Respiratory Care Devices Market Summary
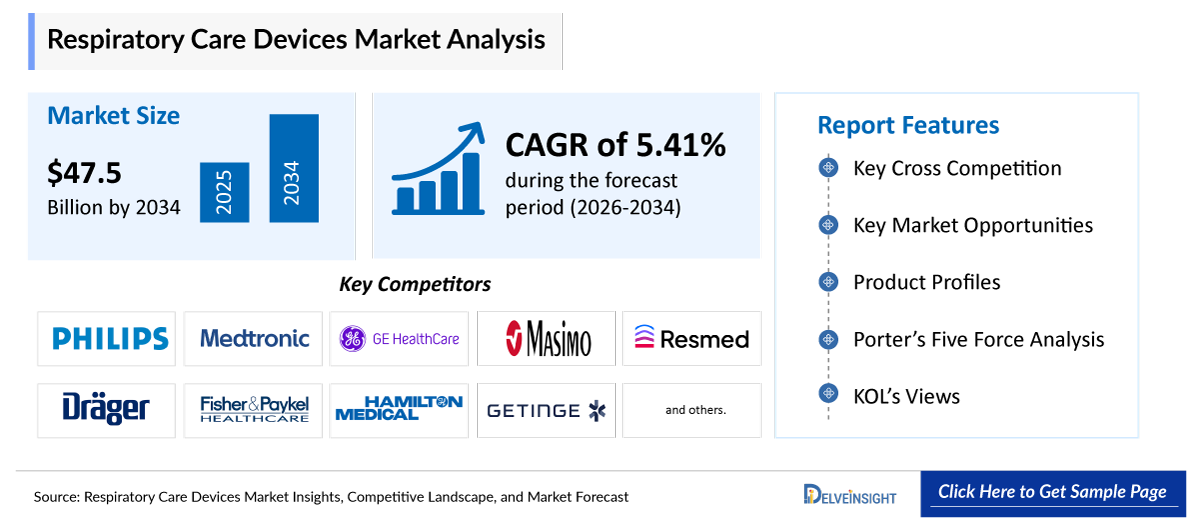
- The Global Respiratory Care Devices Market is expected to increase from ~USD 24,394.06 million in 2025 to USD 47,581.94 million by 2034, reflecting strong and sustained growth.
- The Global Respiratory Care Devices Market is growing at a CAGR of 7.79% during the forecast period from 2026 to 2034.
Respiratory Care Devices Market Insight and Trends
- The Respiratory Care Devices Market is largely propelled by a combination of factors. The growing incidence of chronic respiratory diseases such as COPD, Asthma, Sleep Apnea, and acute respiratory infections has driven greater demand for effective respiratory therapies. In addition, the shift toward home healthcare has increased the need for convenient, continuous respiratory management outside hospital environments. Moreover, continuous innovation and product development in Respiratory Care Devices are further enhancing treatment capabilities and patient outcomes, further supporting market expansion.
- The leading Respiratory Care Devices Companies such as Koninklijke Philips N.V., Medtronic, GE Healthcare, Masimo, ResMed, Dragerwerk AG & CO. KGaA, Fisher & Paykel Healthcare, Hamilton Medical, Getinge AB, Air Liquide Medical Systems, Breas Medical AB, Chart Industries, GaleMed Coproration, OMRON Healthcare, A-M Systems, Invacare Corporation, Vyaire Medical, Polymed, Flexicare (Group) Limited, HomeMedix and others.
- North America is projected to lead the global Respiratory Care Devices Market, driven by several factors. The region has a high prevalence of chronic respiratory conditions such as COPD, asthma, sleep apnea, and acute respiratory infections, which fuels demand for respiratory support devices. Furthermore, the growing transition toward home healthcare settings, driven by patients’ preference to manage chronic respiratory conditions in a comfortable home environment, is accelerating market expansion. Continuous product advancements and strategic innovations by leading companies are also supporting growth by launching technologically advanced, user-friendly Respiratory Care Devices that enhance treatment efficiency and patient convenience.
- In the product type segment of the Respiratory Care Devices market, within therapeutic devices system, ventilators category is estimated to account for the largest market share in 2025.
