medical device
Feb 15, 2023
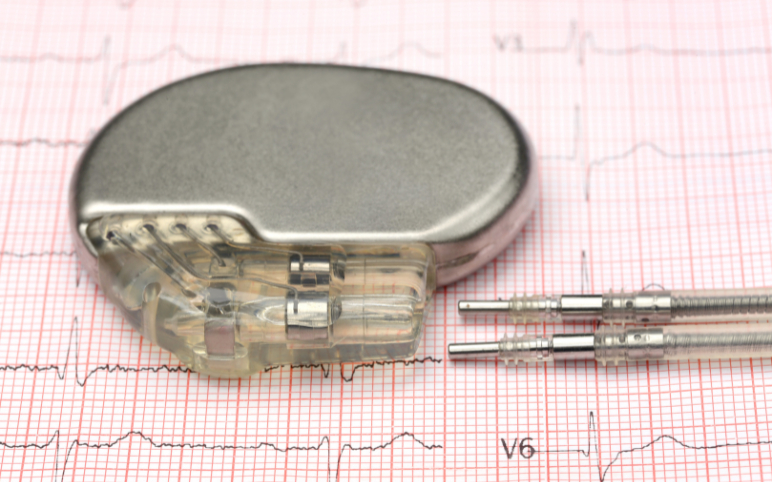
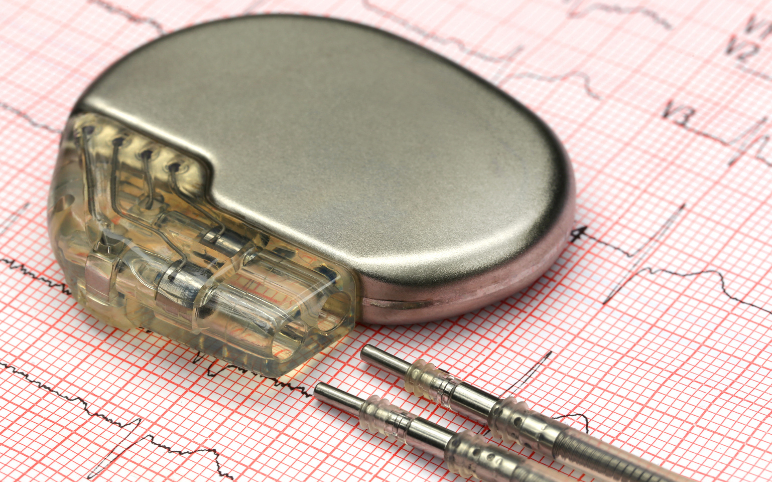
Pacemakers: Analyzing the Market Dynamics and Major Developments
Over the past few decades, the prevalence of cardiovascular diseases has grown significantly, leading to very high mortality and morbidity worldwide. CVDs are a group of disorders of the heart and blood vessels and include coronary heart disease, cerebrovascular disease, rheumatic heart disease, and other condition...
Read More...
Feb 09, 2023
LivaNova Launched SenTiva DUO; Candela Launched Matrix System; 3M Launched Medical Adhesive; MONARCH Platform Performed First Kidney Stones Removal Procedure; Biosense Webster Presented Data of HELIOSTAR Balloon Ablation Catheter; FDA 510(k) Clearance to Aspivix’s Cervical Stabilizer Carevix
LivaNova Launched SenTiva DUO, an Implantable Pulse Generator On February 3, 2023, LivaNova PLC, a market-leading medical technology and innovation company launched SenTiva DUO™, an implantable pulse generator (IPG) with a dual-pin header that delivers VNS Therapy™ for the treatment of drug-resista...
Read More...
Feb 08, 2023
Biochips: An Evolving Technology Driving the Future of Healthcare Market
Technological advancements over the past few decades have significantly transformed our lives and are anticipated to exhibit the trends in the upcoming years. Our living today is simplified to a much larger extent, which seemed unrealistic to imagine over the past few years. With each passing day, technological inn...
Read More...
