing itself as a transformative force in oncology, with demand accelerating so rapidly that manufacturers are under pressure to scale up production.
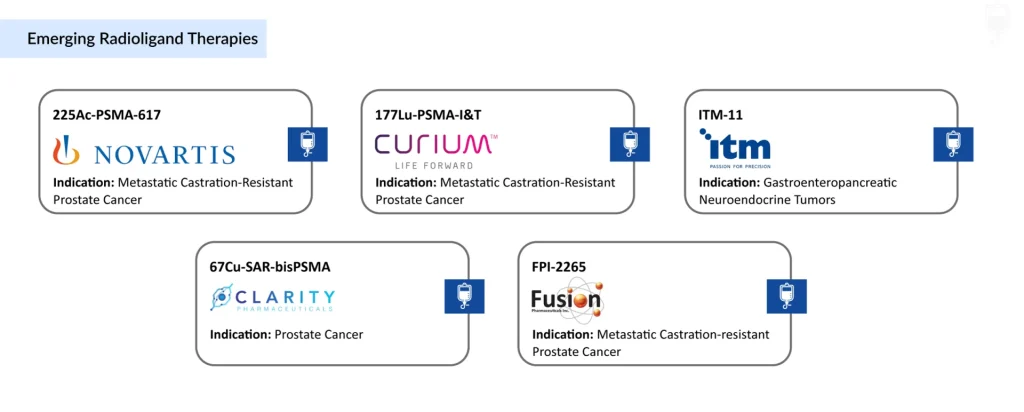
- Among the leaders, Novartis is advancing its late-stage candidate 225Ac-PSMA-617, while a growing competitive landscape is taking shape.
- Companies such as Curium Pharma (177Lu-PSMA-I&T), ITM Isotope Technologies (ITM-11), Fusion Pharmaceuticals (FPI-2265), and Clarity Pharmaceuticals (67Cu-SAR-bisPSMA) are progressing promising assets through mid- to late-stage development.
- Key players, including Bayer, ARTBIO, Convergent Therapeutics, Perspective Therapeutics, PRECIRIX, Ariceum Therapeutics, and Nuclidium, are also driving innovation across prostate cancer, breast cancer, and a broad range of solid tumors.
Radioligand therapy is rapidly emerging as a true game-changer in oncology, so much so that manufacturers are racing against the clock to keep up with demand. This urgency underscores its powerful, transformative potential in reshaping cancer care.
Traditional treatment approaches often fall short for patients with rare, treatment-resistant, or metastatic cancers. Radioligand therapy is stepping in to bridge this gap, offering a promising, life-enhancing option for those with limited alternatives. By delivering precision-targeted treatment, it is redefining the future of personalized cancer care.
Downloads
Click Here To Get the Article in PDF
Recent Articles
- B7-H3, an emerging immune checkpoint molecule in metastatic CRPC and other cancers
- Edwards’s Pascal Precision System; Abbott’s New Spinal Cord Stimulation Device; Imagin to A...
- Early but strong clinical data of Arvinas’s ARV-110 in Men with mCRPC that support a potential pa...
- Notizia
- Phase III RUBY Trial of Jemperli Plus Chemotherapy Updates; FDA Approves Roche’s Vabysmo for RVO;...
Sadaf Javed, Manager of Forecasting at DelveInsght, notes that the currently approved systemic radiotherapeutics are primarily Lu-177–based beta-emitting radioligand therapies, notably PLUVICTO and LUTATHERA. Novartis remains the dominant commercial player and a major purchaser of Lu-177 to support these products and its expanding radioligand therapy pipeline, as per Javed. However, the field is rapidly evolving, with numerous Lu-177–based candidates in development and increasing investment in next-generation alpha-emitting isotopes such as Actinium-225 and Lead-212, Javed further added.
