Pulmonary Arterial Hypertension market forecast
Jul 08, 2025
Merck’s WINREVAIR Granted FDA Priority Review for Pulmonary Arterial Hypertension; KalVista’s EKTERLY Approved by FDA as First Oral On-Demand Therapy for Hereditary Angioedema; Fujifilm’s FF-10832 Receives FDA Orphan Drug Designation for Biliary Tract Cancer; Mustang Bio’s MB-101 Granted FDA Orphan Drug Designation for Glioblastoma and Astrocytomas; Denali’s Tividenofusp Alfa Accepted for FDA Priority Review for Hunter Syndrome
Merck's WINREVAIR Gets FDA Priority Review for Pulmonary Arterial Hypertension Merck, also known as MSD outside the U.S. and Canada, announced that the FDA has accepted and granted Priority Review to the supplemental Biologics License Application (sBLA) for WINREVAIR (sotatercept-csrk). The application seeks to ...
Read More...
May 17, 2019
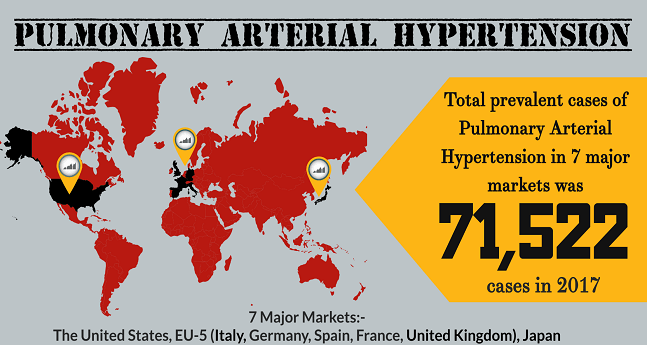
Pulmonary Arterial Hypertension Therapies
Pulmonary Arterial Hypertension (PAH) is a chronic, life-alarming disease that advances over time. It is a rare disease characterized by the increased blood pressure in the arteries of the lungs. It is somehow different from normal high pressure in a way that blood pressure rises only in the arteries that carry blo...
Read More...

-Agonist.png)

