Acute Ischemic Stroke Treatment and Diagnosis Devices: Technologies Transforming Stroke Care
Apr 08, 2026
Table of Contents
Stroke remains one of the most critical challenges in modern medicine, representing a leading cause of long-term disability and mortality worldwide. According to the World Health Organization (WHO), stroke is responsible for approximately 11% of total deaths globally. Within this landscape, Acute Ischemic Stroke (AIS), caused by a sudden blockage in a cerebral artery, accounts for the vast majority of cases. The clinical imperative in AIS is summarized by the mantra “Time is Brain,” as millions of neurons perish every minute that blood flow is obstructed.
In recent years, a paradigm shift has occurred in how we identify and treat these events. The convergence of high-resolution imaging, precision catheter-based interventions, and artificial intelligence is extending the window of opportunity for recovery.
Downloads
Click Here To Get the Article in PDF
Recent Articles
- Boston Scientific Corporation Agreement to Acquire Silk Road Medical; Philips Introduced Duo Veno...
- Prolong’s PP-007 Fast-Tracked for Stroke; FDA Expands JYLAMVO Pediatric Approval; Corcept’s Cushi...
- Roche’s TNKase — First New Acute Ischemic Stroke Drug in Nearly 30 Years
- Wider Administration Window – Need of the Moment for Acute Ischemic Stroke Patients
- Abbott’s New CardioMEMS™ Remote Monitoring Reader Gains FDA Approval; Mobia Medical and Sanford H...
Diagnosis Devices for Acute Ischemic Stroke
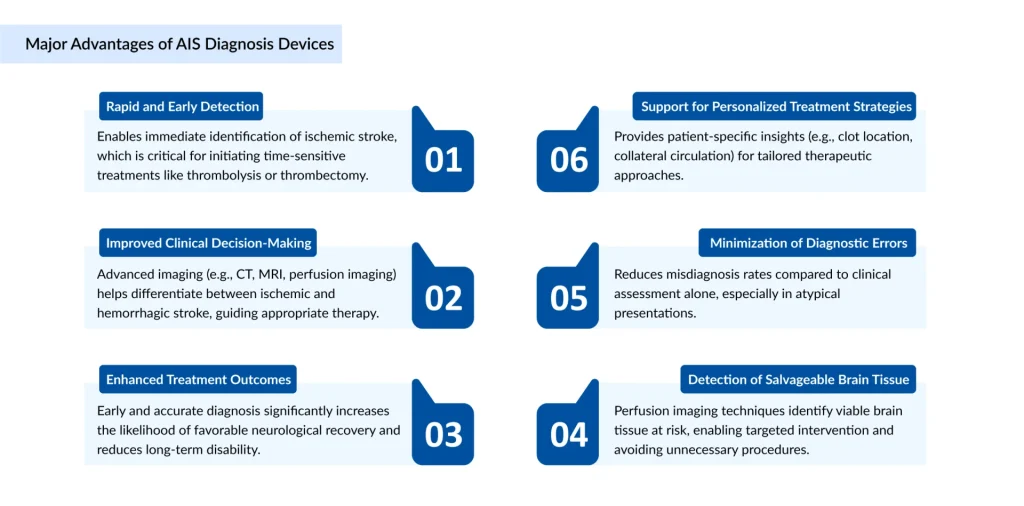
The efficacy of stroke treatment is entirely dependent on the speed and accuracy of the initial diagnosis. Clinical evaluation alone cannot distinguish between an ischemic stroke and a hemorrhagic stroke, making advanced diagnostic imaging the gatekeeper of the treatment pathway.
Computed Tomography (CT) Scanners
CT remains the frontline diagnostic tool for AIS. Its primary role, as guided by FDA and CDC protocols, is to rapidly rule out intracranial hemorrhage, which would contraindicate the use of clot-busting medications. Modern CT technology, including Multi-Detector CT (MDCT) and CT Angiography (CTA), allows clinicians to visualize not only the structure of the brain but also the vascular architecture and blood flow (CT Perfusion). These advancements help identify the “penumbra”, the salvageable brain tissue surrounding the core of the stroke.
Magnetic Resonance Imaging (MRI)
While CT is the standard for speed, MRI is the standard for sensitivity. MRI, particularly Diffusion-Weighted Imaging (DWI), can detect ischemic changes within minutes of onset, far earlier than conventional CT. By utilizing “mismatch” protocols, comparing DWI with Fluid-Attenuated Inversion Recovery (FLAIR) sequences, physicians can estimate the age of a stroke, a critical factor when the patient’s “last known well” time is uncertain.
