J&J and Protagonist’s ICOTYDE Ushers in First-in-Class Oral IL-23 Therapy
Mar 23, 2026
Table of Contents
Summary
- Johnson & Johnson and Protagonist Therapeutics received U.S. FDA approval for ICOTYDE (icotrokinra), the first targeted oral IL-23 receptor peptide therapy for moderate-to-severe plaque psoriasis in adults and adolescents (12+ years, ≥40 kg).
- Once-daily 200 mg oral tablet blocks IL-23 signaling with high affinity; taken on an empty stomach before food for biologic-like efficacy without injections.
- Four pivotal trials (2,500 patients) showed ~70% achieving clear/almost clear skin (IGA 0/1) and 55% PASI 90 at Week 16; superior to SOTYKTU; safety similar to placebo (1.1% difference at Week 16, no new signals at Week 52).
Johnson & Johnson and Protagonist Therapeutics’ ICOTYDE (icotrokinra) approval marks a pivotal inflection point in plaque psoriasis treatment, marrying the power of IL‑23 pathway blockade with the simplicity of a once‑daily oral pill. Positioned as the first and only targeted oral IL‑23 receptor peptide therapy, ICOTYDE is set to redefine first‑line systemic care for patients long trapped in a cycle of inadequate topicals and compromise.
Downloads
Click Here To Get the Article in PDF
Recent Articles
- Revolutionizing Psoriasis Treatment: Advancements in the US Market
- Evaluating the Upcoming Drugs in Pipeline for Major Autoimmune Diseases
- FDA Approves PENBRAYA for Most Common Serogroups Causing Meningococcal Disease; BIMZELX Approved ...
- Taltz (Ixekizumab) in Treatment of Plaque psoriasis
- Amgen Announced the Result of its CodeBreak-200 Trial; FDA Clears Bristol-Myers Squibb’s Deucrava...
A New Chapter in Plaque Psoriasis
Plaque psoriasis remains one of the most visible and burdensome immune‑mediated diseases, affecting more than 8 million Americans and over 125 million people worldwide, with nearly one‑quarter of cases classified as moderate to severe. Beyond the hallmark inflamed, scaly plaques on the scalp, elbows, knees, torso, and high‑impact sites such as the genitals, hands, and feet, psoriasis erodes emotional health, relationships, and daily functioning.
For years, treatment conversations have been polarized: potent injectable biologics at one end and often‑inadequate topical therapies at the other, with a stubborn gap in between. International Psoriasis Council guidance now clearly recommends transitioning to systemic therapy when two cycles of four‑week topical regimens fail, underscoring how many patients require more than creams and ointments.
ICOTYDE: Targeted Science in a Daily Pill
ICOTYDE is an interleukin‑23 receptor antagonist and the first and only targeted oral peptide designed to precisely block IL‑23R, a key upstream driver of the inflammatory cascade in moderate‑to‑severe plaque psoriasis. By binding the IL‑23 receptor with high affinity and potently inhibiting IL‑23 signaling in human T cells, ICOTYDE aims to deliver biologic‑like disease control without needles.
The U.S. FDA has approved ICOTYDE for adults and pediatric patients 12 years and older who weigh at least 40 kg and are candidates for systemic therapy or phototherapy, directly addressing a broad segment of patients underserved by current care algorithms. Dosed as a single 200‑mg tablet once daily upon waking, on an empty stomach and at least 30 minutes before food, the regimen is intentionally built to integrate seamlessly into everyday routines.
ICOTYDE’s label rests on an unusually comprehensive Phase 3 program, ICONIC, spanning four pivotal studies and approximately 2,500 patients across adults and adolescents. The program simultaneously interrogated efficacy and safety in classic plaque disease, high‑impact sites such as scalp and genital psoriasis, and in duplicate head‑to‑head trials versus an active comparator.
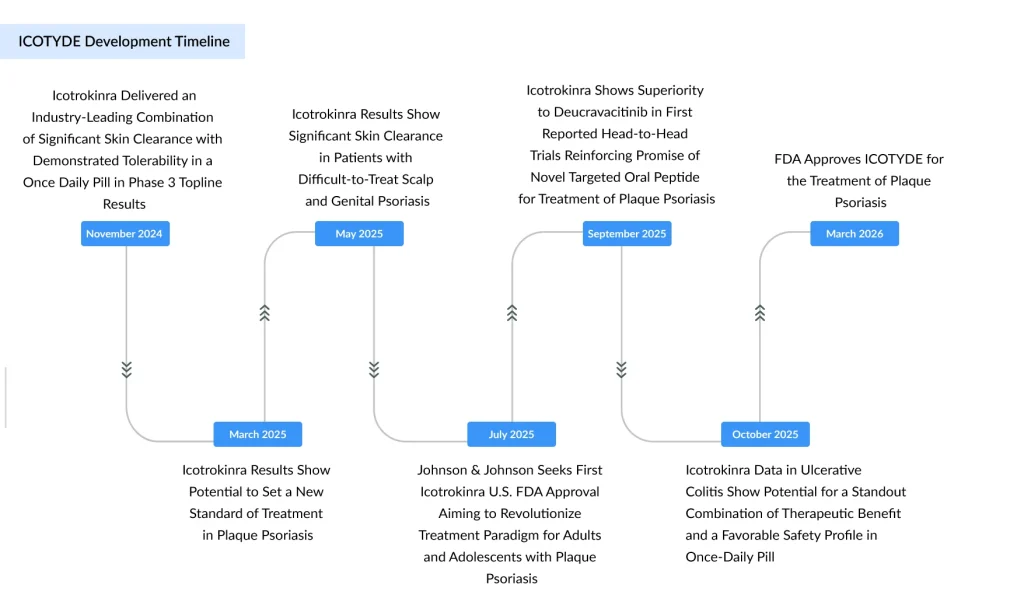
Across these Phase 3 studies, ICOTYDE met all primary endpoints and delivered robust skin clearance outcomes that push the boundaries of what clinicians expect from an oral agent. In head‑to‑head superiority trials, about 70% of patients achieved clear or almost clear skin (IGA 0/1), while 55% reached PASI 90 at Week 16, efficacy levels more typically associated with injectable biologics than with pills.
