Shaping the Future of Idiopathic Membranous Nephropathy Treatment: Breakthroughs in Targeted Therapies
Apr 17, 2026
Table of Contents
Summary
- The epidemiological landscape of IMN highlights a substantial disease burden across the seven major markets. According to DelveInsight’s analysis, IMN accounted for approximately 69,000 prevalent cases in 2025, within a broader pool of nearly 87,000 membranous nephropathy cases. This indicates that IMN constitutes a significant proportion of the overall disease population, reinforcing its importance as a key focus area for therapeutic and market development strategies.
- From a pathophysiological perspective, IMN is strongly defined by antigen-specific mechanisms, particularly involving subepithelial immune complex deposits. Among these, PLA2R and THSD7A remain the most clinically relevant biomarkers. In 2022, around 20,000 patients in the United States were associated with PLA2R, compared to 800 and 4,000 patients linked to THSD7A and other antigens, respectively. This distribution underscores the dominance of PLA2R-driven disease and suggests a clear opportunity for biomarker-targeted therapeutic approaches and precision medicine strategies.
- Despite the absence of formally approved therapies specifically indicated for IMN, the treatment landscape is far from static. Rituximab has emerged as the de facto first-line therapy in clinical practice, driven by strong physician confidence and widespread adoption. Its dominance in 2025 reflects not only its clinical efficacy but also the lack of approved alternatives, positioning it as the current cornerstone of disease management. However, this also highlights a critical gap in regulatory-approved options.
- The shift from broad immunosuppression to more targeted, precision-led approaches and what that implies for future treatment and market dynamics. Therapies such as Felzartamab and Obinutuzumab are anticipated to significantly influence the IMN market size across the 7MM. Their potential to introduce differentiated mechanisms of action and improved clinical outcomes positions them as key drivers of future market growth and competitive dynamics.
- Geographically, the United States continues to lead the IMN market, accounting for approximately 60% of the total share in 2025. This dominance reflects robust healthcare infrastructure, higher diagnosis rates, and early adoption of advanced therapies. In contrast, Japan represents the second-largest market with an 8.0% share, supported by steady improvements in disease awareness and treatment uptake. Epidemiologically, Japan reported around 15,000 IMN cases in 2022, with numbers expected to rise through the 2026–2036 forecast period, indicating a growing patient pool and future market opportunity.
- However, the IMN market continues to face several structural challenges that limit its full potential. Key unmet needs, including the absence of approved therapies, progression of patients to end-stage renal disease, and lack of consensus in treatment approaches, contribute to increased diagnostic, monitoring, and treatment costs. These barriers not only hinder optimal patient outcomes but also restrict market expansion. Addressing these gaps will be critical to improving disease management and unlocking future growth opportunities in the IMN landscape.
Idiopathic membranous nephropathy (IMN) is recognized as a distinct pattern of glomerular injury driven by an autoimmune mechanism. The disease is characterized by the deposition of immune complexes along the glomerular basement membrane, leading to its thickening and structural alterations in podocytes. These pathological changes are primarily mediated by autoantibodies that specifically target podocyte antigens, ultimately resulting in the development of proteinuria.
Downloads
Click Here To Get the Article in PDF
Recent Articles
Recent scientific advancements have led to the identification of several key autoantigens implicated in IMN, including the M-type phospholipase A2 receptor (PLA2R1), thrombospondin type-1 domain-containing 7A (THSD7A), and neural epidermal growth factor-like 1 protein (NELL-1). The detection and quantification of circulating autoantibodies against these antigens have emerged as important clinical tools, supporting both diagnosis and disease monitoring.
Despite these developments, several aspects of IMN pathogenesis remain inadequately understood. Key uncertainties persist regarding the initiation of autoimmunity, the predominance of IgG4 antibodies, the mechanisms underlying spontaneous remission, and the formation of characteristic glomerular lesions. Given the clinical significance of these gaps, a hypothetical model of disease pathogenesis has been proposed. This model suggests that environmental triggers or other external factors may lead to the exposure of antigens such as PLA2R1 and THSD7A in extrarenal tissues, particularly the lungs. This exposure may subsequently initiate an immune response, resulting in the production of circulating autoantibodies that target and damage podocytes within the kidney.
“The prevalence rates are not very well studied since the kidney biopsy performed for diagnosis occurs at various points along the spectrum of disease, and the duration of clinical disease and the number of relapses can impact the total number of people suffering from this disease at any point in time. We use the following statement “the annual incidence rates of MN are estimated at 10–12 per million in North America and 2–17 per million in Europe,” but there are no data beyond this that I am aware of.
