Small Lymphocytic Lymphoma
Dec 11, 2023
Eli’s Jaypirca: A Beacon of Success in the BTK Inhibitor Market
As the battle intensifies among AbbVie, Johnson & Johnson, AstraZeneca, and BeiGene in the BTK inhibitor market, Eli Lilly stands out by forging a new direction in the landscape of blood cancer drugs. Just at the beginning of this month, the FDA granted accelerated approval to Lilly’s Jaypirca. This approval is...
Read More...
Jan 24, 2023
BeiGene’s Brukinsa Approval; FDA Approval to Seagen’s TUKYSA; NICE Recommends Alnylam’s Amvuttra; FDA Approves Brenzavvy for Type 2 Diabetes; Roche’s Tecentriq to be Filed for Early-stage Liver Cancer; FDA Lifts Hold on Astellas’ Pompe Gene Therapy
FDA Approves BeiGene’s Brukinsa for CLL/SLL BeiGene's Brukinsa (zanubrutinib) for chronic lymphocytic leukemia (CLL) and small lymphocytic lymphoma (SLL) has been approved by the US Food and Drug Administration. CLL is a common type of leukemia, accounting for approximately 25% of all new cases each year. SLL is...
Read More...
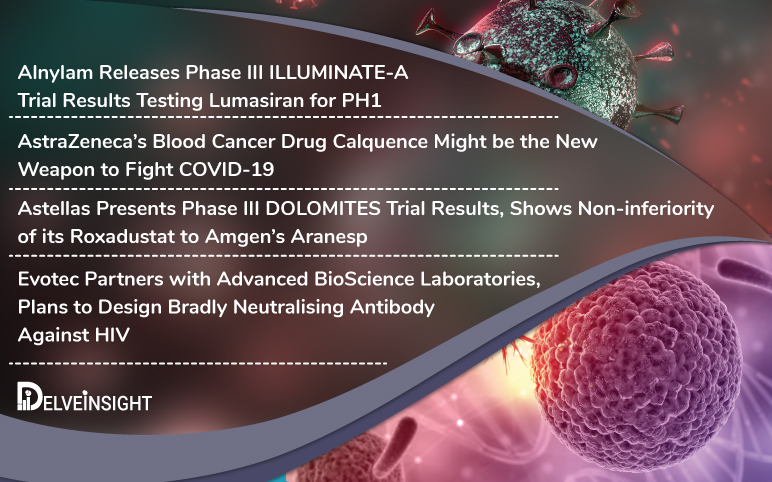
Jun 09, 2020
Alnylam’s lumasiran results; AZ’s oncology drug; Astellas Roxadustat; Evotec’s partnership with ABL
Alnylam Pharmaceuticals has released the Phase III results of the clinical trial ILLUMINATE-A evaluating lumasiran for Primary Hyperoxaluria Type 1 Primary Hyperoxaluria Type 1 (PH1) is a rare disorder that affects kidneys due to an excess buildup of oxalate, which in normal cases is filtered through the kidneys...
Read More...

-Agonist.png)

