Postoperative Ileus Treatment: Innovation Lag in a High-Burden Market
Feb 27, 2026
Table of Contents
Summary
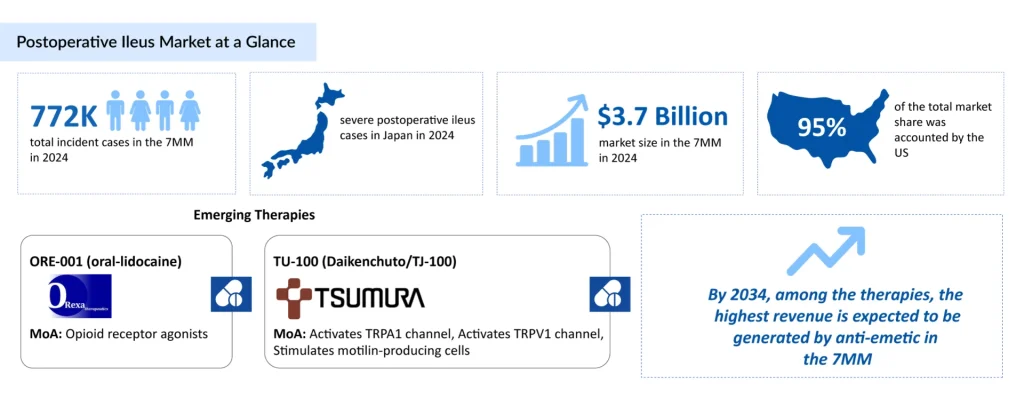
- In 2024, the 7MM reported an estimated 772K new cases of postoperative ileus.
- FDA-approved treatment options include metoclopramide (REGLAN), neostigmine (PROSTIGMIN), and alvimopan (ENTEREG), all of which face generic competition, with alvimopan losing market exclusivity in 2024.
- The development pipeline for postoperative ileus remains sparse, with only a handful of mid-stage candidates, including ORE-001 (lidocaine) and TU-100 (Daikenchuto/TJ-100) in Phase II trials, highlighting slow therapeutic advancement and limited innovation.
- The 7MM market for postoperative ileus was valued at approximately USD 3.7 billion in 2024 and is expected to grow steadily throughout the 2025–2034 forecast period.
Postoperative ileus is a common, temporary impairment of bowel motility that occurs after major surgery, particularly abdominal procedures. It affects up to 25–30% of patients undergoing colorectal surgery and is responsible for nearly 15–20% of all postoperative complications. Abdominal surgery is said to be the most common cause of postoperative ileus. In the United States, in all inpatient gastrointestinal (GI) surgeries, about 6 out of 100 (5.8%-6.3%) developed diagnosed postoperative ileus. Despite the integration of enhanced recovery protocols, the prevalence of postoperative ileus remains variable based on the specific surgical procedure, ranging from 7% to 27%, after colorectal surgery, demonstrating peak occurrence.
Downloads
Click Here To Get the Article in PDF
Recent Articles
The total number of incident cases of postoperative ileus in the 7MM was nearly 771K in 2024. In the US, gastrointestinal/abdominal procedures accounted for approximately 5.6 million cases in 2024, representing a higher number than non-abdominal procedures in the at-risk population for postoperative ileus. Older patients and males are disproportionately affected, with epidemiological analyses consistently identifying them as higher-risk groups for developing clinically significant postoperative ileus.
Ramandeep Singh, Senior Consultant for Forecasting and Analytics at DelveInsight, highlighted that postoperative ileus continues to be a frequent complication after abdominal tumor surgery, with the potential to advance into prolonged postoperative ileus. He emphasized that minimally invasive surgery, early enteral nutrition, and multimodal analgesia are strongly supported by clinical evidence and form key components of ERAS guidelines. Additional interventions, including chewing gum, laxatives, alvimopan, and acupuncture, may also help lower the incidence of postoperative ileus and promote faster recovery.
Characterized by delayed passage of flatus or stool, abdominal distension, nausea, and intolerance to oral intake, postoperative ileus can extend hospital stays by 2–5 days on average and significantly increase healthcare costs. Although traditionally viewed as an unavoidable consequence of surgical stress, growing evidence shows that factors such as opioid use, extensive bowel handling, fluid overload, and underlying comorbidities play major roles, making prevention and early management essential components of modern perioperative care.
Postoperative Ileus Treatment Landscape: An Area in Need of Innovation
Current management of postoperative ileus remains largely supportive, focusing on hydration, electrolyte correction, early ambulation, and minimizing opioid use. While these measures help maintain patient stability, they do not directly restore gastrointestinal motility or significantly shorten recovery time. Despite the clinical impact of postoperative ileus on outcomes and hospital stay, truly effective pharmacologic interventions remain limited.
Historically, alvimopan (ENTEREG), a peripherally acting µ-opioid receptor antagonist (PAMORA), was the only FDA-approved postoperative ileus therapy proven to accelerate gastrointestinal recovery after bowel resection. However, following the discontinuation of the branded product, clinicians now rely on a mix of generic agents and standard supportive care.
