Defibrillators at the Heart of Cardiac Care: Challenges, Innovations, and the Road Ahead
Mar 18, 2026
Table of Contents
Defibrillators are critical medical devices designed to correct life-threatening cardiac arrhythmias, particularly ventricular fibrillation and ventricular tachycardia, which can rapidly lead to sudden cardiac arrest (SCA). By delivering a controlled electrical shock to the heart, defibrillators aim to restore a normal cardiac rhythm and prevent irreversible organ damage or death. According to the World Health Organization (WHO), cardiovascular diseases remain the leading cause of mortality globally, accounting for nearly 17.9 million deaths annually, with sudden cardiac arrest being a major contributor within this burden. Early access to defibrillation is therefore recognized as one of the most effective interventions for improving survival outcomes in cardiac emergencies.
Over the past few decades, defibrillator technology has evolved significantly, transitioning from bulky hospital-based equipment to portable, automated, implantable, and wearable systems. Regulatory agencies such as the FDA have played a central role in ensuring safety, performance, and quality standards for these devices, while global public-health initiatives increasingly emphasize public access to defibrillation.
Downloads
Click Here To Get the Article in PDF
Recent Articles
- Understanding How Cardiac Devices are Aiding in Treatment and Prevention of Heart Disease
- Tepezza receives approval; a new way to treat Alzheimer’s
- Samsung Partnered with Lunit; Boston Scientific Launched VersaVue single-use Flexible Cystoscope;...
- Miracor Medical’s Picso Pivotal Study; Medtronic’s Extravascular ICD; Galaxy Medical’s CENTAURI P...
- LiviWell Receives FDA Clearance for Livi, Pioneering Post-Intercourse Vaginal Care; Medtronic Adv...
Major Types of Defibrillators
Defibrillators can be classified based on their mode of use, level of automation, and intended patient population. Each category serves a distinct clinical or public-health purpose.
Automated External Defibrillators (AEDs)
Automated external defibrillators are portable, user-friendly devices developed for rapid response to sudden cardiac arrest outside of hospital settings. AEDs are designed to be used by individuals with minimal or no medical training, making them suitable for deployment in public places such as airports, schools, offices, railway stations, and sports facilities. The FDA recognizes AEDs as essential emergency devices that analyze heart rhythm automatically and advise or deliver a shock only when clinically indicated.
AEDs operate through adhesive electrode pads placed on the patient’s chest. Once activated, the device analyzes the cardiac rhythm and provides clear voice and visual instructions to guide the rescuer through each step. This automation minimizes human error and reduces hesitation during high-stress situations. From a public-health perspective, the WHO and other health authorities emphasize that early defibrillation within the first few minutes of cardiac arrest significantly improves survival rates.
The AED segment represents one of the fastest-growing areas within the defibrillator market. The automated external defibrillator market is projected to expand at a CAGR of 8.62% between 2026 and 2034. This notable growth is largely driven by the rising prevalence of chronic conditions, particularly cardiovascular diseases such as atrial fibrillation and coronary artery disease (CAD), along with ongoing product innovation by key industry players. Additionally, the growing elderly population is expected to further accelerate the overall expansion of the AED market throughout the forecast period.
Manual and Professional Defibrillators
Manual defibrillators are primarily used in hospitals, emergency departments, intensive care units, and ambulances. Unlike AEDs, these devices require trained healthcare professionals to interpret electrocardiogram (ECG) readings, determine shock timing, and select appropriate energy levels. The FDA categorizes these devices as professional-use medical equipment, subject to stringent regulatory oversight.
Manual defibrillators are often integrated with advanced monitoring systems, enabling continuous ECG display, oxygen saturation measurement, and blood pressure monitoring. Their versatility makes them indispensable in advanced cardiac life support (ACLS) settings, where clinical judgment and rapid decision-making are critical. Although this segment does not experience the same volume-driven growth as AEDs, it remains fundamental to acute care infrastructure worldwide.
