SOTYKTU Expands Its Reach as Bristol Myers Squibb Lands Key Approval in Psoriatic Arthritis
Mar 13, 2026
Summary
- Bristol Myers Squibb’s SOTYKTU has received FDA approval for adults with active psoriatic arthritis, becoming the first and only TYK2 inhibitor authorized for this indication.
- The drug extends its reach beyond its 2022 approval for moderate-to-severe plaque psoriasis, reinforcing BMS’s bid to build a multi‑indication oral immunology platform.
- Approval is based on two pivotal Phase 3 trials, POETYK PsA‑1 and PsA‑2, showing statistically significant improvements in ACR20 and other key PsA endpoints versus placebo, with a safety profile consistent with previous studies.
Bristol Myers Squibb has just scored a pivotal win in immunology: the FDA has approved SOTYKTU (deucravacitinib), an oral, selective tyrosine kinase 2 (TYK2) inhibitor, for the treatment of adults with active psoriatic arthritis (PsA). This makes SOTYKTU the first and only TYK2 inhibitor approved for this indication, extending its reach beyond its original 2022 approval in moderate-to-severe plaque psoriasis. Approval is based on the pivotal Phase 3 POETYK PsA‑1 and POETYK PsA‑2 trials, where once‑daily SOTYKTU 6 mg achieved statistically significant improvements in American College of Rheumatology 20% response (ACR20) and other disease measures versus placebo, with a safety profile broadly consistent with its psoriasis program.
Psoriatic arthritis is a chronic, immune‑mediated inflammatory disease characterized by peripheral arthritis, enthesitis, dactylitis, and skin and nail psoriasis, leading to pain, stiffness, functional impairment, and reduced quality of life. Up to approximately 30% of patients with psoriasis will develop PsA, and many present with multi‑domain disease affecting both joints and skin. Despite the availability of TNF inhibitors, IL‑17 and IL‑23 inhibitors, PDE4 inhibition and JAK inhibitors, there remains a clear need for additional effective, convenient oral therapies, particularly options that combine robust efficacy with a differentiated safety profile versus JAK inhibitors.
Downloads
Click Here To Get the Article in PDF
Recent Articles
- Giant-Cell Arteritis Treatment Beyond Glucocorticoids: Exploring Horizons
- Vitiligo Treatment Drug Pipeline Accelerates with Late-Stage Immune-Targeting Therapies
- Merck’s Gefapixant; Pfizer’s Somatrogon; Gilead’s Viklury; AbbVie’s Skyrizi; Gilead’s...
- Novartis gets CAR-T drug; Shire aims to block; Xeljanz stands; Payer snubs; Lawmakers pass a bill
- Celltrion Announces FDA Nod for New STEQEYMA Presentation to Broaden Pediatric Use; CSL’s ANDEMBR...
Rheumatologists surveyed ahead of the decision indicated that they viewed an oral TYK2 inhibitor as an incremental but meaningful addition to the PsA armamentarium rather than a biologic‑displacing disruptor, reinforcing that residual unmet need is focused on patient preference, convenience, and safety trade‑offs within an already highly treated population.
Deucravacitinib is a highly selective TYK2 inhibitor that binds the regulatory domain allosterically, rather than the active domain targeted by traditional JAK inhibitors. Through TYK2, it modulates signaling of interleukin‑23, interleukin‑12, and type I interferons, cytokine pathways central to the pathogenesis of plaque psoriasis and PsA.
This allosteric mechanism underpins its high selectivity for TYK2 and lack of measurable inhibition of JAK1, JAK2, or JAK3 at clinically relevant concentrations in preclinical work, a differentiation that supports the narrative of a potentially more favorable safety profile compared with pan‑JAK inhibitors. Surveyed rheumatologists were more likely to associate TYK2 inhibition with an improved safety profile versus JAKs, even while remaining cautious about whether efficacy would exceed established JAK inhibitors in PsA.
SOTYKTU was first approved by the FDA in 2022 for adults with moderate‑to‑severe plaque psoriasis who are candidates for systemic therapy or phototherapy, and has since gained approvals for this indication in multiple global markets, supported by more than five years of safety and efficacy data and over 20,000 patient‑years of exposure. Building on that foundation, BMS submitted a supplemental New Drug Application (sNDA) in PsA, with the FDA assigning a Prescription Drug User Fee Act (PDUFA) goal date of March 6, 2026; parallel applications were accepted in China, Japan, and Europe.
The new approval for adults with active PsA follows that review and is explicitly positioned as the first TYK2 inhibitor authorized for this disease, confirming BMS’ strategy of turning SOTYKTU into a multi‑indication, first‑in‑class oral immunology platform therapy.
The PsA development program comprises two large, multicenter, randomized, double‑blind, placebo‑controlled Phase 3 trials: POETYK PsA‑1 (IM011‑054) and POETYK PsA‑2 (IM011‑055). Both enrolled adults with active PsA meeting CASPAR criteria, with at least three swollen and three tender joints, and current or historical plaque psoriasis.