Current therapeutic strategies include prokinetic agents such as metoclopramide, which offer modest benefits but are constrained by central nervous system side effects; NSAIDs and opioid-sparing regimens to reduce gut motility suppression; and PAMORAs like alvimopan and methylnaltrexone, which target peripheral opioid receptors in the gut. While alvimopan has demonstrated efficacy, its cost, safety profile, and limited availability restrict widespread use. Other adjunctive options, such as centrally acting opioid antagonists (e.g., naloxone), antiemetics, laxatives, and intravenous lidocaine, primarily address symptoms or contribute indirectly to bowel recovery.
According to Ramandeep, despite nearly 20 years of limited therapeutic innovation, the management of postoperative ileus still relies largely on supportive care rather than targeted therapies. He emphasized the clear unmet need for safe and effective treatments that can enhance gastrointestinal recovery and improve postoperative outcomes, especially in colorectal surgery, where postoperative ileus continues to impact patient well-being and hospital resource use significantly.
Phase II Therapeutic Candidates Driving Pipeline Progress
Despite the absence of any approved branded therapies for postoperative ileus, the treatment pipeline, though limited, shows strategic progress focused on the disorder’s fundamental mechanisms, including impaired gastrointestinal motility and opioid-induced dysfunction. Two promising Phase II candidates, ORE-001 (Orexa BV) and TU-100 [(Tsumura (TYO: 4540)], are currently advancing in development, intending to improve symptom relief and management of POI across the seven major markets (7MM).
ORE-001 is an oral lidocaine formulation developed by Orexa BV, designed to stimulate food intake and aid recovery in conditions such as postoperative ileus, anorexia, and age-related malnutrition. The company is currently conducting a Phase II clinical trial (NCT05923086) evaluating ORE-001 for the prevention of postoperative ileus. In January 2024, Orexa BV announced the dosing of the first patient in this study, following a June 2023 collaboration with the Clinical Research Organization QPS to conduct its Phase IIa prevention-focused trial.
Meanwhile, TU-100, a traditional Japanese herbal medicine, is being actively explored by Tsumura for its potential to accelerate recovery from postoperative ileus following abdominal surgery. TU-100 enhances gastrointestinal motility by stimulating neural pathways, particularly through increased acetylcholine release via serotonergic receptor activation, and exerts anti-inflammatory effects by reducing neutrophil and macrophage infiltration while improving intestinal blood flow. In March 2024, TU-100 completed a Phase II clinical trial (NCT04742907) for the treatment of postoperative ileus, and by July 2024, Tsumura announced the completion of patient enrollment for its late Phase II trial involving 402 participants.
Together, these investigational therapies underscore a growing clinical interest in targeted strategies aimed at addressing the underlying physiological dysfunctions that contribute to postoperative ileus.
Roadblock in the Postoperative Ileus Market
The postoperative ileus market is projected to experience sluggish growth in the coming years, primarily due to limited pipeline activity and ongoing challenges in achieving consensus on its definition. Currently, there is no universally accepted definition of postoperative ileus; clinical studies, medical guidelines, and hospital protocols often vary in their criteria, basing diagnosis on factors such as bowel inactivity duration, the need for nasogastric decompression, delayed passage of stool or flatus, or extended hospital stays.
Additionally, the adoption of non-pharmacological approaches—such as early patient mobilization and nutritional optimization, may further reduce the dependence on pharmacological interventions. However, prolonged cases of ileus still necessitate close clinical monitoring, as the condition can mimic or develop into severe complications like mechanical obstruction, infection, or anastomotic leaks. These complexities not only elevate clinical risks but also contribute to increased healthcare costs and resource burdens.
What Lies Ahead in the Postoperative Ileus Treatment?
Postoperative ileus remains one of the most common complications following abdominal surgery, sustaining a steady demand for effective therapeutic options. The growing emphasis on Enhanced Recovery After Surgery (ERAS) protocols further supports the adoption of strategies aimed at reducing its incidence and improving patient outcomes.
In 2024, the total market for postoperative ileus across the 7MM was valued at approximately USD 3.7 billion, with steady growth anticipated throughout 2025–2034. According to DelveInsight’s projections, anti-emetic therapies are expected to generate the highest revenue by 2034 within this market.
Currently established management practices, such as bowel rest, IV fluid support, and nasogastric decompression, provide reliable clinical benefits, setting clear benchmarks for evaluating the value of emerging treatment options.
Ongoing scientific progress is also reshaping the therapeutic landscape. Deeper insights into the molecular mechanisms of postoperative ileus, including inflammatory pathways and autonomic nervous system disruption, are paving the way for targeted therapies that may significantly enhance recovery and address long-standing unmet needs.
Promising agents such as TU-100 (Daikenchuto), known for facilitating gastrointestinal recovery and shortening hospital stays with minimal safety concerns, highlight the potential for innovative or adjunctive treatment approaches in the years ahead.

Downloads
Article in PDF