Novartis is pushing the boundaries of radioligand therapy with its late-stage frontrunner 225Ac-PSMA-617. Meanwhile, a competitive wave is building, as players such as Curium Pharma (177Lu-PSMA-I&T), ITM Isotope Technologies (ITM-11), Fusion Pharmaceuticals (FPI-2265), and Clarity Pharmaceuticals (67Cu-SAR-bisPSMA) advance promising candidates into mid- to late-stage development.
Let’s dive deep into the assessment of these 5 promising emerging radioligand therapies poised to make a meaningful impact in prostate cancer and neuroendocrine tumors.
Novartis’ 225Ac‑PSMA‑617
Phase III
Building on the success of beta‑emitting PSMA‑617, Novartis and academic collaborators are advancing 225Ac‑PSMA‑617 as an alpha‑emitting radioligand therapy for advanced prostate cancer. Actinium‑225 delivers a highly potent, short‑range alpha radiation payload that can intensify tumor cell killing while limiting collateral damage to surrounding tissues.
Early clinical experience suggests that 225Ac‑PSMA‑617 can achieve deep prostate‑specific antigen (PSA) responses, including ≥50% reductions in PSA in the majority of treated patients, with manageable toxicity characterized by low‑grade xerostomia. Notably, 225Ac‑PSMA‑617 has also been explored as a “salvage” or escalation option following 177Lu‑PSMA‑617 failure, where combination or sequential approaches have produced encouraging disease control rates and a favorable benefit–risk profile. As more robust randomized data emerge, 225Ac‑PSMA‑617 could redefine how clinicians sequence beta‑ and alpha‑based PSMA therapies in mCRPC.
Curium Pharma’s 177Lu-PSMA-I&T
Phase III
Curium’s 177Lu-PSMA-I&T is rapidly emerging as a frontrunner among next-generation PSMA-targeted radioligand therapies for metastatic castration-resistant prostate cancer—and it’s now advancing into pivotal territory in China. With the Centre for Drug Evaluation (CDE) accepting Curium Shanghai’s IND application, the path is clear for a registrational Phase 3 bridging study in Chinese patients.
This open-label, randomized trial will evaluate 177Lu-PSMA-I&T against standard hormone therapy in ~60 patients, building on robust global data, including insights from the Phase 3 ECLIPSE study. If successful, this effort could accelerate access to cutting-edge PSMA-targeted RLT in China and reinforce lutetium-based therapies as a cornerstone in advanced prostate cancer treatment.
Further strengthening its position, Curium Pharma completed the acquisition of Eczacıbaşı-Monrol’s nuclear business in March 2025, significantly boosting its Lu-177 manufacturing capabilities to support rising global demand and upcoming launches in prostate cancer and neuroendocrine tumor indications.
ITM Isotope Technologies’ ITM-11
Phase III
ITM‑11 (n.c.a. 177Lu‑edotreotide) is an innovative targeted radionuclide therapy designed for patients with gastroenteropancreatic neuroendocrine tumors (GEP‑NETs), a heterogeneous group of typically late‑diagnosed tumors with limited systemic options. ITM‑11 couples the somatostatin analog edotreotide (DOTATOC) with no‑carrier‑added lutetium‑177, delivering beta radiation directly to somatostatin receptor–positive tumor cells while minimizing exposure to normal tissues.
ITM‑11 has demonstrated a favorable safety profile and promising efficacy, including significantly extended progression‑free survival and high tumor‑to‑kidney ratios in earlier studies, which earned orphan designation for GEP‑NETs treatment. The agent is now in two Phase 3 trials: COMPETE for Grade 1–2 GEP‑NETs and COMPOSE for well‑differentiated high‑grade disease. It is supported by positive COMPETE results and an accepted FDA New Drug Application (NDA), with a Prescription Drug User Fee Act (PDUFA) goal date of August 28, 2026. If approved, ITM‑11 could become a new standard RLT option across a broad spectrum of GEP‑NET patients.
