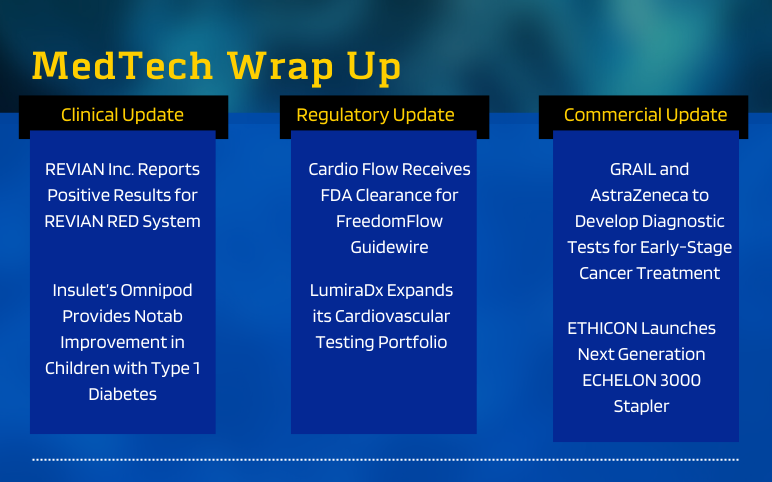
Aug 30, 2021
Venous thromboembolism (VTE) is a condition that is characterized by the development of blood clots in the veins. The condition includes Deep vein thrombosis (DVT) which occurs when a blood clot forms in a deep vein of the lower legs or pelvic area. However, in some cases, the blood clots from pelvic areas may break loose and travel to the lungs through the right side of the heart, known as Pulmonary embolism (PE).
According to the CDC, the condition affects about 900,000 people (1 to 2 per 1,000) each year in the United States, however, the exact number is unknown; with around 60,000-100,000 deaths. Moreover, one-third to one-half of the affected Deep vein thrombosis patients develop long-term complications due to damages in the valves due to clots, referred to as post-thrombotic syndrome (PTS). Venous thromboembolism can occur at any age; however, the most commonly diagnosed patient pool falls in the age group of 60 and above. Further, people who are overweight, have undergone major orthopedic surgeries, or are cancer patients are at a higher risk of developing the condition. Besides, hospitalization is one of the major risk factors for the development of VTE. To give it a number, up to 60% of all the VTE cases occur during or within 90 days of hospitalization, which can be prevented given, proper precautionary measures are taken.
The condition gets progressively worse, yet largely remains underdocumented, misdiagnosed and undertreated owing to the asymptomatic nature of the condition, unclear etiology, and lack of proper screening. However, the condition is preventable. As per the analyses, as many as 70% of the VTE cases are preventable with the help of proper prophylactic treatment, however, the number who received prophylaxis treatment remains considerably low.
Click Here To Get the Article in PDF
The Venous thromboembolism treatment market offers approaches that aim to break the clots and prevent their further development. It majorly comprises blood-thinning agents such as Medication (Anticoagulants, Thrombolytic Agents, Factor Xa Inhibitors, Thrombin Inhibitors). In advanced stages of VTE, available treatment approaches involve Mechanical devices (compression stockings), and Surgical treatment (Pulmonary embolectomy, Vena caval interruption).
However, the available treatment options are often associated with severe side effects. Furthermore, approved anti-coagulants such as (heparin, warfarin, enoxaparin, dalteparin) are not approved to be sued for an extended duration in patients who are at high risk. Thus, there is a need for a new generation of novel anticoagulants (NOACs) that are safe, can be used for the longer term, and are associated with fewer side effects. These include rapid onset and offset of action, predictable pharmacokinetics, and fewer drug interactions, which eliminate the need for regular coagulation monitoring associated with conventional anticoagulant therapy.