Request for unlocking the report of the @ Respiratory Care Devices Market
Respiratory Care Devices Market Size and Forecasts | |
|
Report Metrics |
Details |
|
2025 Market Size |
USD 24,394.06 million |
|
2034 Projected Market Size |
USD 47,581.94 million |
|
Growth Rate (2026-2034) |
7.79% CAGR |
|
Largest Market |
North America |
|
Fastest Growing Market |
Asia-Pacific |
|
Market Structure |
Moderately Concentrated |
Factors Contributing to the Growth of the Respiratory Care Devices Market
Increasing prevalence of chronic respiratory conditions such as COPD, asthma, sleep apnea, and acute respiratory infections leading to a surge in Respiratory Care Devices:
The increasing prevalence of chronic respiratory conditions such as chronic obstructive pulmonary disease (COPD), asthma, sleep apnea, and acute respiratory infections is significantly driving the demand for Respiratory Care Devices. As the number of patients requiring long-term oxygen therapy, ventilation support, inhalation therapy, and sleep monitoring continues to rise, healthcare facilities and homecare settings are increasingly adopting devices such as ventilators, nebulizers, oxygen concentrators, and CPAP/BiPAP machines. Additionally, growing air pollution levels, smoking habits, aging populations, and recurrent viral outbreaks further contribute to the expanding patient pool, thereby accelerating the overall growth of the Respiratory Care Devices market.
Increasing preference for home healthcare escalating the market of Respiratory Care Devices:
The increasing preference for home healthcare is significantly boosting the Respiratory Care Devices market, as more patients seek convenient, cost-effective, and long-term management solutions outside of hospitals. The growing prevalence of chronic respiratory diseases such as COPD and asthma, along with the rising aging population, has led to higher demand for portable oxygen concentrators, nebulizers, CPAP devices, and home ventilators. Advances in compact, user-friendly, and connected respiratory devices further support remote monitoring and improved patient compliance, thereby accelerating market growth.
Increase in product development activities:
The increase in product development activities is significantly boosting the Respiratory Care Devices market, as manufacturers are introducing advanced, portable, and technologically enhanced solutions to meet rising patient needs. Innovations such as smart ventilators, connected oxygen concentrators, AI-enabled monitoring systems, and improved CPAP devices are enhancing treatment efficiency, patient comfort, and remote monitoring capabilities. Continuous research and development efforts, along with product upgrades and new launches, are expanding clinical applications and improving outcomes, thereby driving overall market growth.
Respiratory Care Devices Market Report Segmentation
This Respiratory Care Devices market report offers a comprehensive overview of the global Respiratory Care Devices market, highlighting key trends, growth drivers, challenges, and opportunities. It covers detailed market segmentation by Product Type (Therapeutic Devices {Ventilators [Invasive Ventilators and Non-Invasive Ventilators], Positive Airway Pressure (PAP) Devices [Continuous Positive Airway Pressure (CPAP) Devices, Bilevel PAP (BPAP) Devices, and Automatic positive airway pressure (APAP) devices], Nebulizers [Jet Nebulizers, Ultrasonic Nebulizers, and Mesh Nebulizers], and Others}, Monitoring & Diagnostic Devices {Spirometers [Table Top and Portable], Pulse Oximeters [Fingertip Pulse Oximeters, Handheld Pulse Oximeters, Benchtop Pulse Oximeters, and Wearable Pulse Oximeters], Capnography Devices [Side Stream Capnography Devices, Main Stream Capnography Devices, and Micro Stream Capnography Devices], Polysomnography Devices [Stationary Devices and Portable Devices], and Others}, Airway Management Devices {Endotracheal Tubes, Tracheostomy Tubes, and Others}, and Consumables & Accessories), End-Users (Hospitals and Clinics, Ambulatory Surgical Centers, Homecare Settings and Others), and geography. The report provides valuable insights into the competitive landscape, regulatory environment, and market dynamics across major markets, including North America, Europe, and Asia-Pacific. Featuring in-depth profiles of leading industry players and recent product innovations, this report equips businesses with essential data to identify market potential, develop strategic plans, and capitalize on emerging opportunities in the rapidly growing Respiratory Care Devices market.
Respiratory Care Devices are medical devices designed to diagnose, monitor, and treat disorders related to the respiratory system, helping patients maintain adequate breathing and lung function. These devices are used in hospitals, clinics, emergency settings, and home healthcare to manage conditions such as asthma, Chronic Obstructive Pulmonary Disease (COPD), sleep apnea, and respiratory failure. Common examples include ventilators, oxygen concentrators, nebulizers, inhalers, CPAP/BiPAP machines, and spirometers.
The Respiratory Care Devices Market is propelled by several interrelated factors that collectively strengthen its growth trajectory. A major driver is the rising global burden of chronic respiratory disorders such as chronic obstructive pulmonary disease, asthma, sleep apnea, and acute respiratory infections. The long-term and often progressive nature of these conditions necessitates continuous monitoring, oxygen therapy, ventilatory assistance, and airway management, thereby increasing reliance on advanced respiratory support technologies.