– Boston University School of Medicine, Department of Medicine, Renal Section
Currently, there is no definitive or disease-specific therapy, and management primarily relies on supportive care, including diuretics and renin-angiotensin system blockers such as ACE inhibitors and angiotensin II receptor blockers, although their impact is modest. Immunosuppressive therapies, such as corticosteroids, alkylating agents, calcineurin inhibitors (Cyclosporin A and tacrolimus), and mycophenolate mofetil, are used in selected patients, but their efficacy remains variable and is often limited by safety concerns. Consequently, overall prognosis has improved only marginally over the past decades, with a substantial proportion of patients progressing to end-stage renal disease. Emerging evidence highlighting the central role of B cells in disease pathogenesis has shifted focus toward targeted therapies, particularly B-cell depletion strategies, which offer the potential for more effective and safer disease control by reducing pathogenic antibody production.
IMN Treatment Challenges
Many immunosuppressive therapies used in IMN, such as alkylating agents and calcineurin inhibitors, are associated with significant safety concerns, including increased risk of infections, organ toxicity, and long-term complications like malignancy with agents such as cyclophosphamide. These risks often necessitate cautious use, close monitoring, and, in some cases, treatment discontinuation, thereby complicating disease management and limiting the widespread adoption of current IMN therapeutic options.
Despite recent advancements, primary Membranous Nephropathy (pMN) is characterized by a heterogeneous pathophysiology involving multiple autoantibody targets beyond PLA2R, including THSD7A and NELL-1. This diversity adds to diagnostic complexity and highlights the need for more nuanced disease classification. As a result, patients may require differentiated therapeutic strategies based on their underlying immunologic profile. Therapies focused on a single pathway may not adequately address all subtypes of the disease, underscoring the importance of developing broader or more personalized treatment approaches to achieve optimal clinical outcomes.
Pipeline Advancements in IMN
Emerging therapies are reshaping the treatment landscape of IMN by transitioning from broad immunosuppression to targeted, mechanism-driven approaches. Agents such as obinutuzumab, which enhance B-cell depletion, exemplify this shift toward precision therapy. Compared to conventional treatments associated with toxicity and relapse risk, these novel therapies enable deeper and more sustained immunological remission while supporting a more personalized, biomarker-driven treatment strategy, ultimately improving safety and long-term renal outcomes.
In June 2025, Biogen Inc. announced the initiation of dosing in the global clinical study, PROMINENT. The Phase III study evaluated the efficacy and safety of the investigational drug felzartamab compared to tacrolimus in adults diagnosed with primary membranous nephropathy.
In June 2025, Ono Pharmaceutical Co., Ltd. and Vertex Pharmaceuticals Incorporated announced an exclusive collaboration and license agreement for the development and commercialization of Vertex’s povetacicept in Japan and South Korea. Under the terms of the agreement, Vertex will receive an upfront payment, as well as certain regulatory and commercial milestone payments and tiered royalties. Ono will utilize its extensive development expertise to help advance Vertex’s clinical trials for povetacicept and will be responsible for obtaining marketing authorizations in Japan and South Korea. Following approval, Ono will be solely responsible for commercializing povetacicept in these regions. The agreement includes povetacicept for both IgAN and pMN, with the potential to add other indications.
Obinutuzumab: Hoffmann-La Roche
Obinutuzumab, an anti-CD20 monoclonal antibody, is currently being evaluated in a Phase III trial for pMN. It has shown a favorable safety and efficacy profile in clinical studies and is already approved for conditions such as lymphocytic leukemia and follicular lymphoma. Real-world retrospective data indicate strong therapeutic potential in pMN, with high immunological (92%) and clinical remission rates (83.6%), alongside improvements in proteinuria, serum albumin, and renal function over a median follow-up of 13 months. Notably, its efficacy appears consistent across both first-line and alternative settings, including in patients refractory to prior therapies and those negative for anti-PLA2R antibodies, underscoring its promise as an effective treatment option.
Modifying the glycan tree structure at the Fc fragment of obinutuzumab increases affinity for FcgRIII, thereby potentiating antibody-dependent cellular cytotoxicity by natural killer cells and antibody-dependent cellular phagocytosis by macrophages. These B-cell depletion mechanisms contrast with the primarily complement-dependent cytotoxicity of rituximab. When combined with chemotherapy, obinutuzumab is more effective than rituximab in treating patients with certain B-cell malignancies.
Supported by positive clinical study results, it offers a promising safety profile and convenient dosing, positioning it as a differentiated, next-generation pharmacological approach in the evolving idiopathic membranous nephropathy landscape, with its launch anticipated in 2027.