To meet the unmet needs in the Venous thromboembolism therapy market, several pharmaceutical companies including Tetherex Pharmaceuticals, Ono Pharmaceutical, Quercis Pharma, Verseon Corporation, Velox Therapeutics, among several others are developing innovative therapies including monoclonal antibodies, NOACs, and so on.
| Therapy | Company | Phase |
| SelK2 | Tetherex Pharmaceuticals | Phase II |
| ONO-7684 | Ono Pharmaceutical | Phase I |
| Isoquercetin | Quercis Pharma | Phase III |
| VE-1902 | Verseon Corporation | – |
| – | Velox Therapeutics | Preclinical |
DelveInsight estimates that with the major influx of pharmaceutical companies, the launch of emerging therapies and expected to rise in the prevalent cases, the Venous thromboembolism market is expected to witness an exponential boom in the forecasted period 2021-30. The launch of novel anticoagulants shall offer a major pharmacological benefit over the available ones in terms of safety, tolerability, and effectiveness.

Aug 30, 2021
Venous thromboembolism (VTE) is a condition that is characterized by the development of blood clots in the veins. The condition includes Deep vein thrombosis (DVT) which occurs when a blood clot forms in a deep vein of the lower legs or pelvic area. However, in some cases, the blood clots from pelvic areas may break loose and travel to the lungs through the right side of the heart, known as Pulmonary embolism (PE).
According to the CDC, the condition affects about 900,000 people (1 to 2 per 1,000) each year in the United States, however, the exact number is unknown; with around 60,000-100,000 deaths. Moreover, one-third to one-half of the affected Deep vein thrombosis patients develop long-term complications due to damages in the valves due to clots, referred to as post-thrombotic syndrome (PTS). Venous thromboembolism can occur at any age; however, the most commonly diagnosed patient pool falls in the age group of 60 and above. Further, people who are overweight, have undergone major orthopedic surgeries, or are cancer patients are at a higher risk of developing the condition. Besides, hospitalization is one of the major risk factors for the development of VTE. To give it a number, up to 60% of all the VTE cases occur during or within 90 days of hospitalization, which can be prevented given, proper precautionary measures are taken.
The condition gets progressively worse, yet largely remains underdocumented, misdiagnosed and undertreated owing to the asymptomatic nature of the condition, unclear etiology, and lack of proper screening. However, the condition is preventable. As per the analyses, as many as 70% of the VTE cases are preventable with the help of proper prophylactic treatment, however, the number who received prophylaxis treatment remains considerably low.
The Venous thromboembolism treatment market offers approaches that aim to break the clots and prevent their further development. It majorly comprises blood-thinning agents such as Medication (Anticoagulants, Thrombolytic Agents, Factor Xa Inhibitors, Thrombin Inhibitors). In advanced stages of VTE, available treatment approaches involve Mechanical devices (compression stockings), and Surgical treatment (Pulmonary embolectomy, Vena caval interruption).
However, the available treatment options are often associated with severe side effects. Furthermore, approved anti-coagulants such as (heparin, warfarin, enoxaparin, dalteparin) are not approved to be sued for an extended duration in patients who are at high risk. Thus, there is a need for a new generation of novel anticoagulants (NOACs) that are safe, can be used for the longer term, and are associated with fewer side effects. These include rapid onset and offset of action, predictable pharmacokinetics, and fewer drug interactions, which eliminate the need for regular coagulation monitoring associated with conventional anticoagulant therapy.
To meet the unmet needs in the Venous thromboembolism therapy market, several pharmaceutical companies including Tetherex Pharmaceuticals, Ono Pharmaceutical, Quercis Pharma, Verseon Corporation, Velox Therapeutics, among several others are developing innovative therapies including monoclonal antibodies, NOACs, and so on.
| Therapy | Company | Phase |
| SelK2 | Tetherex Pharmaceuticals | Phase II |
| ONO-7684 | Ono Pharmaceutical | Phase I |
| Isoquercetin | Quercis Pharma | Phase III |
| VE-1902 | Verseon Corporation | – |
| – | Velox Therapeutics | Preclinical |
DelveInsight estimates that with the major influx of pharmaceutical companies, the launch of emerging therapies and expected to rise in the prevalent cases, the Venous thromboembolism market is expected to witness an exponential boom in the forecasted period 2021-30. The launch of novel anticoagulants shall offer a major pharmacological benefit over the available ones in terms of safety, tolerability, and effectiveness.