In parallel, the growing preference for home-based treatment is reshaping care delivery models, as patients seek convenient, cost-effective, and comfortable management options outside hospital settings. This trend has further accelerated demand for portable, compact, and user-friendly devices such as home ventilators, CPAP machines, and oxygen concentrators. Additionally, continuous technological advancements including smart connectivity features, improved battery life, enhanced filtration systems, and better patient-interface designs are improving treatment adherence and clinical outcomes. Together, these factors are expanding the patient pool, encouraging healthcare providers to adopt innovative solutions, and driving sustained global market expansion of Respiratory Care Devices during the forecast period from 2026 to 2034.
Get More Insights into the Report @ Respiratory Care Devices Market
What are the latest Respiratory Care Devices Market Dynamics and Trends?
The global Respiratory Care Devices market has witnessed strong growth in recent years, largely due to the increasing burden of chronic respiratory diseases such as chronic obstructive pulmonary disease, asthma, and sleep Apnea. Moreover, the growing preference for home-based care and continuous technological advancements in device design are further improving treatment effectiveness, enhancing portability, and increasing overall patient comfort.
According to the World Health Organization (2025), global cases of chronic obstructive pulmonary disease were projected to increase by 23% worldwide by 2050. Moreover, as per the same source (2024), tobacco smoking accounted for more than 70% of chronic obstructive pulmonary disease cases in high-income countries. Patients with COPD often need oxygen therapy, non-invasive ventilation, nebulizers, and monitoring devices to manage symptoms such as breathlessness and reduced lung function. Additionally, the increasing number of COPD diagnoses, particularly among the aging population and individuals with a history of smoking or exposure to air pollution, is further driving consistent demand for hospital-based as well as home-based respiratory equipment, thereby escalating the overall Respiratory Care Devices Market.
Moreover the asthma remains another major growth driver. As per the recent data provided by the World Health Organization (2025), more than 250 million people worldwide were living with asthma, highlighting it as a significant global health burden. Patients with Asthma regularly depend on devices such as inhalers, nebulizers, spacers, and peak flow meters to manage airway inflammation, ensuring effective drug delivery, monitoring lung function, and preventing sudden exacerbations. Since asthma requires continuous treatment and periodic monitoring, the consistent use and replacement of these devices significantly increase their demand. This sustained and recurring need for respiratory support equipment directly contributes to the growth and expansion of the Respiratory Care Devices market.
Additionally, the increasing shift toward home-based healthcare is significantly driving growth in the Respiratory Care Devices Market, as patients and providers increasingly prefer convenient, cost-effective treatment options outside hospitals. This trend has led to the introduction and regulatory approvals of several innovative homecare-focused respiratory products. For example, in January 2026, Inogen launched its Aurora™ line of CPAP masks with FDA 510(k) clearance to expand into sleep apnea treatment for home use, improving patient comfort and ease of therapy.
However, the increase in product development activities is further boosting the overall market of Respiratory Care Devices. For instance, in July 2025, Synchrony Medical, a medical technology company focused on advancing respiratory care, received U.S. Food and Drug Administration (FDA) 510(k) clearance to market its LibAirty™ Airway Clearance System in the United States. Thus, the factors mentioned above are expected to boost the overall market of Respiratory Care Devices during the forecast period from 2026 to 2034.
However, the increasing number of product recalls is acting as a significant limiting factor for the Respiratory Care Devices Market, as recalls due to device malfunctions, safety concerns, or quality issues negatively impact manufacturer reputation and consumer trust. Recalls also lead to financial losses, regulatory scrutiny, and temporary product shortages, which hinder market growth. Additionally, the stringent regulatory approval process for Class II and Class III respiratory devices further restrains the market. These devices require extensive clinical testing, strict compliance with safety and performance standards, and lengthy review timelines before approval. The high cost and time involved in meeting regulatory requirements delay product launches and discourage smaller manufacturers from entering the market, thereby limiting overall industry expansion.
Respiratory Care Devices Market Segment Analysis
Respiratory Care Devices Market by Product Type (Therapeutic Devices {Ventilators [Invasive Ventilators and Non-Invasive Ventilators], Positive Airway Pressure (PAP) Devices [Continuous Positive Airway Pressure (CPAP) Devices, Bilevel PAP (BPAP) Devices, and Automatic positive airway pressure (APAP) devices], Nebulizers [Jet Nebulizers, Ultrasonic Nebulizers, and Mesh Nebulizers] and Others}, Monitoring & Diagnostic Devices {Spirometers [Table Top and Portable], Pulse Oximeters [Fingertip Pulse Oximeters, Handheld Pulse Oximeters, Benchtop Pulse Oximeters, Wearable Pulse Oximeters], Capnography Devices [Side Stream Capnography Devices, Main Stream Capnography Devices, Micro Stream Capnography Devices], Polysomnography Devices [Stationary Devices and Portable Devices], Others}, Airway Management Devices {Endotracheal Tubes, Tracheostomy Tubes, and Others}, and Consumables & Accessories), End-Users (Hospitals and Clinics, Ambulatory Surgical Centers, Homecare Settings and Others), and Geography (North America, Europe, Asia-Pacific, and Rest of the World).