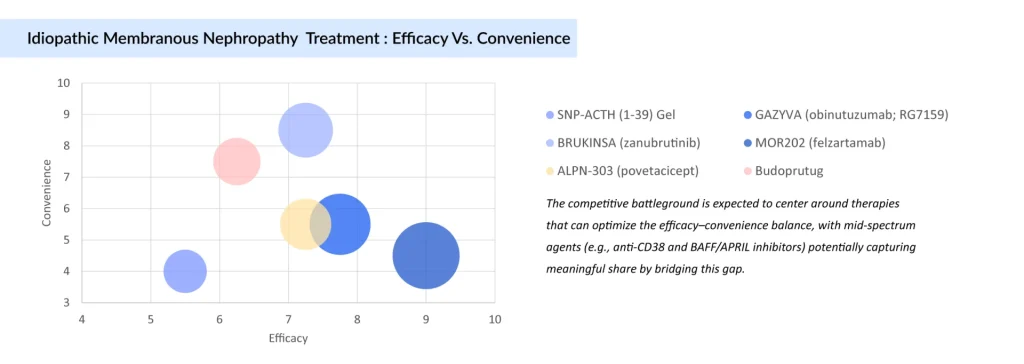
Pipeline differentiation is shifting toward convenience without compromising efficacy. The IMN treatment landscape is transitioning from high-efficacy, infusion-based immunosuppressive therapies toward more patient-friendly, targeted approaches, with convenience emerging as a key differentiator in future adoption.
While B-cell–depleting agents such as obinutuzumab continue to anchor the high-efficacy segment, their clinical use is constrained by treatment burden and monitoring requirements. In parallel, next-generation agents, particularly oral therapies like zanubrutinib, are well positioned to disrupt the market by offering significantly improved convenience with competitive efficacy, contingent on robust late-stage validation.
Unmet Needs in IMN Treatment
IMN continues to present significant unmet needs, primarily driven by the absence of a clearly approved, disease-specific therapy with a favorable risk–benefit profile. While current management focuses on reducing proteinuria and preserving renal function, the optimal timing for initiating immunosuppressive therapy, the most appropriate treatment choice, and the identification of patients who would derive the greatest benefit remain areas of ongoing uncertainty. Furthermore, the lack of a personalized treatment approach limits the ability to tailor therapies based on individual disease characteristics. Although advances such as the identification of PLA2R and other autoantigens have improved the understanding of disease pathogenesis, these insights have not yet been fully translated into targeted, highly effective therapies, leaving a substantial gap in clinical management.
In addition to therapeutic limitations, IMN imposes a considerable economic burden on both patients and healthcare systems. The absence of standardized diagnostic and monitoring pathways often leads to repeated investigations and increased healthcare utilization, negatively impacting patient quality of life and resource allocation. High costs associated with biomarker testing, such as anti-PLA2R assays, and long-duration therapies further exacerbate this burden, highlighting the need for more cost-effective and streamlined management strategies.
Current treatment options are also associated with notable shortcomings. Patients frequently experience significant disease-related complications, including nephrotic syndrome and its associated risks, while available immunosuppressive therapies carry the potential for serious adverse effects such as infections, malignancies, and long-term toxicity. Although therapies like rituximab have improved tolerability profiles, challenges such as relapse risk and treatment-related toxicity persist, and the role of combination regimens remains unclear.
Collectively, these gaps represent a strong opportunity for the IMN market. The clear need for safer, more effective, and targeted therapies creates a favorable environment for innovation, particularly in the development of precision medicines and B-cell-directed treatments. Additionally, there is significant scope for advancements in biomarker-driven diagnosis, patient stratification, and cost-efficient treatment approaches. Companies that can address these unmet needs by delivering therapies with improved efficacy, reduced adverse effects, and better long-term outcomes are well-positioned to capture substantial value in the evolving IMN therapeutic landscape.
IMN Market Growth Outlook
The IMN treatment market is expected to witness steady growth, driven by improved diagnostics, rising disease awareness, and a shift toward targeted therapies. While current management relies largely on off-label immunosuppressants, increasing adoption of B-cell-targeted treatments is reshaping the landscape. Advances in biomarker-based diagnosis and a growing pipeline of novel therapies are likely to enable more personalized and effective treatment approaches. Overall, the market holds strong future potential, with growth supported by innovation aimed at improving efficacy, reducing relapse, and minimizing safety concerns.
The treatment of IMN involves supportive care and immunosuppressive therapies based on disease severity. Initial management focuses on blood pressure control, renin–angiotensin system inhibitors, and measures to manage complications like edema and hyperlipidemia, aiming to reduce proteinuria and support remission. For moderate to high-risk patients, immunosuppressive therapy is initiated, with rituximab as the preferred first-line option due to its efficacy and safety profile. Alternatives include corticosteroids with alkylating agents and calcineurin inhibitors, though the latter are associated with higher relapse rates. Other treatments are less commonly used due to variable efficacy. Overall, while current therapies improve outcomes, their use is limited by safety concerns and relapse risk, highlighting the need for more targeted and durable options.
