Travere Turns Setback into Success with FILSPARI Breakthrough in FSGS
Apr 20, 2026
For decades, patients with focal segmental glomerulosclerosis had no approved treatment. Then Travere Therapeutics did the unthinkable: it turned a Phase 3 setback into a historic approval.
Imagine being diagnosed with a progressive kidney disease, one that scars your kidneys from the inside, strips proteins from your blood, and steadily marches you toward organ failure, only to be told there is no approved treatment for it. That has been the reality for patients with focal segmental glomerulosclerosis (FSGS) for generations. Roughly seven in every one million people are diagnosed with FSGS. In 2024, the diagnosed prevalent cases of FSGS were highest in the US compared to the other 7MM.
DelveInsight estimates that the country alone accounted for 36% of the diagnosed prevalent cases among the 7MM. The disease damages the small filtering units of the kidney, causing protein to spill into urine, triggering hypertension, severe swelling, and ultimately kidney failure. Until now, nephrologists could only manage symptoms, never target the disease directly with a regulatory-backed therapy. That changed on April 13, 2026. On that day, the FDA granted full approval to FILSPARI (sparsentan), developed by Travere Therapeutics, making it the first and only FDA-approved medicine for FSGS in history. The announcement sent Travere’s stock surging nearly 40% in pre-market trading. But the real story isn’t about share prices. It’s about a drug that refused to quit, and the patients who waited far too long for it.
Downloads
Click Here To Get the Article in PDF
Recent Articles
- Advancements in IgA Nephropathy (IgAN) Treatment: From Standard-of-care (SoC) to Emerging Therapies
- Incyte’s ZYNYZ Approved as First-Line Treatment for Advanced Anal Cancer; Amneal Gets FDA Nod for...
- Focal Segmental Glomerulosclerosis Market: Unfolding Insights into the Future
- Travere’s FILSPARI Approval Sparks Rivalry in the IgA Nephropathy Space
FILSPARI is not your typical kidney drug. Rather than suppressing the immune system, it works through a dual-receptor mechanism, simultaneously blocking the endothelin A receptor (ETAR) and the angiotensin II subtype 1 receptor (AT1R). These two pathways are believed to be key drivers of the inflammation, scarring, and glomerular damage that characterize FSGS.
By targeting both simultaneously, FILSPARI attacks the disease from two angles at once, reducing proteinuria, protecting the kidney’s delicate filtering cells, and guarding against the fibrosis that would otherwise quietly destroy kidney architecture over time.
This first-in-class mechanism isn’t new to Travere’s portfolio. FILSPARI had already earned its regulatory stripes, first receiving accelerated FDA approval in February 2023 for IgA nephropathy treatment, another rare kidney disease, and then converting that to a full traditional approval in September 2024. That track record paved the way for the FSGS push.
The FDA’s full approval of Travere Therapeutics’ FILSPARI for FSGS is not just a regulatory milestone; it’s a story of scientific perseverance in the face of clinical adversity. Here’s the twist: FILSPARI failed its primary endpoint in the pivotal Phase 3 DUPLEX trial. Data from the 108-week study showed that the drug did not significantly outperform Sanofi’s AVAPRO (irbesartan) on kidney function as measured by eGFR slope. In most drug development stories, that’s where the chapter ends. But Travere refused to close the book.
While FILSPARI stumbled on one metric, it excelled on another that arguably matters more to patients, proteinuria reduction, a key marker of kidney disease progression and a recognized surrogate endpoint aligned with KDIGO (Kidney Disease: Improving Global Outcomes) guidelines.
The DUPLEX trial, the largest head-to-head interventional study in FSGS to date, showed that patients on FILSPARI achieved a statistically significant 46% reduction in proteinuria from baseline at Week 108, compared to just 30% for those on the maximum labeled dose of irbesartan. Published in the New England Journal of Medicine, the data also revealed higher rates of partial and complete remission and a lower incidence of end-stage kidney disease in FILSPARI-treated patients.
Travere further bolstered its case with findings from the earlier Phase 2 DUET study, which demonstrated a more than twofold decrease in proteinuria compared to irbesartan. Together, the combined package was compelling enough for the FDA to grant full approval, making FILSPARI the first and only approved medicine for FSGS in the United States.
CEO Eric Dube called the approval “a historic milestone for people living with FSGS, who for the first time have an FDA-approved medicine for this rare and devastating condition.” He added that FILSPARI would be available immediately for nephrologists to prescribe.