By Product Type: Under Therapeutic Devices Segment, Ventilators Category Projected to Register Highest Revenue Share
Within the product type segmentation of the Respiratory Care Devices market, under therapeutic devices segment, ventilators category is projected to dominate, accounting for an estimated 20% market share in 2025. Ventilators play a crucial role in boosting the overall Respiratory Care Devices market, as they represent one of the most essential and high-value segments within respiratory support technologies. Clinically, ventilators are indispensable for managing patients with acute respiratory failure, chronic obstructive pulmonary disease (COPD), severe asthma, pneumonia, ARDS, and post-surgical respiratory complications. The rising incidence of respiratory disorders, growing ICU admissions, increasing premature births requiring neonatal ventilation, and expanding geriatric population have significantly increased demand for both invasive and non-invasive ventilators. Additionally, the shift toward home healthcare has driven adoption of portable and home-use ventilators, further expanding the addressable patient population. Technological advancements such as smart ventilation modes, touchscreen interfaces, integrated monitoring systems, and remote connectivity are improving patient outcomes and encouraging healthcare providers to upgrade existing systems, thereby accelerating market growth.
From a commercial perspective, ventilators are capital-intensive devices with high unit prices, generating substantial revenue compared to other respiratory devices like nebulizers or inhalers. Hospitals and healthcare systems allocate significant budgets for ICU infrastructure, where ventilators are a core component, ensuring consistent procurement demand. Major manufacturers such as Medtronic, Koninklijke Philips N.V., ResMed Inc., GE HealthCare, and Drägerwerk AG dominate this segment, continuously launching upgraded models and expanding global distribution networks. For example, in November 2024, CorVent Medical, a manufacturer of medical devices and mechanical ventilators, announced that its RESPOND ventilator had received U.S. FDA 510(k) clearance.
The ventilator segment also drives recurring revenue streams through accessories, consumables (breathing circuits, filters), service contracts, and maintenance agreements. Furthermore, government investments in pandemic preparedness and critical care infrastructure have strengthened long-term procurement strategies, making ventilators not only a life-saving clinical tool but also a commercially strategic growth engine within the broader Respiratory Care Devices market. As a result, the ventilators category is projected to witness substantial growth within the overall Respiratory Care Devices market throughout the forecast period.
By End-Users: Hospitals and Clinics Category Dominates the Market
Hospitals and clinics hold a dominant position in the Respiratory Care Devices market as they serve as the main centers for diagnosis, emergency treatment, and specialized respiratory care. Patients with acute and chronic respiratory disorders, including chronic obstructive pulmonary disease, asthma, pneumonia, and acute respiratory distress syndrome, often require urgent medical attention, continuous monitoring, and access to advanced therapeutic devices such as ventilators, oxygen delivery systems, and diagnostic monitors resources that are primarily available in hospital and clinical settings. Hospitals are equipped with ICUs and dedicated respiratory care units capable of managing critical and high-risk patients.
The presence of skilled healthcare professionals, including pulmonologists and respiratory therapists, ensures effective use and management of respiratory devices. Additionally, high patient volumes, particularly for emergency care and surgical procedures, contribute to increased device demand. Supportive reimbursement policies, government funding, and substantial procurement budgets further enable hospitals and clinics to acquire advanced, high-cost respiratory equipment, reinforcing their leading role in the market.
