Emerging TIL Therapies Poised to Broaden the Landscape Beyond AMTAGVI
Mar 06, 2026
Summary
- Iovance’s AMTAGVI has become the first personalized TIL therapy to receive regulatory clearance, gaining accelerated FDA approval for use in advanced melanoma following PD-1 inhibitor treatment.
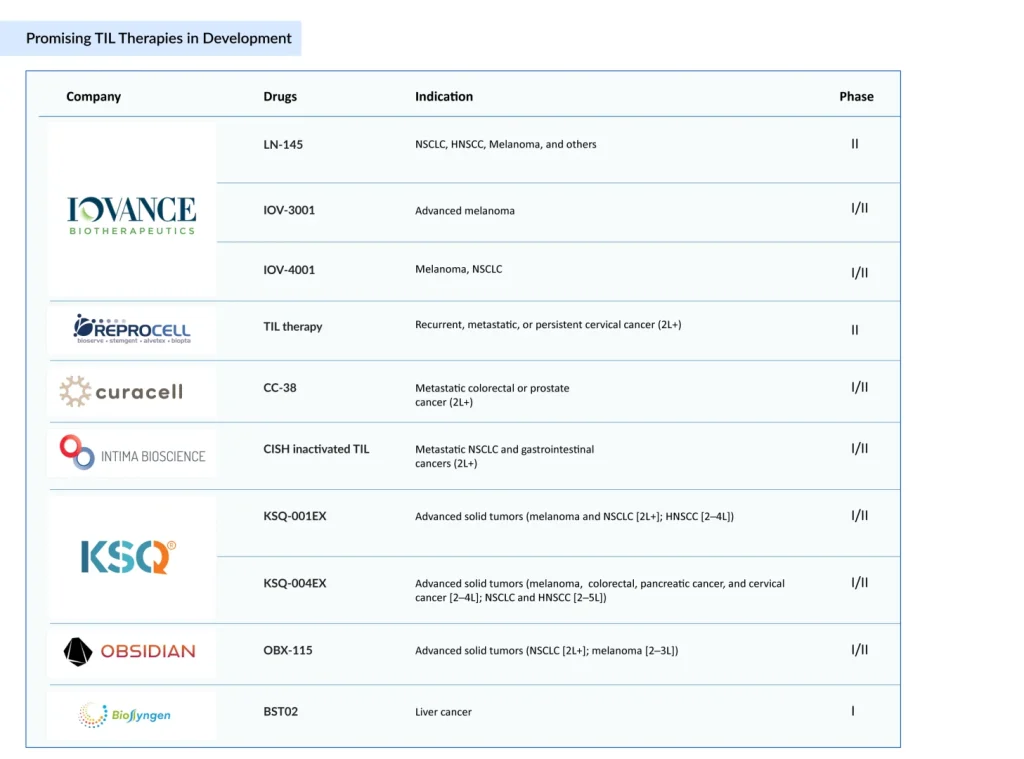
- AMTAGVI approval has sparked intensified activity across the field, with companies such as Obsidian Therapeutics (OBX-115), KSQ Therapeutics (KSQ-001EX, KSQ-004EX), Curacell Holding (CC-38), and Intima Bioscience (CISH-inactivated TIL), among others, advancing their own next-generation TIL candidates.
- In addition to these therapies, Iovance is broadening its TIL pipeline with candidates such as LN-145, IOV-4001, and IOV-3001, while investigating combination treatments and expanding into new indications like NSCLC and endometrial cancer.
The origins of TIL therapy trace back to the 1980s, when Steven Rosenberg first characterized LAK cells and tumor-infiltrating lymphocytes. By 1986, IL-2–expanded TILs had demonstrated markedly improved tumor regression in early preclinical studies.
Iovance Biotherapeutics’ AMTAGVI (lifileucel) has since emerged as a milestone in oncology, becoming the first T-cell therapy for a solid tumor to receive authorization from the FDA. This personalized, single-administration TIL treatment is indicated for adults with unresectable or metastatic melanoma who have already undergone standard first-line PD-1 blockade and, when applicable, targeted therapy with a BRAF or BRAF/MEK inhibitor for BRAF-V600–mutant disease, addressing a population with substantial unmet need in later treatment lines. Its accelerated approval in February 2024 provided AMTAGVI with early market leadership, contributing to strong initial adoption and positive revenue trends expected to continue beyond 2024.