Feb 01, 2023
Analyzing How Medical Coding is Playing a Conducive Role in Healthcare Service Delivery
Over the past few decades, technological innovations have led to immense growth and development in the healthcare industry and has simplified the patient treatment journey to a significant extent. The growth in high-tech medical devices, equipment, innovative targeted therapies, and ingenious procedures and techniq...
Read More...
Jan 19, 2023
First Patient Enrolled in Kaizen Clinical Study in Japan; Koneksa’s At-Home Mobile Spirometry Clinical Trial; Medcura’s LifeGel™ Absorbable Surgical Hemostat; FDA Clearance to Abbott’s NAVITOR; Enovis’s DynaClip Delta™ and DynaClip Quattro™ bone staples; Bausch + Lomb Acquired AcuFocus
First Patient Enrolled in Kaizen Clinical Study in Japan On January 11, 2023, Cardiovascular Systems, Inc., a medical device company developing and commercializing innovative interventional treatment systems for patients suffering from peripheral and coronary artery disease, announced the initiation of the...
Read More...
Jan 18, 2023
How Are Innovations in Drug Delivery Devices Solving the Complex Challenges in the Market?
Drug delivery is one of the highly evolving topics in the healthcare industry. Over the past few decades, there has been enormous growth in the drug delivery devices market owing to the active participation of the global MedTech giants, the rise in investments, and technological advancements in the development of n...
Read More...
Dec 29, 2022
PAVmed-Novosound’s Partnership; J&J Acquires Abiomed; Approval to Sysmex’s Assay Kit to Identify Amyloid Beta (Aβ) Accumulation; SI-BONE Announced FDA Clearance for Expanded Rod Compatibility with the iFuse Bedrock Granite Implant System; EOFlow’s Wearable Insulin Pump ‘EOPatch’; Icecure’s ProSense System, a Cryoablation Therapy
PAVmed Entered into a Partnership with Novosound for Ultrasound Imaging Technology On December 22, 2022, PAVmed Inc., a diversified commercial-stage medical technology company, operating in the medical device, diagnostics, and digital health sectors, announced that the company has entered into a partnership with...
Read More...
Dec 28, 2022
How is Active Implantable Medical Devices Market Evolving?
Over the past few years, the growth and development in electronics, micro and nano-fabrication, and wireless technology have significantly enhanced the quality and effectiveness of healthcare delivery systems, and treatment outcomes and led to various insights in the biological investigations through research and d...
Read More...
Dec 22, 2022
Medtronic’s Hugo Robotic-Assisted Surgery System Trial; Insightec’s Pivotal LIBERATE Clinical Trial; FDA Granted EMA for Thermo Fisher Scientific’s Monkeypox Test; Life Spine’s Trulift Lateral Expandable Spacer System & Lateral Pate System; Fujifilm & Inspirata Announces Asset Purchase Agreement; Abbott Launched SCS System for Chronic Pain
Medtronic Announced First Patient Enrolment for Hugo™ Robotic-Assisted Surgery System in US Clinical Trial On December 15, 2022, Medtronic, a global healthcare technology leader, announced that the first patient was enrolled in the Expand URO US clinical trial for the Hugo™ robotic-assisted surgery...
Read More...
Dec 15, 2022
BioGX’s ‘pixl’ Portable qPCR Platform; Stryker’s Citrefix Suture Anchor System; Boston Scientific to Acquire Majority Stake of Acotec Scientific; FDA Approved the Dexcom G7 CGM System; Agilent’s NSCLC Liquid Biopsy Companion Diagnostic Test Approval; ClearPoint Neuro’s Prism™ Neuro Laser Therapy
BioGX Announced Ex-US Commercial Launch of ‘pixl™’ Portable qPCR Platform On December 8, 2022, BioGX, a global provider of easy molecular diagnostic solutions, announced the Ex-US commercial release of BioGX's portable pixl™ qPCR real-time PCR platform for use with BioGX CE-IVD approved assays.&nbs...
Read More...







-Agonist.png)