Fusion’s FPI-2265
Phase II
Fusion Pharmaceuticals’ FPI‑2265 is an actinium‑225–based PSMA‑targeted radioconjugate that exemplifies the field’s shift toward alpha‑emitting RLTs in prostate cancer. Targeting PSMA with a 225Ac payload, FPI‑2265 is being evaluated in patients with mCRPC in the Phase 2 TATCIST trial and in the Phase 2/3 AlphaBreak registrational program.
The TATCIST and AlphaBreak trials are designed to include both PSMA‑RLT–naïve patients and those previously treated with 177Lu‑based PSMA agents such as PLUVICTO, positioning FPI‑2265 as a potential option across multiple lines of therapy. Early data from TATCIST and the initiation of the AlphaBreak trial underscore the promise of alpha‑based PSMA targeting to deliver more potent, focused radiation to resistant disease while maintaining a manageable safety profile. As Fusion builds out its manufacturing and actinium‑225 supply infrastructure, FPI‑2265 could emerge as a leading alpha RLT in the mCRPC landscape.
Clarity’s 67Cu-SAR-bisPSMA
Phase II
Clarity Pharmaceuticals’ 67Cu‑SAR‑bisPSMA brings a distinct copper‑based theranostic platform to the PSMA RLT arena. The agent is being investigated in SECuRE, a US Phase I/IIa trial in mCRPC, where patients receive escalating single doses of 67Cu‑SAR‑bisPSMA after diagnostic imaging with 64Cu‑SAR‑bisPSMA. Initial cohorts at 4, 8, and 12 GBq have shown no dose‑limiting toxicities and compelling signals of antitumor activity, including marked reductions in PSMA PET uptake and PSA declines.
In one notable SECuRE case, a patient receiving two cycles of 8 GBq achieved a complete response by RECIST, with PSA falling from 47.2 ng/mL to undetectable levels and complete resolution of PSMA uptake on 64Cu‑SAR‑bisPSMA PET imaging. Reflecting the strength of early data and the high unmet need in mCRPC, the FDA has granted Fast Track Designation to 67Cu‑SAR‑bisPSMA, the third Fast Track for Clarity’s SAR‑bisPSMA platform within six months, supporting a strategy to move the product into large pre‑chemotherapy mCRPC populations and combinations with androgen receptor pathway inhibitors. Coupled with Clarity’s proprietary SAR chelator technology and streamlined copper‑64/67 supply model, 67Cu‑SAR‑bisPSMA could help address the scalability challenges that have historically constrained PSMA‑RLT access.
Beyond the frontrunners, a strong pipeline is rapidly taking shape across the radioligand therapy landscape. Industry players like Bayer (225Ac-PSMA-Trillium, 225Ac-Pelgifatamab, 225Ac-GPC3), ARTBIO (212Pb-NG001), Convergent Therapeutics (CONV01-α), Perspective Therapeutics (VMT-α-NET, VMT01, PSV359), PRECIRIX (CAM-FAP-Ac-225), Ariceum Therapeutics (225Ac-SSO110, ATT001), and Nuclidium (NU101, NU201) are all advancing innovative candidates across prostate cancer, breast cancer, and a wide spectrum of solid tumors.
In conclusion, these emerging RLTs highlight how rapidly the field is evolving, from beta to alpha emitters, from lutetium to copper, and from late‑line salvage therapy toward earlier disease settings and broader tumor types. As pivotal data readouts and regulatory milestones approach over the next few years, oncologists may soon have a much richer, more personalized toolbox of radioligand options to match individual tumor biology, treatment history, and logistical realities.

Downloads
Article in PDF
Recent Articles
- Edwards’s Pascal Precision System; Abbott’s New Spinal Cord Stimulation Device; Imagin to A...
- Prostate Cancer Market Experiences an Influx of the Pharma Players Veering the Market Ahead
- Clover’s SCB-1019 RSV Revaccination Study Clears U.S. IND; FDA Approves AMVUTTRA for ATTR-CM CV R...
- Notizia
- CEPI Grants $41.3 Million to Valneva; Innovent Achieves Phase III Success for Mazdutide; GSK’s BL...