Downloads
Click Here To Get the Article in PDF
Sadaf Javed, Manager of Forecasting and Analytics at DelveInsight, said that AMTAGVI’s uptake is highly dependent on expanding the authorized treatment centers network; insufficient center onboarding could slow adoption and revenue growth.
Following AMTAGVI approval, competitive activity has expanded, with Obsidian Therapeutics (OBX-115), KSQ Therapeutics (KSQ-001EX and KSQ-004EX), Curacell Holding (CC-38), Intima Bioscience (CISH inactivated TIL), and others progressing next-generation TIL candidates.
Alongside these programs, Iovance continues to expand its own TIL portfolio with candidates like LN‑145, IOV‑4001, and IOV-3001, exploring combination strategies and new indications such as NSCLC and endometrial cancer.
Let’s dive deeper into the promising TIL-based therapies under development for solid tumors.
Iovance Biotherapeutics’ LN‑145, IOV‑4001, and IOV-3001
LN-145 is a form of autologous TIL therapy, which works by activating the patient’s own white blood cells to attack cancer cells. To develop LN-145, doctors extract T-cells from the patient’s tumor and expand them in the lab. LN-145-S1 is produced through an adjusted manufacturing process that selects a specific subset of these T-cells. These cells may be able to identify, target, and destroy tumor cells.
In November 2025, Iovance (NASDAQ: IOVA) reported that it anticipates completing enrollment for the IOV-LUN-202 study in 2026, aiming to support a supplemental BLA submission for lifileucel in nonsquamous NSCLC, with a possible market launch in 2027. That same month, the company also announced that early data from the IOV-END-201 trial, evaluating lifileucel in previously treated advanced endometrial cancer, is expected in early 2026.
IOV-4001 uses gene-editing TALEN technology licensed from Cellectis (EPA: ALCLS). This technology disables the gene responsible for producing the PD-1 protein. By removing this key obstacle that limits T-cell activity against tumors, IOV-4001 may represent a more advanced, next-generation TIL therapy for various solid tumors. The Phase 2 efficacy stage of the IOV-GM1-201 study is ongoing, enrolling patients with previously treated advanced melanoma and NSCLC to assess the treatment’s effectiveness.
IOV-3001 is a redesigned IL-2 CDR-grafted molecule that selectively targets cells expressing IL2R beta-gamma while reducing activation of Tregs that depend on IL2R alpha-beta-gamma. Its optimized half-life increases overall exposure and may lower peak concentrations, potentially decreasing IL-2–related side effects. Dose escalation for IOV-3001 is ongoing. In preclinical testing, the agent showed promising improvements in safety, less frequent dosing needs, and robust effector T-cell expansion. Phase 2 development is anticipated to begin in 2026.
REPROCELL’s TIL therapy
TIL therapy is a form of adoptive immunotherapy where TILs are extracted from a patient’s tumor, expanded to large numbers in the lab, and then infused back into the patient. In June 2023, REPROCELL (TYO: 4978) announced a joint research agreement with Keio University to transfer technology for producing TILs for Advanced Medicine B, a Phase II clinical study testing TIL therapy combined with low-dose interleukin-2 in patients with cervical cancer.
KSQ Therapeutics’ KSQ-001EX and KSQ-004EX
KSQ Therapeutics is developing two advanced TIL-based treatments, KSQ-001EX and KSQ-004EX, aimed at multiple solid tumors. Each therapy uses a distinct mechanism intended to boost effectiveness, expand the types of tumors that can be addressed, and support longer-lasting responses.
KSQ-001EX is an experimental eTIL product created with the company’s ExPRESS manufacturing method, which disables the SOCS1 gene through CRISPR/Cas9 editing. KSQ’s Immune CRISPRomics platform pinpointed SOCS1 as a key regulator that limits TIL anti-tumor activity, engraftment, and persistence. The FDA has authorized an Investigational New Drug (IND) application enabling a Phase 1/2 trial of KSQ-001EX.
KSQ-004EX is another investigational eTIL therapy, featuring a dual edit that knocks out both Regnase-1 and SOCS1 using CRISPR/Cas9, and is also produced with the ExPRESS process. Through the company’s in vivo CRISPR2 screening method, this gene pair emerged as the most promising dual-edit combination for maximizing anti-tumor activity. The FDA has likewise cleared an IND application for a Phase 1/2 clinical study of KSQ-004EX.