Practical Hurdles Shaping Current and Emerging Therapies
A major challenge in IMN is its complex and heterogeneous autoimmune biology, which continues to limit the success of emerging targeted therapies. Although antigens such as PLA2R have transformed disease understanding, variability in immune pathways leads to inconsistent treatment responses. For instance, while B-cell–depleting therapy such as rituximab has shown meaningful remission rates, outcomes still vary widely across patients, reflecting underlying biological diversity and the need for more personalized therapeutic approaches.
Closely linked to this is the limited predictive power of current biomarkers, which restricts optimal patient stratification. Anti-PLA2R antibodies are widely used for diagnosis and monitoring, yet their ability to predict long-term response or relapse remains imperfect. This limitation is evident in clinical practice, where therapies such as rituximab and newer agents like obinutuzumab are often administered without clear evidence of response, leading to trial-and-error treatment strategies.
Another significant hurdle is the lack of a universally accepted treatment paradigm, particularly with the influx of emerging biologics. While rituximab has increasingly become a preferred first-line option due to a better safety profile compared to cyclophosphamide, its remission rates are still suboptimal, prompting investigation into combination strategies.
Safety and tolerability concerns remain a persistent barrier, especially given the reliance on immunosuppressive mechanisms. Conventional agents are associated with infections, malignancy risk, and nephrotoxicity, while even newer biologics are not devoid of adverse effects. This creates a delicate balance between efficacy and long-term safety, particularly in a disease where spontaneous remission can occur in a subset of patients.
Finally, market-level barriers such as limited commercial incentives and high therapy costs further restrict the adoption of emerging therapies. The relatively low prevalence of IMN reduces large-scale investment, while expensive biologics and combination regimens may face reimbursement challenges. This is particularly relevant for newer targeted therapies, which, despite promising efficacy, may not be readily accessible across all healthcare settings.

Frequently Asked Questions
GAZYVA and SNP-ACTH (1–39) Gel are expected to emerge as key future competitors in the idiopathic membranous nephropathy market following their anticipated launch around 2027. Their entry is likely to intensify competition by offering alternative treatment options with potentially improved efficacy and safety profiles. As a result, these therapies may capture a meaningful share of the market, particularly among patients who are refractory to or intolerant of existing treatments. This increased competition is expected to gradually shift market dynamics, leading to a redistribution of market share from currently established therapies, while also expanding the overall market through improved treatment adoption and patient outcomes.
The emerging pipeline for IMN includes a diverse set of therapies targeting different immune pathways, reflecting a shift toward more targeted and mechanism-driven treatment approaches. Key late-stage candidates include Obinutuzumab (GAZYVA), an anti-CD20 monoclonal antibody in Phase III, and Felzartamab (MOR202), an anti-CD38 monoclonal antibody also in Phase III, both focusing on B-cell and plasma cell depletion. Additionally, Zanubrutinib (BRUKINSA), a BTK inhibitor in Phase II/III, represents a small-molecule approach targeting B-cell signaling pathways. Earlier-stage therapies include Budoprutug, a CD19-targeting monoclonal antibody in Phase II, and NKX019, a CAR-NK cell therapy targeting CD19 in Phase I/II, highlighting the growing interest in cell-based immunotherapies. Overall, the pipeline demonstrates a strong emphasis on targeting B cells and related immune mechanisms, with the potential to offer more effective and durable treatment options in the future.
The key unmet needs in IMN include the absence of a disease-specific approved therapy, reliance on off-label treatments with variable efficacy, and significant safety concerns associated with immunosuppressive agents. There is also a lack of clear guidance on treatment initiation and patient selection, along with high relapse rates that limit long-term disease control. Additionally, gaps in biomarker-driven diagnosis and patient stratification, coupled with the overall economic burden of management, highlight the need for more targeted, safer, and durable therapeutic options.
According to DelveInsight, in the 7MM, the IMN market was approximately USD 130 million in 2022. The market is projected to grow at a CAGR of about 24% from 2026 through 2036. The growth of the IMN market is driven by emerging therapies, increasing awareness, and earlier diagnosis, alongside continuous advancements in treatment options. These factors are expanding access to care and supporting the overall rise in market size.
Future transformation in IMN therapy and patient care will be driven by the shift toward targeted and precision-based treatments, particularly B-cell and plasma cell-directed therapies. Advancements in biomarker-based diagnostics, such as autoantibody monitoring, will enable earlier diagnosis, better patient stratification, and more personalized treatment decisions. In addition, a robust pipeline of novel agents with improved efficacy and safety profiles is expected to reduce relapse rates and treatment-related toxicity. The integration of these innovations with evolving clinical guidelines and increased focus on individualized care will significantly enhance disease management and long-term patient outcomes.
Downloads
Article in PDF