Implantable Cardioverter-Defibrillators (ICDs)
Implantable Cardioverter-Defibrillators are surgically implanted devices designed for patients at high risk of sudden cardiac death due to underlying heart conditions. ICDs continuously monitor heart rhythm and automatically deliver electrical therapy when they detect life-threatening arrhythmias. According to the U.S. National Institutes of Health (NIH), ICDs have been shown to significantly reduce mortality in patients with prior ventricular arrhythmias or severe cardiomyopathy.
Modern ICDs combine defibrillation and pacing functions, allowing them to correct both dangerously fast and abnormally slow heart rhythms. The FDA closely regulates ICDs as high-risk medical devices, requiring rigorous clinical evaluation before approval.
The ICD market continues to expand steadily, supported by increasing diagnosis rates of cardiac arrhythmias, technological refinement, and broader clinical guidelines recommending preventive implantation in high-risk patients. Innovations such as subcutaneous ICDs, which avoid placing leads inside the heart, are also gaining attention due to reduced procedural complications.
Wearable and Extravascular Systems
Wearable cardioverter-defibrillators and emerging extravascular systems represent an intermediate solution between external and implantable devices. Wearable defibrillators are typically prescribed for patients who are temporarily at risk of sudden cardiac arrest, such as those awaiting ICD implantation or recovering from certain cardiac events. These devices continuously monitor heart rhythm and deliver therapy if a dangerous arrhythmia occurs.
From a regulatory standpoint, the FDA recognizes wearable defibrillators as important options for short-term risk management. DelveInsight reports suggest that while this segment is smaller in size compared to AEDs and ICDs, it is gaining momentum due to growing emphasis on personalized and transitional cardiac care.
Advantages of Defibrillators
The primary advantage of defibrillators lies in their ability to save lives by rapidly restoring normal heart rhythm during cardiac emergencies. According to the FDA and WHO, early defibrillation is one of the most effective interventions for sudden cardiac arrest, particularly when combined with cardiopulmonary resuscitation (CPR). Survival rates decline significantly with each passing minute without defibrillation, underscoring the importance of rapid device access.
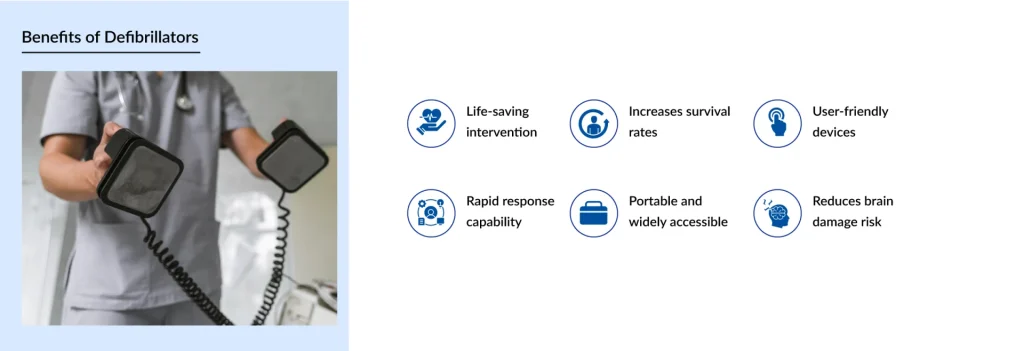
Defibrillators also enhance long-term patient outcomes by reducing the risk of recurrent fatal arrhythmias. Implantable systems, in particular, provide continuous protection for high-risk individuals, offering peace of mind and measurable reductions in mortality. From a healthcare system perspective, these devices help reduce the burden of emergency interventions and intensive care admissions by preventing catastrophic cardiac events.
Technological advancements further enhance these benefits. Automated rhythm analysis, improved energy delivery algorithms, and device reliability have collectively improved safety and effectiveness. Government health agencies consistently emphasize that widespread defibrillator availability and proper regulatory oversight are central to improving population-level cardiac outcomes.
Challenges with Defibrillators
Despite their clinical value, defibrillators face several challenges. Cost remains a major barrier, particularly for implantable and advanced wearable systems. High device prices, surgical expenses, and ongoing maintenance requirements can limit access in low- and middle-income regions. The WHO has repeatedly highlighted disparities in access to life-saving medical technologies, including defibrillators, across different healthcare systems.