Equally critical is the safety story: rates of adverse reactions with ICOTYDE were within 1.1% of placebo through Week 16, and no new safety signals emerged through Week 52, supporting long‑term use in a chronic disease that demands durable control. This balance of deep efficacy and a favorable safety profile is a central pillar of how ICOTYDE is being positioned as a potential new standard in systemic psoriasis care.
Regulators have explicitly framed ICOTYDE as a first‑line systemic option for moderate‑to‑severe plaque psoriasis, opening the door for earlier, targeted intervention. The drug is also the first FDA‑approved daily peptide pill that targets the IL‑23 receptor and delivers complete skin clearance in plaque psoriasis, creating a new mechanistic and formulation class within the oral space.
That differentiation matters in a competitive landscape where Bristol Myers Squibb’s oral TYK2 inhibitor SOTYKTU previously enjoyed a multi‑year head start and has become a key growth driver. Data from ICONIC‑ADVANCE 1 and 2 showed ICOTYDE not only outperformed placebo but also demonstrated significant superiority to SOTYKTU in achieving high‑level clearance measures, positioning J&J’s pill as a formidable new entrant in the oral systemic segment.
ICOTYDE is the product of a long‑running strategic partnership between Johnson & Johnson and Protagonist Therapeutics that began in 2017, built around Protagonist’s proprietary peptide technology platform. Discovered jointly by scientists from both companies, ICOTYDE advanced through Phase 1 under Protagonist’s leadership, after which J&J assumed responsibility for late‑stage development and global commercialization.
The FDA green light triggers a $50 million milestone payment to Protagonist, with the potential for up to $580 million in additional regulatory and sales milestones and tiered royalties ranging from 6% to 10% on global net sales. For Protagonist, these economics and validation of its platform, alongside the pending FDA decision for rusfertide, signal the beginning of a multi‑year, product‑driven growth cycle.
For J&J, ICOTYDE is strategically timed as the company navigates loss of exclusivity for its immunology blockbuster STELARA and works to extend its leadership via newer assets like TREMFYA and now this first‑in‑class oral IL‑23R peptide. The approval reinforces J&J’s broader immunology vision: translating cutting‑edge science into accessible, patient‑centric therapies that can be deployed earlier in the disease journey.
Beyond Skin: Building an IL‑23‑Centric Franchise
While ICOTYDE’s first indication is moderate‑to‑severe plaque psoriasis, its development arc clearly signals ambitions that go far beyond the skin. Multiple Phase 3 ICONIC studies are underway in IL‑23‑driven conditions, including active psoriatic arthritis (ICONIC‑PsA 1 and 2), moderately‑to‑severely active ulcerative colitis (ICONIC‑UC), and moderately‑to‑severely active Crohn’s disease (ICONIC‑CD).
If successful, these programs could enable J&J and Protagonist to build a multi‑indication, oral IL‑23R franchise that complements existing injectable IL‑23 biologics while speaking directly to patients’ demand for less invasive, more convenient options. Such an evolution would not only expand the commercial footprint of ICOTYDE but also reinforce IL‑23 as a central axis in systemic inflammatory disease management.
Redesigning the Treatment Conversation
Perhaps the most profound impact of ICOTYDE will be on the everyday treatment decisions made in dermatology clinics. With a once‑daily targeted oral peptide now available as a first‑line systemic option for plaque psoriasis, the conversation can move from “How long can we stretch topicals?” to “How quickly can we achieve and sustain deep skin clearance with a regimen that fits the patient’s life?”
For patients who have long associated high‑efficacy psoriasis control with needles, infusion centers, or complex dosing schedules, ICOTYDE introduces a new proposition: biologic‑like outcomes with the familiarity of a pill and a safety profile that closely mirrors placebo over the first 16 weeks. For J&J and Protagonist, it is both a scientific milestone and a strategic beachhead, one that could reshape the systemic psoriasis landscape, and possibly the broader IL‑23‑driven disease space, for years to come.

Downloads
Article in PDF
Recent Articles
- Revolutionizing Psoriasis Treatment: Advancements in the US Market
- Plaque psoriasis drug outshines in phase III trial; Immunovant announces pause in trial of IMVT-1...
- Bristol-Myers Squibb’s Opdivo & Yervoy Combo Trial; Sarepta’s Gene Therapy SRP-9001 for DMD;...
- Taltz (Ixekizumab) in Treatment of Plaque psoriasis
- Amgen Announced the Result of its CodeBreak-200 Trial; FDA Clears Bristol-Myers Squibb’s Deucrava...