Respiratory Care Devices Market Regional Analysis
North America Respiratory Care Devices Market Trends
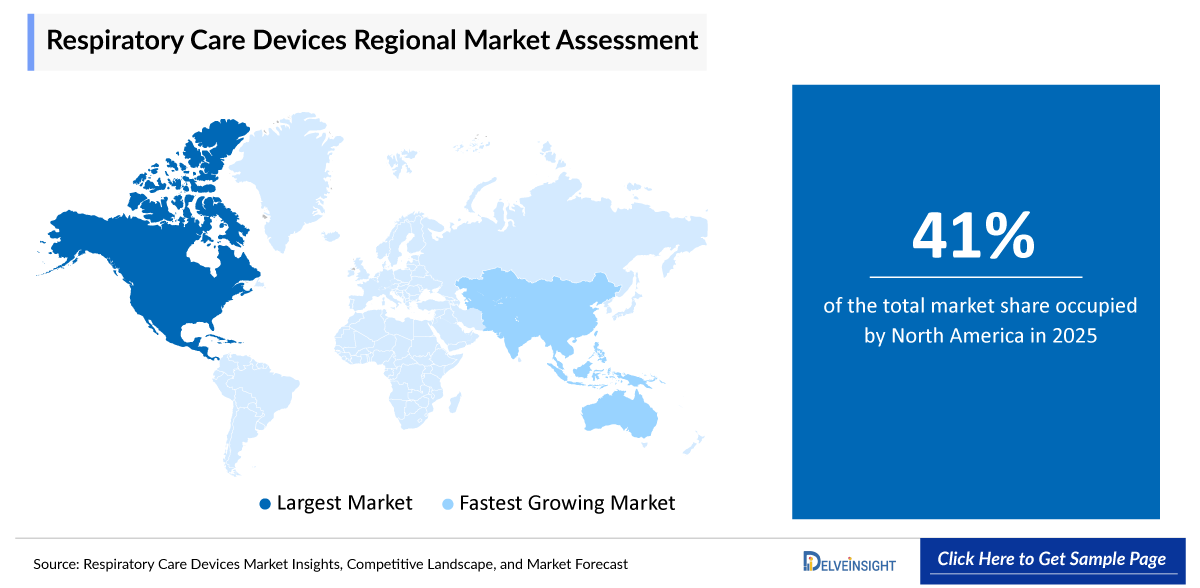
North America is expected to account for the highest proportion of 41% of the Respiratory Care Devices market in 2025, out of all regions. This dominance is driven by rising the prevalence of chronic respiratory conditions such as chronic obstructive pulmonary disease, asthma, sleep apnea, and acute respiratory infections. Additionally, the shift toward home healthcare and ongoing technological innovations in user-friendly, efficient devices are further boosting demand and reinforcing the region’s market dominance.
According to the Asthma and Allergy Foundation of America (2025), over 28 million people in the U.S. had asthma, representing approximately 1 in 12 individuals. Of these, more than 23 million were adults aged 18 and older. Being a chronic respiratory condition affecting millions globally, the disease necessitates ongoing management and effective treatment, driving demand for devices such as inhalers, nebulizers, spirometers, and smart monitoring systems. These devices are essential not only for immediate symptom relief but also for long-term disease control and prevention of attacks, particularly in home care settings, thereby contributing significantly to the growth of the Respiratory Care Devices market.
Moreover, according to the American Medical Association (2025), interrupted breathing during sleep affected an estimated 30 million people in the U.S. Patients with sleep apnea suffer from repeated breathing interruptions during sleep, resulting in reduced oxygen levels and associated health issues such as cardiovascular complications and daytime fatigue. This ongoing need for effective management drives demand for therapeutic devices, including Continuous Positive Airway Pressure (CPAP) machines, Bi-level Positive Airway Pressure (BiPAP) devices, oral appliances, and oxygen therapy systems, thereby contributing significantly to the growth of the Respiratory Care Devices Market.
However, the increase in product development activities across the region is further boosting the overall market of Respiratory Care Devices. For instance, in December 2024, Inogen, Inc., a medical technology company offering innovative respiratory products for use in the homecare setting, announced that it had received U.S. Food and Drug Administration (FDA) 510(k) clearance for the SIMEOX 200 Airway Clearance Device, expanding the company’s ability to market and address the needs of patients with chronic respiratory diseases in the U.S. Thus, all the above-mentioned factors are anticipated to propel the market for Respiratory Care Devices in North America during the forecast period.
Europe Respiratory Care Devices Market Trends
Europe is witnessing strong growth in the Respiratory Care Devices Market due to the rising prevalence of chronic conditions such as COPD, asthma, sleep apnea, and acute respiratory infections. This, combined with a shift toward home healthcare and ongoing product innovation, drives demand for advanced, patient-friendly respiratory solutions.
According to the European Federation of Allergy and Airways Diseases (2025), more than 36 million Europeans were living with chronic obstructive pulmonary disease, with a median prevalence of 3,230 per 100,000 in men and 2,202 per 100,000 in women. Patients rely on devices such as oxygen therapy systems, nebulizers, inhalers, ventilators, and portable monitoring tools to manage symptoms, prevent exacerbations, and maintain daily activities. Thus, the growing number of individuals affected by COPD is steadily increasing the demand for these Respiratory Care Devices, thereby fueling market growth.
Additionally, according to the European Respiratory Society (2024), there were 27 million asthma cases in Western Europe, with the condition affecting an estimated 12% of the population in the UK. Patients use devices such as inhalers, nebulizers, spacers, peak flow meters, and smart monitoring systems to control airway inflammation, prevent sudden asthma episodes, and monitor lung function. The ongoing need for these devices, both for daily management and emergency care, generates steady demand, driving substantial growth in the Respiratory Care Devices market. Hence, all the factors mentioned above are expected to drive the market for Respiratory Care Devices in Europe during the forecast period.
