Tenosynovial Giant Cell Tumor Treatment Landscape: A Dual-Modality Race Targeting CSF1R
Apr 03, 2026
Table of Contents
Summary
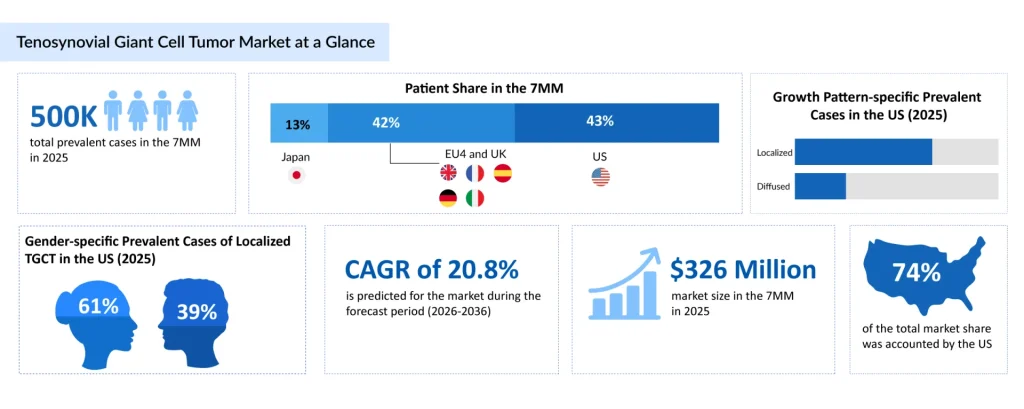
- According to DelveInsight’s estimates, nearly 580,000 prevalent cases of TGCT were reported across the 7MM in 2025, with approximately 460,000 cases classified as localized TGCT and nearly 120,000 as diffuse TGCT; these subtypes differ notably in clinical presentation, recurrence rates, and overall disease burden, and the total cases are expected to increase during the forecast period.
- Despite being a rare condition, TGCT has attracted increasing interest from pharmaceutical companies due to its chronic nature and high unmet need, particularly in diffuse cases where surgery alone is often insufficient. Currently, limited systemic treatment options are available. This growing adoption of CSF1R inhibitors signals a shift toward chronic disease management in TGCT, transitioning from episodic surgical intervention to long-term pharmacological control. Although approved therapies remain limited, the expanding pipeline of candidates under clinical evaluation reflects increasing industry confidence and a competitive push to address this gap.
- Historically, TGCT has been treated primarily with surgical synovectomy, using arthroscopic or open techniques, sometimes combined with adjuvant radiotherapy. Localized disease is generally curable with surgery, while diffuse TGCT is associated with high recurrence rates. Open surgical approaches in diffuse disease can lead to increased morbidity, including postoperative stiffness and pain.
- According to DelveInsight’s analysis, the TGCT market size was found to be approximately USD 326 million in the leading markets (the United States, the EU4 (Germany, France, Italy, and Spain), the United Kingdom, and Japan) in 2025, underlining the evolving treatment landscape supported by improved disease awareness and the gradual uptake of novel CSF1R-targeted therapies.
- Key unmet needs in the TGCT landscape include the lack of curative treatment options, high recurrence rates post-surgery, limited long-term safety data for systemic therapies, and the absence of standardized treatment guidelines. Addressing these gaps will be critical for advancing disease management and unlocking the full market potential.
TGCT is a group of neoplastic disorders that involve synovium-lined tendon sheaths, synovial joints, and adjacent soft tissue. They are divided based on localized and diffuse subtypes. TGCT is an abnormal growth of tissue derived from the synovium that causes activation of immune cells, specifically macrophages, leading to the formation of a mass. These tumors are often classified by their growth pattern and location. These pathological distinctions are important because variability in the clinical and biological features of these neoplasms affects treatment. TGCTs are usually benign lesions arising from the tendon sheath. DelveInsight has estimated the tumour localization of diffuse TGCT to be maximum in the knee, with nearly 33,010 cases in 2025 in the United States, which might reach nearly 58,110 cases by 2036.