According to Sadaf, if clinical results are positive, these therapies could compete with other TIL programs by offering superior efficacy, applicability across more tumor types, and potentially longer-lasting responses.
Obsidian Therapeutics’ OBX-115
OBX-115 is an advanced TIL therapy created using Obsidian’s cytoDRIVE platform. These TILs are genetically modified to produce membrane-bound interleukin-15 (mbIL-15), which enhances T-cell expansion, durability, and antitumor function, while removing the need for the high-dose IL-2 typically responsible for significant toxicity in standard TIL treatments. OBX-115 has been granted both Fast Track Designation and Regenerative Medicine Advanced Therapy status by the US FDA.
In June 2025, Obsidian Therapeutics released early Phase I safety and efficacy results from the Phase I/II Agni-01 multicenter trial evaluating OBX-115, an engineered, tumor-derived autologous TIL product featuring a pharmacologically controlled mbIL-15 construct, in patients with advanced or metastatic melanoma that no longer responds to immune checkpoint inhibitors. These findings were later presented at the 2025 ASCO annual meeting.
Biosyngen’s BST02
BST02 is an innovative adoptive cell therapy that utilizes a patient’s own expanded TILs. It relies on growing TILs harvested from the patient without the need for high-dose interleukin-2. Its capacity for cryopreservation marks a major step forward for TIL-based treatments, removing many travel and logistical hurdles.
In May 2025, Biosyngen reported that results from the Phase I study of BST02 would be presented at ASCO 2025. In February 2024, the company announced that BST02 had been granted Fast Track Designation by the US FDA for treating liver cancers, including hepatocellular carcinoma and cholangiocarcinoma.
Intima Bioscience’s CISH inactivated TIL
The cytokine-inducible SH2-containing protein (CISH) acts as an intracellular immune checkpoint in cancer, dampening T-cell receptor (TCR) signaling and reducing recognition of tumor neoantigens. By interacting with PLC-γ1, an early signaling component of the TCR pathway, CISH suppresses antigen-driven cytokine production and limits T-cell proliferation. Blocking CISH in T cells is thought to counteract tumor immune escape across cancer types, independent of PD-L1 status. In May 2025, Intima Bioscience reported first-in-human results using CRISPR-mediated CISH knockout in T cells given to patients with metastatic colorectal cancer at the 2025 American Association for Cancer Research Annual Meeting.
Curacell Holding’s CC-38
CuraCell’s first-generation therapy is a TIL product enhanced for stronger cytotoxic activity using CytoPLY. When paired with a proprietary dosing schedule and checkpoint inhibition, the treatment delivers a synergistic and durable effect while requiring a gentler pre-treatment process for patients. Early patient-specific applications of CC-38 have yielded encouraging outcomes, with both complete and partial responses observed in a varied population of cold solid-tumor patients, highlighting the therapy’s strength and wide potential use.
In its initial clinical program, CC-38 is being developed for advanced prostate and colorectal cancers, areas with significant unmet medical needs. In August 2025, CuraCell announced that the German regulatory body Paul-Ehrlich-Institut had approved its Clinical Trial Application, allowing the launch of a Phase I/IIa trial to evaluate the autologous TIL therapy CC-38 in patients with metastatic colorectal and metastatic prostate cancer.
In summary, the accelerating clinical progress of next-generation TIL programs, from Iovance Biotherapeutics and its advancing LN-145, IOV-4001, and IOV-3001 portfolio to emerging platforms from REPROCELL, KSQ Therapeutics, Obsidian Therapeutics, Biosyngen, Intima Bioscience, and Curacell Holding, signals a turning point for solid-tumor immunotherapy.
Collectively, these therapies are pushing TIL therapy treatment beyond its traditional limitations by incorporating gene editing, cytokine engineering, next-generation IL-2 alternatives, improved manufacturing, and more durable cell-product designs. As programs expand into Phase I/II and Phase II studies across melanoma, NSCLC, endometrial, cervical, colorectal, liver, and prostate cancers, the field is poised for broader applicability, reduced toxicity, and more convenient treatment logistics.
If current trials read out positively, this wave of engineered and optimized TIL therapies could substantially reshape the competitive landscape, challenging existing autologous and allogeneic cell therapies, accelerating regulatory momentum, and ultimately enabling TILs to become a mainstream modality for cancers that have been historically resistant to immunotherapy.

Downloads
Article in PDF