The Current Scenario for Interstitial Cystitis(IC)
Mar 27, 2017
Interstitial cystitis (IC), also known as painful bladder syndrome, is a chronic disease that results in recurring discomfort or pain in the bladder and the surrounding pelvic region. This is considered as multiple bladder conditions as it can lead to many other problems. Interstitial cystitis can be of two types: Non-Ulcerative (90% of IC cases) and Ulcerative (5%-10% of IC cases), where the exact cause is unknown. However, researchers believe that many factors are at play which may contribute to the development of the condition like Bladder trauma, Bladder over-distention, pelvic floor muscle dysfunction, and bacterial infection (cystitis), etc. Damage to the bladder wall allows particles in the urine, such as potassium, to leak into the bladder lining.
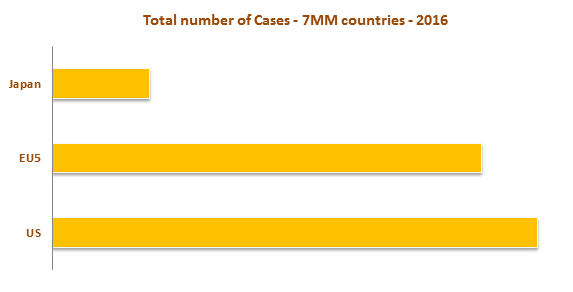
The diagnosis of interstitial cystitis can be challenging as the patient poses a variety of symptoms that may be difficult to diagnose and may also lead to misdiagnosis, underdiagnosis and delayed diagnosis. United States has the highest incident probable IC Cases. 90% of IC cases occur in women. Though only 10% of IC cases occur in men, it is believed that is underestimated due to misdiagnosis.
Downloads
Click Here To Get the Article in PDF
Recent Articles
- Transthyretin Amyloidosis (ATTR) Competitive Landscape, Market Insights, Epidemiology and Market
- mTOR Inhibitors-Emerging therapy in various therapeutic areas
- Progressive Supranuclear Palsy– A neurodegenerative disorder
- Vitiligo – High Unmet Need Indeed!
- Urinary Incontinence: Epidemiology Insight on Leaking Bladder
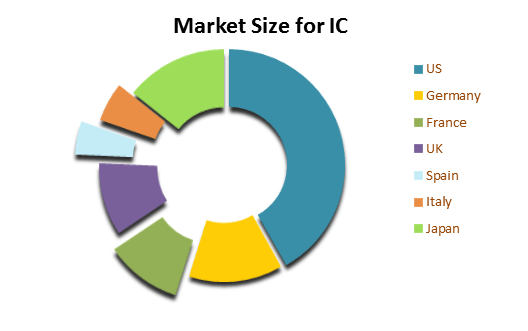
The Interstitial Cystitis market is mainly driven by Elmiron, which is the only approved oral drug by FDA specifically for IC. Another option includes bladder instillations like Cystistat, Hyacyst, Uracyst, etc., which are not approved by FDA. NSAIDs, Anti-depressants, anti-histamines, etc are also used for controlling symptoms of IC. The overall market of IC in 7 major markets for Elmiron and bladder instillations was found to be around USD 261.3 millions, of which major share is of U.S.
With drugs in the pipeline for IC, the market is expected to grow at a steady rate. There is no cure for interstitial cystitis (IC). However, many treatment options are available to help relieve the symptoms of bladder pain, urgency, and frequency. The combination of various treatments is the best approach to treating interstitial cystitis.
Insight by:
Anuj Thakur
Analyst
DelveInsight is a leading Business Consulting and Market Research Firm. We help our clients to find answers relevant to their business, facilitating their decision-making. DelveInsight also serves as a knowledge partner for business strategy and market research. We provide comprehensive analytical reports across various therapeutic indications. DelveInsight has a database of 3000+ high-quality analytical reports.
Downloads
Article in PDF
Recent Articles
- TRAIL Receptor Agonists: Emerging target therapies against Cancer
- Bio-manufacturing with Spider Silk for pre-term babies
- Interstitial Cystitis: A big opportunities for emerging drugs
- Colony Stimulating Factor 1 Receptor (CSF1R) and Its connection with Memory Retention
- Biohaven’s trial ; GSK integrates; FDA Approval; Phase III trial