Downloads
Click Here To Get the Article in PDF
Recent Articles
- Ono’s ROMVIMZA Triumph Sparks Battle with Daiichi Sankyo’s TURALIO in TGCT Space
- EU Approves Galderma’s NEMLUVIO for Atopic Dermatitis and Prurigo Nodularis; GSK’s Penmenvy Wins ...
- Ipsen and Skyhawk Therapeutics Partnership; SynOx Therapeutics’ Phase III Trial; Roche’s Alecensa...
- IDEAYA and Servier Reports Successful Uveal Melanoma Trial; SynOx Therapeutics Highlights Positiv...
- FDA Grants Priority Review to Merck’s Application for KEYTRUDA Plus Padcev; Roche and Carmot Ther...
TGCT most commonly occurs in large joints, with the knee being the most frequently affected site, followed by the hip, ankle, and shoulder. Localized TGCT often involves smaller joints such as the fingers, hands, and wrist, typically arising from the tendon sheath. In contrast, diffuse TGCT more commonly affects larger, weight-bearing joints and is associated with extensive synovial involvement. The distribution of disease is largely driven by synovial tissue presence rather than external risk factors, with no strong geographic variation reported.
TGCT demonstrates a slight female predominance, with women being more frequently affected than men. Clinically, patients typically present with joint pain, swelling, stiffness, and limited range of motion, often leading to functional impairment and reduced quality of life.
Diagnosis of TGCT relies on a combination of clinical assessment and imaging modalities. Magnetic Resonance Imaging (MRI) is the preferred diagnostic tool, enabling visualization of synovial proliferation, joint effusion, and hemosiderin deposition, which are characteristic of the disease. Definitive diagnosis is established through histopathological evaluation following biopsy or surgical resection.
Recent research literature highlights that TGCT is gaining significant attention due to converging scientific and clinical breakthroughs. A key driver is the clear identification of CSF1/CSF1R biology as the central disease mechanism. At the molecular level, TGCT is driven by overexpression of Colony-stimulating Factor 1 (CSF1), often due to gene rearrangements, leading to the recruitment of macrophages that contribute to tumor growth. This underlying biology underscores the importance of accurate diagnosis and targeted therapeutic approaches in the management of TGCT.
| Comparative Analysis of TGCT Management Strategies | ||
| Parameter | Surgery | Systemic Therapies |
| Role in treatment | Primary treatment, especially in localized TGCT | Used in advanced, unresectable, or recurrent TGCT |
| Recurrence risk | High in diffuse TGCT | Aims to control disease progression |
| Administration | Surgical procedure | Long-term pharmacological treatment |
| Cost and resource | High procedural cost | Long-term treatment cost |
| Safety profile | Surgical risks, post-operative complications | Systemic adverse effects (e.g., liver toxicity) |
Why CSF1R became the prime target in the TGCT?
The genetic and cellular underpinnings of TGCT have made CSF1/CSF1R signaling an attractive, druggable node. Overexpression of CSF1 attracts and activates CSF1R‑positive macrophages that populate and sustain the tumor mass, creating a dependency on this ligand–receptor loop.The first wave of systemic agents exploited this vulnerability: CSF1R‑targeting therapies and off‑label tyrosine kinase inhibitors were deployed in advanced or unresectable cases to shrink tumor volume and improve pain, stiffness, and function. More recently, dedicated CSF1R inhibitors, small molecules, and antibody‑based agents alike, have begun to redefine the standard of care for patients who are not candidates for curative surgery or for whom surgery would cause unacceptable morbidity.
Approved CSF1R‑targeted small molecules: TURALIO and ROMVIMZA
TURALIO (pexidartinib, Daiichi Sankyo) was the first systemic therapy specifically approved for TGCT, and it set the template for small‑molecule intervention in this disease. Pexidartinib is an oral small‑molecule inhibitor that targets CSF1R alongside other kinases, including FMS‑like tyrosine kinase 3 and KIT, directly disrupting macrophage‑driven tumor growth.
In the Phase III ENLIVEN trial, pexidartinib achieved an objective response rate of 39.3% versus 0% with placebo in 120 TGCT patients, alongside clear improvements in tumor volume, mobility, physical function, stiffness, and pain. These benefits were tempered by notable hepatic toxicity, elevations in AST, ALT, and bilirubin led to treatment discontinuations and prompted stringent risk‑management measures, but the drug established proof of concept that systemically blocking CSF1R can deliver meaningful and durable responses.