Asia-Pacific Respiratory Care Devices Market Trends
The Asia Pacific region is experiencing robust growth in the Respiratory Care Devices market, driven by the increasing prevalence of chronic respiratory conditions, including COPD, asthma, sleep apnea, and acute respiratory infections.
According to Global Asthma Report (2022), in 2021, about 35 million suffered from asthma in India. Asthma is a significant driver of the Respiratory Care Devices Market due to its widespread prevalence, chronic nature, and need for continuous management. Patients use inhalers, nebulizers, CPAP/BiPAP devices, and monitoring systems to manage symptoms, prevent flare-ups, and maintain lung function, creating steady demand for these devices. The rising incidence of asthma, particularly among children and urban populations, is enlarging the patient base, thereby further driving growth in the Respiratory Care Devices Market.
Additionally, according to the Institute for Health Metrics and Evaluation (2026), in 2023, the age-standardized prevalence rate of Chronic Obstructive Pulmonary Disease (COPD) was highest in South Asia, at 3,044.18 per 100,000 population. Patients with chronic obstructive pulmonary disease frequently face breathing difficulties, decreased lung capacity, and recurring exacerbations, creating a need for therapeutic and monitoring devices such as oxygen therapy systems, nebulizers, inhalers, ventilators, and portable monitoring equipment. This ongoing demand for effective respiratory management is driving growth in the Respiratory Care Devices market.
Hence, all the factors mentioned above are expected to drive the market for Respiratory Care Devices in Asia-Pacific during the forecast period.
Who are the major players in the Respiratory Care Devices Market?
The following are the leading companies in the Respiratory Care Devices market. These companies collectively hold the largest market share and dictate industry trends.
- Koninklijke Philips N.V
- Medtronic
- GE Healthcare
- Masimo
- ResMed
- Drägerwerk AG & Co. KGaA
- Fisher & Paykel Healthcare
- Hamilton Medical
- Getinge AB
- Air Liquide Medical Systems
- Breas Medical AB
- Chart Industries
- GaleMed Corporation
- OMRON Healthcare
- A-M Systems
- Invacare Corporation
- Vyaire Medical
- Polymed
- Flexicare (Group) Limited
- HomeMedix
- Others
How is the competitive landscape shaping the Respiratory Care Devices Market?
The competitive landscape of the Respiratory Care Devices market is increasingly dynamic, driven by technological innovation, strategic collaborations, and geographic expansion. Leading players are focusing on developing advanced, user-friendly devices such as portable ventilators, smart oxygen concentrators, and connected CPAP/BiPAP machines to meet growing patient demand, particularly in home healthcare settings. Companies are also engaging in mergers, acquisitions, partnerships, and licensing agreements to strengthen their market presence and expand product portfolios.
Emerging players and startups are contributing to niche innovations, including AI-enabled respiratory monitoring and wearable respiratory devices, which intensify competition. Additionally, the market is shaped by regulatory approvals and compliance, as obtaining clearances such as FDA 510(k) or CE marks can provide a competitive edge. Overall, the competitive environment is fostering continuous innovation, improved patient-centric solutions, and expanded global reach, making the market highly dynamic and growth-oriented.
Recent Developmental Activities in the Respiratory Care Devices Market
- In July 2025, Synchrony Medical, a medical technology company focused on advancing respiratory care, received U.S. Food and Drug Administration (FDA) 510(k) clearance to market its LibAirty™ Airway Clearance System in the United States.
- In January 2025, Linshom Medical announced that the U.S. Food and Drug Administration had granted 510(k) clearance for the company’s two-piece, wearable respiratory sensor.
- In December 2024, Inogen, Inc., a medical technology company offering innovative respiratory products for use in the homecare setting, announced that it had received U.S. Food and Drug Administration (FDA) 510(k) clearance for the SIMEOX 200 Airway Clearance Device, expanding the company’s ability to market and address the needs of patients with chronic respiratory diseases in the U.S.
- In February 2024, ResMed launched its AirCurve 11 series devices, the company’s latest bilevel devices featuring two levels of support inspiratory positive airway pressure (IPAP) and expiratory positive airway pressure (EPAP) integrated with digital technology to simplify treatment for healthcare providers and help patients initiate and adhere to sleep apnea therapy.