Kirk Campbell, MD, president of the National Kidney Foundation, echoed that sentiment, noting that the approval gives nephrologists a validated, guideline-consistent option in a space that had been operating on hope and off-label workarounds for far too long.
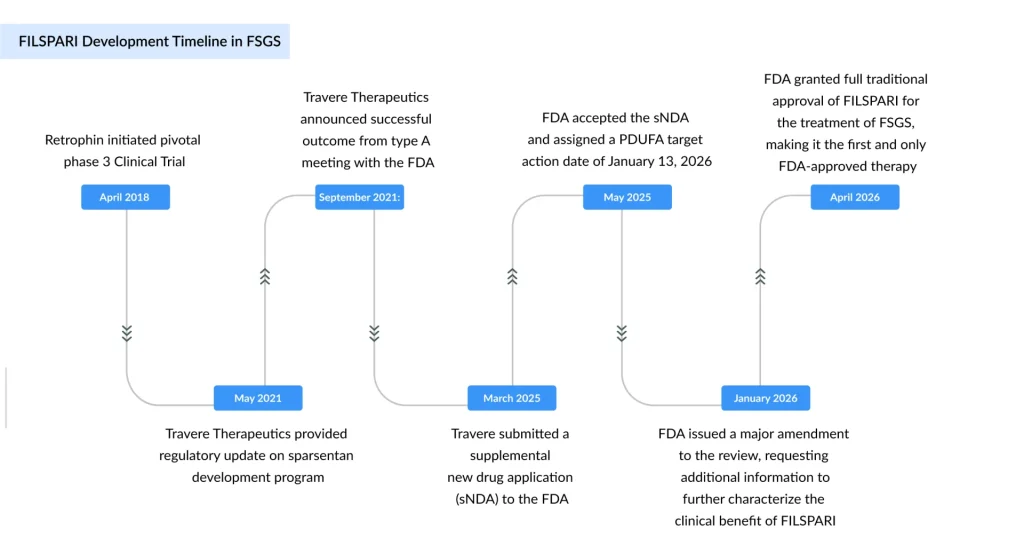
The path wasn’t just scientifically uncertain; it was administratively bumpy too. Travere filed its supplemental New Drug Application for FSGS in early 2025. The FDA accepted it in May 2025 and set an initial target action date of January 13, 2026. But in January, the agency announced it needed an additional three months to review additional submissions, pushing the deadline to April 13, 2026. For a company betting heavily on this approval, every month of uncertainty carries real weight.
When the approval finally landed, right on the extended deadline, it validated both the science and Travere’s willingness to stay in the ring. The commercial stakes are significant. Travere estimates the total addressable FSGS population in the U.S. at more than 30,000 patients, with the drug becoming immediately available for nephrologists to prescribe.
Sadaf Javed, manager of forecasting at DelveInsight, projected that FILSPARI could earn $70 million in the FSGS indication alone in 2027, with peak FSGS sales potentially exceeding $2 billion before patent expiration in 2033.
Travere already has an established commercial footprint from FILSPARI’s IgAN business, which generated $322 million in revenue in 2025, a 144% year-on-year jump following the drug’s full approval for that indication. The FSGS expansion doesn’t require building commercial infrastructure from scratch; it layers onto an existing platform.
Perhaps the most compelling takeaway from FILSPARI’s FSGS approval is what it says about drug development in the rare disease space. Clinical trials don’t always yield neat, decisive victories. Primary endpoints get missed. Timelines get extended. The data gets complicated.
The question is whether the totality of evidence, the mechanism, the proteinuria data, the remission rates, and the real-world disease trajectory for untreated patients makes a compelling enough case for regulators to act.
In this case, it did. And for the tens of thousands of patients with FSGS who have spent years navigating a condition with no dedicated treatment, that decision carries a weight no stock chart can fully capture. FILSPARI’s approval is a reminder that in medicine, “failed trial” doesn’t always mean “failed drug.” Sometimes, it just means the story is more complicated than a single endpoint, and that staying the course matters.

Downloads
Article in PDF
Recent Articles
- Focal Segmental Glomerulosclerosis Market: Unfolding Insights into the Future
- Travere’s FILSPARI Approval Sparks Rivalry in the IgA Nephropathy Space
- Incyte’s ZYNYZ Approved as First-Line Treatment for Advanced Anal Cancer; Amneal Gets FDA Nod for...
- Advancements in IgA Nephropathy (IgAN) Treatment: From Standard-of-care (SoC) to Emerging Therapies