ROMVIMZA (vimseltinib, Ono Pharmaceutical) represents the next generation of oral CSF1R inhibition and underscores the market’s momentum toward finely tuned small molecules. Approved by the US FDA in February 2025 for adults with symptomatic TGCT in whom surgery would worsen functional limitation or cause severe morbidity, ROMVIMZA subsequently secured European Commission approval later that year for a similar patient population.
In contrast, Japan currently lacks approved drug therapies for TGCT, where treatment primarily relies on surgical procedures, along with radiation and systemic approaches to manage the disease. By 2025, TURALIO and ROMVIMZA stood as the only approved systemic TGCT therapies in the US, with ROMVIMZA also available in the EU, while Japan remained reliant on surgical and non‑approved systemic options. Collectively, these agents helped drive the total TGCT market in the 7MM to an estimated USD ~320 million in 2025, with the US accounting for nearly USD 240 million of that value.
The rise of monoclonal antibodies and next‑wave small molecules
The TGCT pipeline now clearly bifurcates into highly selective small‑molecule CSF1R inhibitors and antibody‑based CSF1R antagonists, each bringing distinct pharmacology, dosing formats, and potential positioning across the patient journey.
Abbisko Therapeutics’ Pimicotinib: Refining oral CSF1R blockade
Pimicotinib (ABSK021, Abbisko Therapeutics) is an orally available, highly selective small‑molecule inhibitor of CSF1R designed specifically for macrophage‑driven diseases, including TGCT. By inhibiting CSF1R signaling, pimicotinib aims to reduce macrophage‑mediated tumor proliferation and inflammation while potentially offering a more refined safety and tolerability profile than earlier multi‑target agents.
The program has advanced rapidly: Abbisko’s New Drug Application for pimicotinib in TGCT was formally accepted by the US FDA in January 2026, and the drug is one of the most advanced late‑stage candidates in the TGCT pipeline with expected US approval around 2027.
According to Stuti Mahajan, consulting manager at DelveInsight, pimicotinib is projected to emerge as a key revenue contributor among emerging therapies, reflecting both its oral route and its fit for long‑term systemic disease control.
SynOx Therapeutics’ Emactuzumab: Antibody‑driven CSF1R inhibition
On the biologics front, emactuzumab (SynOx Therapeutics) exemplifies how monoclonal antibodies can be harnessed to modulate the same pathway from a different therapeutic angle. Emactuzumab is described as a potent and selective CSF1R inhibitor with platform potential across macrophage‑driven inflammatory, fibrotic, and neovascular conditions. Administered intravenously, it blocks CSF1R signaling mediated not only by CSF1 but also by IL‑34, offering a broader lever over macrophage activation and survival.
SynOx is evaluating emactuzumab in the Phase III TANGENT study for patients with unresectable localized or diffuse TGCT, directly addressing one of the most difficult‑to‑treat subsets. Patient enrollment for TANGENT was completed in August 2025, and top‑line results are anticipated in the first quarter of 2026, a milestone that could open an antibody‑based systemic option to complement or even compete with oral CSF1R inhibitors.
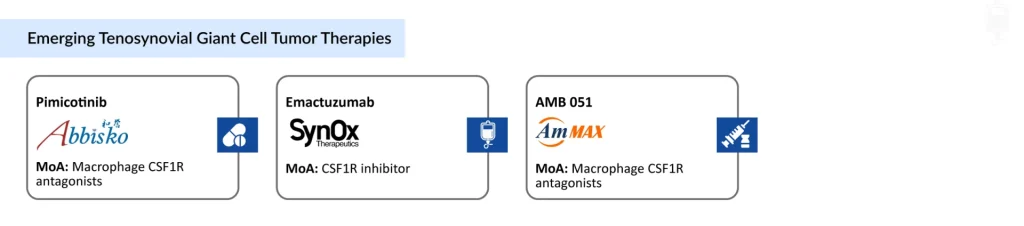
AmMax Bio’s AMB 051: Local CSF1R antagonism inside the joint
AMB 051 (AmMax Bio) highlights a third strategic avenue: local, intra‑articular CSF1R antagonism. Positioned as a macrophage CSF1R antagonist in Phase II development with intra‑articular administration, AMB 051 aims to concentrate its effect within the affected joint while potentially limiting systemic exposure.