The magnetic resonance imaging devices market was valued at USD 8.2 billion in 2025, growing at a CAGR of 6.54% during the forecast period from 2026 to 2034 to reach USD 13.5 billion by 2034. The rising prevalence of various chronic disorders, such as cardiovascular diseases, cancer, and neurological conditions, is fueling the demand for precise diagnostic tools like MRI, which offers superior imaging capabilities for soft tissues and organs.
Transcranial Doppler Ultrasound (TCD)
TCD offers a non-invasive, portable method to monitor cerebral blood flow. It is particularly valuable for bedside assessments and real-time monitoring of clot dissolution (recanalization). Because it does not use ionizing radiation or contrast agents, it is an essential tool for continuous monitoring in intensive care settings to detect potential re-occlusion or vasospasm.
Portable and Prehospital Stroke Detection Devices
The frontier of stroke diagnosis has moved from the hospital to the ambulance. Emerging prehospital devices, such as microwave tomography headsets and portable “Mobile Stroke Units” equipped with small-scale CT scanners, are designed to begin the diagnostic process during transit. These technologies aim to shorten the “door-to-needle” and “door-to-puncture” times, ensuring that treatment begins the moment a patient reaches the hospital, or even before.
Treatment Devices for Acute Ischemic Stroke
Once a diagnosis is confirmed, the goal is rapid revascularization. While pharmacological thrombolysis (IV-tPA) remains a staple, the advent of endovascular therapy has revolutionized the treatment of Large Vessel Occlusions (LVOs).
Mechanical Thrombectomy Devices
Mechanical thrombectomy has become the gold standard for treating LVOs. These devices are physically navigated into the brain to remove the obstructing clot.
- Stent Retrievers: These are self-expanding mesh devices that “trap” the clot within their struts, allowing the surgeon to pull the obstruction out of the body.
- Aspiration Catheters: Utilizing high-power suction, these catheters “vacuum” the clot directly from the artery.
The mechanical thrombectomy devices market was valued at USD 849.70 million in 2025, growing at a CAGR of 6.88% during the forecast period from 2026 to 2034 to reach USD 1.4 billion by 2034. The demand for mechanical thrombectomy devices is being propelled by several key factors that collectively underscore the market’s strong growth. Foremost among these is the rising global prevalence of neurological and vascular disorders, including acute ischemic stroke and deep vein thrombosis, which continues to drive the need for effective and timely interventions.
Balloon Guide Catheters (BGCs)
A BGC is positioned in the carotid artery and inflated to temporarily arrest or reverse blood flow during the thrombectomy procedure. This prevents “clot fragmentation,” where small pieces of the clot break off and travel deeper into the brain, causing new strokes in different territories.
The balloon catheters market was valued at USD 4.9 billion in 2025, growing at a CAGR of 7.63% during the forecast period from 2026 to 2034, to reach USD 8.9 billion by 2034. The rising prevalence of cardiovascular diseases, brain aneurysms, and pelvic region cancers is significantly boosting the market for balloon catheters. As cardiovascular conditions become more common, the demand for balloon catheters increases.
Microcatheters and Guidewires
These are the “navigational” tools of the neurointerventionalist. Extremely thin and flexible, microcatheters are tracked over guidewires to reach the delicate, winding vessels of the brain. Modern iterations feature hydrophilic coatings and varying stiffness levels to navigate complex anatomy without damaging the vessel walls.
Neurovascular Access Systems
The success of any procedure depends on stable access. Neurovascular access systems provide a “highway” from the entry point (usually the groin or wrist) to the brain. These systems have evolved to be increasingly kink-resistant and “trackable,” allowing for the rapid delivery of therapeutic devices even in patients with tortuous vascular paths.
Emerging Technologies in Acute Ischemic Stroke Devices
Emerging technologies in acute ischemic stroke devices are rapidly transforming the landscape of stroke intervention, particularly in the management of large vessel occlusion (LVO). Mechanical thrombectomy has become the cornerstone of treatment, and recent innovations are focused on improving first-pass recanalization, procedural efficiency, and safety. Next-generation devices include advanced stent retrievers, large-bore aspiration catheters, and combination systems that integrate both approaches. These devices are designed with improved flexibility, trackability, and clot engagement capabilities, enabling better navigation through complex cerebrovascular anatomy and higher rates of successful reperfusion.