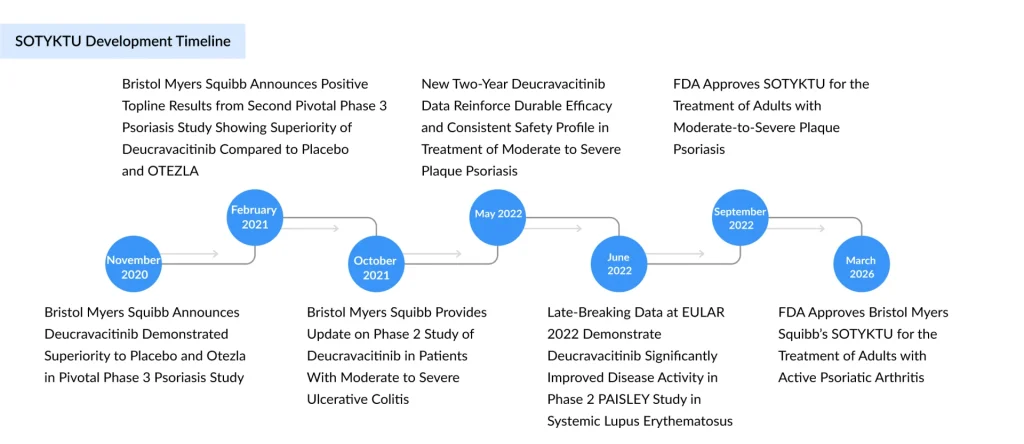
POETYK PsA‑1 enrolled about 670 patients who were biologic DMARD‑naïve, while POETYK PsA‑2 enrolled roughly 624–730 patients who were either bDMARD‑naïve or had prior exposure to TNF inhibitors, reflecting a more heterogeneous, real‑world‑like population. In both trials, patients received deucravacitinib 6 mg once daily or placebo, with a 16‑week placebo‑controlled period followed by active treatment up to 52 weeks; PsA‑2 additionally included an apremilast arm as a safety reference without formal efficacy comparison.
The primary endpoint in both trials was the proportion of patients achieving ACR20 at Week 16, with key secondary endpoints spanning minimal disease activity (MDA), skin responses such as PASI‑75, and patient‑reported outcomes including the Health Assessment Questionnaire‑Disability Index (HAQ‑DI) and SF‑36 measures.
Across both POETYK PsA‑1 and PsA‑2, significantly more patients receiving SOTYKTU achieved ACR20 at Week 16 than those on placebo. In POETYK PsA‑1, approximately 54% of patients on deucravacitinib reached ACR20 versus about 34% with placebo; in POETYK PsA‑2, ACR20 responses were again around 54% for SOTYKTU compared with roughly 39% for placebo, with p‑values in both studies indicating strong statistical significance.
Higher thresholds of response, including ACR50 and ACR70, numerically favored SOTYKTU as well, although these endpoints were not always multiplicity‑adjusted, and thus are reported as supportive rather than confirmatory efficacy evidence. Importantly, a greater proportion of patients achieved minimal disease activity on SOTYKTU versus placebo, reinforcing the drug’s ability to drive multi‑domain disease control in both joint and skin manifestations.
The PsA market is mature, with most patients under rheumatologist care already receiving a biologic or JAK inhibitor, and new entrants gaining share primarily through brand‑to‑brand switching rather than expansion of the treated population. In this context, SOTYKTU is expected to compete most directly with other advanced oral therapies, particularly JAK inhibitors such as XELJANZ and RINVOQ, and the PDE4 inhibitor OTEZLA, rather than with injectable biologics.
Sadaf Javed, Manager of Forecasting and Analytics at DelveInsight, viewed SOTYKTU as a “substantial advance” for PsA, driven largely by its oral route and perceived safety differentiation from JAK inhibitors, although most still characterized TYK2 inhibition as an incremental rather than disruptive innovation at this stage.
Javed also pointed out that the PsA indication is a critical lever to expand SOTYKTU’s revenue base beyond plaque psoriasis, where uptake to date has lagged initial blockbuster ambitions. In 2025, SOTYKTU generated approximately 291 million dollars in sales, a 19% increase versus 2024 but still modest relative to its long‑term potential, underscoring the need for label expansion and broader rheumatology penetration.
The new indication not only opens access to a substantial PsA population but also enhances BMS’ broader positioning in rheumatology, a specialty where the company historically has had a smaller footprint compared with some peers. Success in PsA is also expected to support future launches in systemic lupus erythematosus and Sjögren’s disease, both areas where deucravacitinib is in clinical development.
The FDA approval of SOTYKTU for adults with active PsA marks a key milestone in the evolution of TYK2 inhibition from a dermatology‑focused concept to a broader rheumatologic platform, validating BMS’ multi‑indication investment strategy around deucravacitinib. Over the next several years, the brand’s trajectory will depend on its ability to demonstrate sustained, real‑world effectiveness and safety, carve out a distinct role versus JAK inhibitors and biologics, and secure favorable payer positioning in a crowded class.
Competitive pressure is expected to intensify, particularly from Takeda’s zasocitinib (TAK‑279) and other emerging TYK2 agents that may pursue best‑in‑class claims based on efficacy, safety, or dosing convenience. Head‑to‑head or indirect comparative data, alongside post‑marketing safety experience, will likely shape prescriber preferences among TYK2 inhibitors and determine whether SOTYKTU can maintain its first‑in‑class advantage.
For now, however, the approval provides clinicians and patients with a new, once‑daily oral option that targets validated pathogenic pathways through a novel, allosteric mechanism, with robust Phase 3 data supporting improvements in joints, skin, function, and quality of life in PsA. If BMS executes effectively on medical education, real‑world evidence generation, and market access, the PsA label expansion could mark the inflection point needed for SOTYKTU to transition from an underperforming launch to a cornerstone asset in the company’s immunology portfolio.

Downloads
Article in PDF
Recent Articles
- Zevra’s MIPLYFFA Niemann-Pick Disease Approval; FASENRA Approved for Eosinophilic Granulomatosis;...
- Vitiligo Treatment Drug Pipeline Accelerates with Late-Stage Immune-Targeting Therapies
- Celltrion Announces FDA Nod for New STEQEYMA Presentation to Broaden Pediatric Use; CSL’s ANDEMBR...
- Eli Lilly and Company to Acquire Ajax Therapeutics; Sun Pharma Moves to Acquire Organon in Defini...
- Giant-Cell Arteritis Treatment Beyond Glucocorticoids: Exploring Horizons