If successful, Mahajan said, AMB 051 could carve out a niche between surgery and fully systemic therapy, particularly for patients with localized but recurrent disease where joint‑level control is paramount and repeated resections are undesirable.
Challenges despite pipeline progress in TGCT
While emerging therapies, particularly CSF1/CSF1R inhibitors, are reshaping the treatment landscape of TGCT, most investigational and recently approved options are still positioned as adjunctive or second-line treatments, especially for patients with unresectable, recurrent, or diffuse disease. Surgery continues to remain the primary standard of care, despite its limitations, highlighting the absence of definitive, curative first-line systemic options. Additionally, many novel therapies still require robust long-term safety, durability, and real-world effectiveness data to support broader clinical adoption and consistent regulatory acceptance.
Several critical gaps persist in the broader TGCT landscape
- TGCT often presents with non-specific symptoms and is frequently misdiagnosed, with a significant proportion of patients experiencing delayed diagnosis (>2 years in some cases), complicating early intervention strategies. This highlights a significant opportunity to strengthen early diagnostic pathways, including improved MRI access and streamlined specialist referral networks to enable timely and accurate diagnosis.
- There remains considerable uncertainty in selecting optimal therapy (surgery vs systemic treatment vs multimodal approaches), resulting in inconsistent clinical practices and suboptimal patient outcomes globally. This underscores a key opportunity to develop standardized treatment guidelines and decision-making frameworks to optimize patient outcomes.
- Despite advances in understanding CSF1-driven biology, there is still a lack of reliable biomarkers for disease monitoring, patient stratification, and prediction of treatment response, limiting precision medicine approaches, presenting a critical opportunity to advance biomarker research and enable more precise, personalized treatment strategies.
- Surgical management is associated with high recurrence rates (up to ~50–67% in diffuse TGCT), often necessitating repeat interventions that negatively impact quality of life and increase healthcare burden. This emphasizes the need for durable, disease-modifying therapies that can reduce recurrence and minimize reliance on repeated surgical procedures.
- Variability in patient-reported outcomes (PROs) and clinical endpoints across studies limits cross-trial comparisons and slows the establishment of standardized efficacy benchmarks.
TGCT treatment market outlook for the future
The TGCT treatment approaches are undergoing a gradual but meaningful shift from a predominantly surgery-centric model toward a more diversified and mechanism-driven approach. According to DelveInsight’s analysis, the total market size of TGCT in the 7MM is expected to reach approximately USD 3,330 million by 2036, driven by increased adoption of targeted therapies, rising recurrence in diffuse TGCT, and expanding treatment options.
Key Growth Drivers
- Therapeutic targeting of the CSF1/CSF1R axis, supported by clinical evidence from agents such as pexidartinib and emerging therapies like vimseltinib, has established this pathway as a key driver of TGCT pathogenesis and enabled the development of systemic treatment options for patients with advanced or unresectable disease.
- Frequent post-surgical recurrences continue to expand the addressable population for systemic therapies, highlighting the need for more durable treatment approaches.
- Increasing recognition of long-term joint damage and functional impairment is driving a shift toward earlier intervention and therapies that go beyond symptomatic relief.
- Emerging candidates such as pimicotinib and emactuzumab are strengthening the development landscape, with the potential to improve efficacy, tolerability, and overall patient outcomes.
Key Restraints
- Despite their efficacy, CSF1R inhibitors are associated with notable safety risks, including hepatotoxicity, which necessitates regular monitoring and restricts use in certain patient populations. Uncertainty regarding long-term safety and durability of response further limits physician confidence, particularly in cases where TGCT is not immediately life-threatening.
- The high cost of systemic therapies, combined with the need for continuous treatment and monitoring, creates a significant economic burden for both patients and healthcare systems. Reimbursement limitations and disparities in healthcare infrastructure across markets further restrict access, thereby impacting overall market penetration.