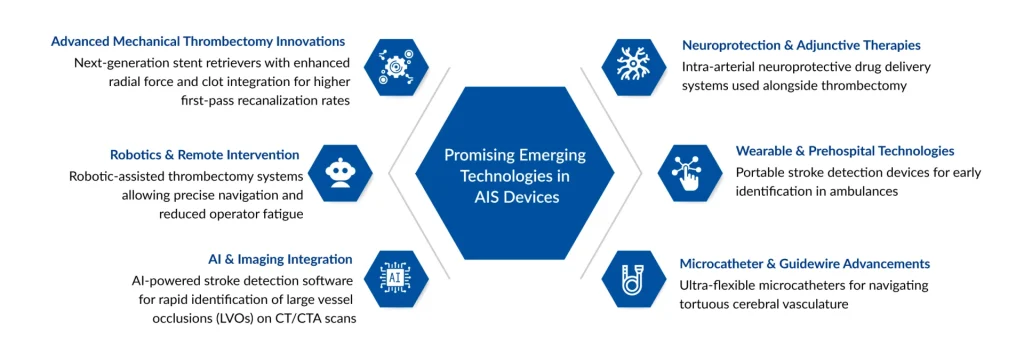
A major frontier in AIS device innovation is the integration of artificial intelligence (AI) and automation. AI-powered imaging platforms are increasingly used to rapidly detect occlusions, assess perfusion deficits, and guide treatment decisions in real time. Machine learning algorithms can analyze CT and MRI data to identify stroke regions and predict outcomes with accuracy comparable to expert clinicians, thereby reducing time-to-treatment, a critical determinant of patient outcomes. Additionally, experimental advancements in autonomous or robot-assisted thrombectomy systems are being explored, where AI can assist in catheter navigation and procedural planning, potentially expanding access to stroke care in resource-limited settings.
Another key area of innovation lies in advanced neuroimaging and smart diagnostic tools that complement interventional devices. Technologies such as dual-energy CT, spectral CT, and perfusion imaging software enable more precise characterization of ischemic tissue and viable penumbra. These imaging advancements improve patient selection for thrombectomy and extend treatment windows beyond traditional limits. Furthermore, “smart” catheters and sensor-enabled devices are being investigated to provide real-time feedback on clot composition, blood flow, and vessel wall interaction, allowing for more personalized and adaptive interventions.
Finally, emerging adjunctive and hybrid technologies are aiming to enhance the efficacy of existing stroke devices. These include novel thrombolytic delivery systems, nanotechnology-based clot targeting, and regenerative approaches that support neuroprotection and recovery. There is also growing interest in expanding device indications to medium and distal vessel occlusions, supported by improved miniaturized tools and refined endovascular techniques. Together, these innovations signal a shift toward more precise, data-driven, and minimally invasive stroke care, with the ultimate goal of improving functional outcomes and reducing the global burden of stroke.
Future Outlook of Acute Ischemic Stroke Devices
The economic and clinical landscape of the AIS market is characterized by robust growth and intense innovation. The global market for acute ischemic stroke treatment and diagnosis was valued at approximately USD 4.6 billion in 2025. This market is projected to expand at a CAGR of 4.85%, reaching an estimated USD 6.8 billion by 2034. This growth is fueled by a rising geriatric population, an increase in the prevalence of risk factors like hypertension and obesity, and the expansion of the “treatment window” from 6 hours to up to 24 hours in eligible patients.
The competitive landscape is dominated by several key medical technology giants and innovative startups. Companies such as Medtronic PLC, Abbott, Stryker, Johnson & Johnson Services, Inc., Terumo Corporation, Toro Neurovascular, Ceretrieve, Perfuze Ltd., CERENOVUS Inc., Rapid Medical, Boston Scientific Corporation, MicroVention, Inc., F. Hoffmann-La Roche Ltd., Boehringer Ingelheim International GmbH, Sanofi, Pfizer, Inc., AstraZeneca, Penumbra, Inc., Inari Medical, Inc., InspireMD, Inc., and others are at the forefront of device development. Recent milestones, such as the FDA clearance of the ROADSAVER Carotid Stent System and the launch of Penumbra’s Lightning Flash 2.0 for rapid clot removal, underscore the industry’s commitment to procedural efficiency. Moving forward, the industry is expected to focus on “smarter” devices, integrating real-time sensors and AI-assisted navigation, to ensure that the life-saving potential of stroke technology is accessible to more patients, faster than ever before.

Downloads
Article in PDF
Recent Articles
- Abbott’s New CardioMEMS™ Remote Monitoring Reader Gains FDA Approval; Mobia Medical and Sanford H...
- Roche’s TNKase — First New Acute Ischemic Stroke Drug in Nearly 30 Years
- Prolong’s PP-007 Fast-Tracked for Stroke; FDA Expands JYLAMVO Pediatric Approval; Corcept’s Cushi...
- Wider Administration Window – Need of the Moment for Acute Ischemic Stroke Patients
- Boston Scientific Corporation Agreement to Acquire Silk Road Medical; Philips Introduced Duo Veno...