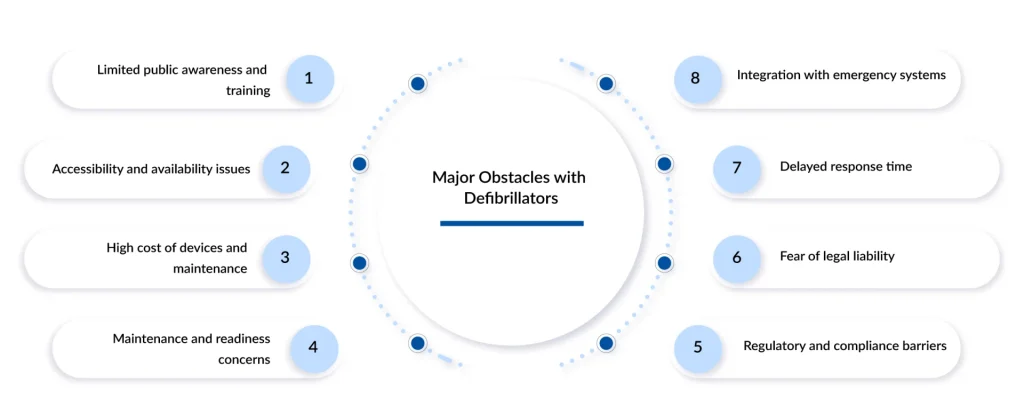
Another challenge involves awareness and training. While AEDs are designed for ease of use, their effectiveness depends on bystander willingness and confidence to act during emergencies. Government health agencies continue to stress the need for public education, standardized training programs, and clear emergency response protocols.
Regulatory complexity also presents hurdles, especially as devices become more technologically sophisticated. Ensuring cybersecurity, data privacy, and long-term device reliability requires ongoing collaboration between manufacturers and regulatory authorities such as the FDA.
Future Outlook
The global defibrillator market is expected to maintain steady growth at a CAGR of ~3% by 2034, driven by rising cardiovascular disease prevalence, aging populations, and increasing emphasis on preventive cardiac care. AEDs and ICDs remain the dominant segments, while wearable and extravascular systems represent emerging growth areas.
Several global medical device manufacturers are at the forefront of defibrillator innovation and commercialization, shaping both clinical practice and market evolution. Companies such as Medtronic, Abbott, and Boston Scientific are widely recognized as leaders in implantable cardioverter-defibrillator technologies, supported by extensive product portfolios and long-standing clinical evidence.
In the external and public-access defibrillation space, organizations including Koninklijke Philips N.V., ZOLL Medical Corporation, and Stryker play a central role, particularly in advancing automated external defibrillators and professional emergency response systems. At the same time, manufacturers such as Biotronik, Nihon Kohden Corporation, and BPL Medical Technologies contribute diversified defibrillation solutions across regional and global markets. Collectively, these companies invest heavily in research and development, focusing on innovations in battery longevity, rhythm-analysis algorithms, advanced materials, device connectivity, and patient-centered design, thereby continuously enhancing the safety, reliability, and clinical effectiveness of defibrillator technologies.
From a public-health standpoint, the future of defibrillators lies in expanded access, improved affordability, and deeper integration with healthcare systems. Governments and regulatory bodies are increasingly advocating for wider public access to defibrillation programs, particularly in high-traffic and high-risk environments. Technological innovation, supported by regulatory oversight, is expected to further enhance device effectiveness while reducing complications.
In conclusion, defibrillators remain indispensable tools in modern cardiovascular care. Their ability to prevent sudden cardiac death, combined with continuous technological and regulatory progress, positions them as central components of both emergency response and long-term cardiac management strategies worldwide.

Downloads
Article in PDF
Recent Articles
- Pacemakers: Analyzing the Market Dynamics and Major Developments
- MEDIT’s i700 Intraoral Scanner; Phantom Neuro and Blackrock’s Next-Generation Assistive Devices; ...
- Major Drugs Decisions for Cardiovascular Diseases to Watch Through 2022
- Tepezza receives approval; a new way to treat Alzheimer’s
- Samsung Partnered with Lunit; Boston Scientific Launched VersaVue single-use Flexible Cystoscope;...