- Surgery remains deeply entrenched as the standard of care, especially in localized TGCT, where outcomes are often curative. Clinician familiarity, established treatment protocols, and immediate symptomatic relief associated with surgical excision contribute to its continued preference. This reliance slows the adoption of newer systemic therapies, particularly in earlier lines of treatment.
- The absence of validated biomarkers for disease monitoring, patient stratification, and prediction of treatment response poses a significant challenge. This limits the ability to identify patients who would benefit most from specific therapies and results in variability in treatment approaches, ultimately hindering the implementation of personalized care strategies.
Future Direction
- The future treatment is expected to move beyond a single-modality approach, with increasing integration of surgery, systemic therapies, and supportive care. Multimodal strategies will likely be tailored based on disease severity and progression, aiming to maximize efficacy while minimizing treatment-related morbidity.
- Advances in understanding TGCT biology and patient heterogeneity are expected to drive more individualized treatment approaches. Factors such as disease subtype (localized vs diffuse), tumor burden, surgical feasibility, and patient preferences will play a greater role in guiding therapy selection and optimizing outcomes.
- As more TGCT therapeutic options become available, defining the optimal sequencing and combination of treatments will become increasingly important. Clinicians will need to balance efficacy, safety, and long-term outcomes to develop treatment algorithms that provide sustained disease control while minimizing adverse effects.
- The entry of new therapies and expanding clinical evidence base are expected to intensify competition within the TGCT market. This will likely lead to improved therapeutic differentiation, pricing dynamics, and broader access, ultimately contributing to a more mature and innovation-driven market strategy across the 7MM.

Frequently Asked Questions
Currently, surgical resection remains the primary treatment for TGCT, particularly in localized disease; however, it is associated with high recurrence rates, especially in diffuse TGCT. Repeated surgeries can lead to joint damage, functional impairment, and increased morbidity. While systemic therapies such as pexidartinib and vimseltinib have improved outcomes in unresectable cases, their use is limited by safety concerns, including hepatotoxicity, and restricted eligibility, highlighting the need for safer and more durable treatment options.
The TGCT market is expected to grow significantly due to increasing disease awareness, improved diagnostic capabilities such as MRI, and the introduction of targeted therapies inhibiting the CSF1/CSF1R pathway. The anticipated launch of emerging therapies like pimicotinib and emactuzumab, along with a strong pipeline from companies such as Daiichi Sankyo and Ono Pharmaceutical, is expected to further drive market expansion during the forecast period (2026-2036).
Despite recent advancements, TGCT management still faces several unmet needs, including limited effective options for recurrent and diffuse disease, lack of long-term safety data for systemic therapies, and challenges in balancing efficacy with tolerability. Additionally, high treatment costs and limited accessibility in certain regions further restrict optimal patient care, emphasizing the need for novel, affordable, and targeted therapies.
Diagnosis of TGCT is primarily based on clinical evaluation, imaging techniques such as MRI, and confirmation through biopsy. However, due to its nonspecific symptoms, such as joint swelling and pain, it is often misdiagnosed or delayed. Differentiating between localized and diffuse forms is critical for treatment planning but can be challenging, leading to potential delays in appropriate intervention.
The future treatment paradigm for TGCT is expected to shift toward a more integrated approach combining surgery with targeted systemic therapies. As newer CSF1R inhibitors and monoclonal antibodies advance through clinical development, treatment strategies will likely focus on reducing recurrence, improving functional outcomes, and minimizing toxicity. Personalized treatment approaches based on disease severity and patient characteristics are also expected to play a key role in optimizing long-term management.
Downloads
Article in PDF
Recent Articles
- FDA Grants Priority Review to Merck’s Application for KEYTRUDA Plus Padcev; Roche and Carmot Ther...
- EU Approves Galderma’s NEMLUVIO for Atopic Dermatitis and Prurigo Nodularis; GSK’s Penmenvy Wins ...
- Ipsen and Skyhawk Therapeutics Partnership; SynOx Therapeutics’ Phase III Trial; Roche’s Alecensa...
- Ono’s ROMVIMZA Triumph Sparks Battle with Daiichi Sankyo’s TURALIO in TGCT Space
- IDEAYA and Servier Reports Successful Uveal Melanoma Trial; SynOx Therapeutics Highlights Positiv...