- In July 2023, Dräger announced that it had received 510(k) clearance from the U.S. Food & Drug Administration (FDA) for its Evita V600, V800, and Babylog VN800 devices, designed for mechanical ventilation ranging from adults to premature babies.
|
Report Metrics |
Details |
|
Study Period |
2023 to 2034 |
|
Base Year |
2025 |
|
Forecast Period |
2026 to 2034 |
|
Respiratory Care Devices Market CAGR |
7.79% |
|
Key Companies in the Respiratory Care Devices Market |
Koninklijke Philips N.V., Medtronic, GE Healthcare, Masimo, ResMed, Dragerwerk AG & CO. KGaA, Fisher & Paykel Healthcare, Hamilton Medical, Getinge AB, Air Liquide Medical Systems, Breas Medical AB, Chart Industries, GaleMed Coproration, OMRON Healthcare, A-M Systems, Invacare Corporation, Vyaire Medical, Polymed, Flexicare (Group) Limited, HomeMedix and others. |
|
Respiratory Care Devices Market Segments |
by Product Type, by End-Users, and by Geography |
|
Respiratory Care Devices Regional Scope |
North America, Europe, Asia Pacific, Middle East, Africa, and South America |
|
Respiratory Care Devices Country Scope |
U.S., Canada, Mexico, Germany, United Kingdom, France, Italy, Spain, China, Japan, India, Australia, South Korea, and key Countries |
Respiratory Care Devices Market Segmentation
· Respiratory Care Devices by Product Type Exposure
o Therapeutic Devices
§ Ventilators
§ Invasive Ventilators
§ Non-Invasive Ventilators
§ Positive Airway Pressure (PAP) Devices
§ Continuous Positive Airway Pressure (CPAP) Devices
§ Bilevel PAP (BPAP) Devices
§ Automatic Positive Airway Pressire (APAP) Devices
§ Nebulizers
§ Jet Nebulizers
§ Ultrasonic Nebulizers
§ Mesh Nebulizers
§ Others
o Monitoring & Diagnostic Devices
§ Spirometers
§ Table Top
§ Portable
§ Pulse Oximeters
§ Fingertip Oulse Oximeters
§ Handheld Pulse Oximeters
§ Benchtop Pulse Oximeters
§ Wearable Pulse Oximeters
§ Capnography Devices
§ Side Stream Capnography Devices
§ Main Stream Capnography Devices
§ Micro Stream Capnography Devices
§ Polysomnography Devices
§ Stationary Devices
§ Portable Devices
§ Others
o Airway Management Devices
§ Endotracheal Tubes
§ Tracheostomy Tubes
§ Others
o Consumables & Accessories
· Respiratory Care Devices End-Users Exposure
o Hospitals and Clinics
o Ambulatory Surgical Centers
o Homecare Settings
o Others
· Respiratory Care Devices Geography Exposure
o North America Respiratory Care Devices Market
§ United States Respiratory Care Devices Market
§ Canada Respiratory Care Devices Market
§ Mexico Respiratory Care Devices Market
o Europe Respiratory Care Devices Market
§ United Kingdom Respiratory Care Devices Market
§ Germany Respiratory Care Devices Market
§ France Respiratory Care Devices Market
§ Italy Respiratory Care Devices Market
§ Spain Respiratory Care Devices Market
§ Rest of Europe Respiratory Care Devices Market
o Asia-Pacific Respiratory Care Devices Market
§ China Respiratory Care Devices Market
§ Japan Respiratory Care Devices Market
§ India Respiratory Care Devices Market
§ Australia Respiratory Care Devices Market
§ South Korea Respiratory Care Devices Market
§ Rest of Asia-Pacific Respiratory Care Devices Market
o Rest of the World Respiratory Care Devices Market
§ South America Respiratory Care Devices Market
§ Middle East Respiratory Care Devices Market
§ Africa Respiratory Care Devices Market
Respiratory Care Devices Market Recent Industry Trends and Milestones (2022-2026)
|
Category |
Key Developments |
|
Respiratory Care Devices Product Approvals |
Linshom Medical- Respiratory Sensors (FDA), Synchrony Medical- LibAirty™ Airway Clearance System (FDA), Inogen, Inc.- SIMEOX 200 Airway Clearance Device (FDA). |
|
Acquisition in the Respiratory Care Devices Market |
ResMed acquired VirtuOx and SunMed agreed to acquire the Respiratory Health business from Avanos Medical. |
|
Company Strategy |
Koninklijke Philips N.V- focuses its strategy on strengthening its position in the respiratory care market through innovation, digital health integration, and patient-centric solutions. The company emphasizes developing connected and smart respiratory devices, such as advanced ventilators, CPAP/BiPAP machines, and home respiratory monitoring systems, to improve treatment outcomes and patient convenience. ResMed - focuses on innovating and expanding its portfolio of connected Respiratory Care Devices, including CPAP, BiPAP, ventilators, and cloud-based digital health solutions. The company emphasizes home healthcare solutions and remote patient monitoring to improve treatment adherence and outcomes. |
|
Emerging Technology |
Smart inhalers & wearables, advanced portable devices, advanced nebulization, ai-powered monitoring systems, smart CPAP and BiPAP machines with connected apps, telemedicine integration, and others. |
Impact Analysis
AI-Powered Innovations and Applications:
AI‑powered innovations are transforming the Respiratory Care Devices market by significantly improving patient outcomes, operational efficiency, and cost‑effectiveness. Advanced machine learning algorithms and real‑time data analytics enable devices such as smart ventilators, connected spirometers, and AI‑enhanced CPAP (Continuous Positive Airway Pressure) machines to continuously monitor respiratory parameters, detect anomalies early, and adapt therapy settings dynamically, leading to more personalized and precise care. Integration of predictive analytics helps clinicians anticipate exacerbations in chronic respiratory conditions like COPD (Chronic Obstructive Pulmonary Disease) and asthma, reducing hospital readmissions and enabling proactive interventions. Additionally, cloud‑based platforms and remote monitoring capabilities have expanded telehealth services, allowing healthcare providers to manage patients outside traditional clinical settings and improving access to care in underserved areas. Operationally, AI‑driven workflow automation enhances device management and maintenance, reduces clinician workload, and minimizes human error. However, the adoption of AI in respiratory care also brings challenges, including data privacy concerns, regulatory compliance hurdles, the need for robust interoperability standards, and potential workforce displacement due to automation. Despite these challenges, AI innovations are poised to deliver substantial clinical and economic value, driving growth and shaping the future landscape of the Respiratory Care Devices market.
U.S. Tariff Impact Analysis on Respiratory Care Devices Market:
The introduction and adjustment of U.S. tariffs on imported Respiratory Care Devices have created notable repercussions across the domestic market, influencing costs, supply chains, and competitive dynamics. Tariffs on key components and finished products especially those sourced from major manufacturing hubs in Asia and Europe have elevated procurement costs for U.S. healthcare providers and device manufacturers, leading to higher end‑user prices for ventilators, nebulizers, CPAP machines, and related accessories. This cost inflation pressures hospital budgets and insurers, potentially reducing adoption rates for newer or more advanced technologies. At the same time, tariffs have incentivized some manufacturers to diversify supply chains, accelerate onshoring of production, or shift to tariff‑free materials where possible, bolstering domestic manufacturing resilience. Smaller suppliers, however, often bear disproportionate burdens from increased input costs and disrupted supplier relationships, which can constrain innovation and market entry. Furthermore, trade tensions and tariff uncertainty can dampen investment in the respiratory care sector, as companies hesitate to commit capital amid unpredictable policy environments. While tariffs aim to protect U.S. industry and reduce reliance on foreign sources, they also risk slowing growth, constraining affordability, and reshaping competitive landscapes within the Respiratory Care Devices market.
How This Analysis Helps Clients
- Cost Management: By understanding the tariff landscape, clients can anticipate cost increases and adjust pricing strategies accordingly, ensuring profitability.
- Supply Chain Optimization: Clients can identify alternative sourcing options and diversify their supply chains to reduce dependency on high-tariff regions, enhancing resilience.
- Regulatory Navigation: Expert guidance on navigating the evolving regulatory environment helps clients maintain compliance and avoid potential legal challenges.
- Strategic Planning: Insights into tariff impacts enable clients to make informed decisions about manufacturing locations, partnerships, and market entry strategies.
Startup Funding & Investment Trends
|
Company Name |
Total Funding |
Main Products |
Stage of Development |
Core Technology |
|
VQ Biomedical |
$5 million |
Intravascular Oxygenation Catheter |
Seed round |
Used to provide supplemental oxygen directly into the blood within a vein (typically the vena cava). |
|
Inhaler |
£1.5 million |
Cardboard Dry Powder Inhaler (DPI) |
- |
Uses a pre‑metered dose of dry powder formulation contained in blisters, capsules, or sealed pockets integrated into a rigid or foldable cardboard housing. |
Key Takeaways from the Respiratory Care Devices Market Report Study
- Market size analysis for the current Respiratory Care Devices Market Size (2025), and market forecast for 8 years (2026 to 2034)
- Top key product/technology developments, mergers, acquisitions, partnerships, and joint ventures happened over the last 3 years.
- Key companies dominating the Respiratory Care Devices market.
- Various opportunities available for the other competitors in the Respiratory Care Devices market space.
- What are the top-performing segments in 2025? How these segments will perform in 2034?
- Which are the top-performing regions and countries in the current Respiratory Care Devices market scenario?
- Which are the regions and countries where companies should have concentrated on opportunities for the Respiratory Care Devices Market Growth in the future?